Abstract
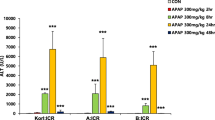
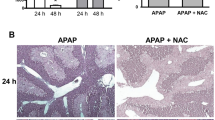
Acetaminophen (APAP) is known to cause a breach of the blood–bile barrier in mice that, via a mechanism called futile bile acid (BA) cycling, increases BA concentrations in hepatocytes above cytotoxic thresholds. Here, we compared this mechanism in mice and rats, because both species differ massively in their susceptibility to APAP and compared the results to available human data. Dose and time-dependent APAP experiments were performed in male C57BL6/N mice and Wistar rats. The time course of BA concentrations in liver tissue and in blood was analyzed by MALDI-MSI and LC–MS/MS. APAP and its derivatives were measured in the blood by LC–MS. APAP-induced liver damage was analyzed by histopathology, immunohistochemistry, and by clinical chemistry. In mice, a transient increase of BA in blood and in peri-central hepatocytes preceded hepatocyte death. The BA increase coincided with oxidative stress in liver tissue and a compromised morphology of bile canaliculi and immunohistochemically visualized tight junction proteins. Rats showed a reduced metabolic activation of APAP compared to mice. However, even at very high doses that caused cell death of hepatocytes, no increase of BA concentrations was observed neither in liver tissue nor in the blood. Correspondingly, no oxidative stress was detectable, and the morphology of bile canaliculi and tight junction proteins remained unaltered. In conclusion, different mechanisms cause cell death in rats and mice, whereby oxidative stress and a breach of the blood–bile barrier are seen only in mice. Since transient cholestasis also occurs in human patients with APAP overdose, mice are a clinically relevant species to study APAP hepatotoxicity but not rats.







Similar content being viewed by others
Data availability
All data are included in the manuscript.
References
Adams KJ, Pratt B, Bose N et al (2020) Skyline for small molecules: a unifying software package for quantitative metabolomics. J Proteome Res 19(4):1447–1458. https://doi.org/10.1021/acs.jproteome.9b00640
Bernal W, Wendon J (2013) Acute liver failure. N Engl J Med 369(26):2525–2534. https://doi.org/10.1056/NEJMra1208937
Cohen SD, Pumford NR, Khairallah EA et al (1997) Selective protein covalent binding and target organ toxicity. Toxicol Appl Pharmacol 143(1):1–12. https://doi.org/10.1006/taap.1996.8074
Cover C, Mansouri A, Knight TR et al (2005) Peroxynitrite-induced mitochondrial and endonuclease-mediated nuclear DNA damage in acetaminophen hepatotoxicity. J Pharmacol Exp Ther 315(2):879–887. https://doi.org/10.1124/jpet.105.088898
Davern TJ 2nd, James LP, Hinson JA et al (2006) Measurement of serum acetaminophen-protein adducts in patients with acute liver failure. Gastroenterology 130(3):687–694. https://doi.org/10.1053/j.gastro.2006.01.033
Davidson DG, Eastham WN (1966) Acute liver necrosis following overdose of paracetamol. Br Med J 2(5512):497–499. https://doi.org/10.1136/bmj.2.5512.497
Ghallab A, Myllys M, Holland CH et al (2019) Influence of liver fibrosis on lobular zonation. Cells 8(12):1556. https://doi.org/10.3390/cells8121556
Ghallab A, Hassan R, Myllys M et al (2021a) Subcellular spatio-temporal intravital kinetics of aflatoxin B(1) and ochratoxin A in liver and kidney. Arch Toxicol 95(6):2163–2177. https://doi.org/10.1007/s00204-021-03073-5
Ghallab A, Myllys M, Friebel A et al (2021b) Spatio-temporal multiscale analysis of western diet-fed mice reveals a translationally relevant sequence of events during NAFLD progression. Cells 10(10):2516. https://doi.org/10.3390/cells10102516
Ghallab A, Hassan R, Hofmann U et al (2022) Interruption of bile acid uptake by hepatocytes after acetaminophen overdose ameliorates hepatotoxicity. J Hepatol 77(1):71–83. https://doi.org/10.1016/j.jhep.2022.01.020
Ghallab A, Gonzalez D, Strangberg E et al (2024) Inhibition of the renal apical sodium dependent bile acid transporter prevents cholemic nephropathy in mice with obstructive cholestasis. J Hepatol 80(2):268–281. https://doi.org/10.1016/j.jhep.2023.10.035
Gow PJ, Jones RM, Dobson JL, Angus PW (2004) Etiology and outcome of fulminant hepatic failure managed at an Australian liver transplant unit. J Gastroenterol Hepatol 19(2):154–159. https://doi.org/10.1111/j.1440-1746.2004.03273.x
Gunawan BK, Liu ZX, Han D, Hanawa N, Gaarde WA, Kaplowitz N (2006) c-Jun N-terminal kinase plays a major role in murine acetaminophen hepatotoxicity. Gastroenterology 131(1):165–178. https://doi.org/10.1053/j.gastro.2006.03.045
Hanawa N, Shinohara M, Saberi B, Gaarde WA, Han D, Kaplowitz N (2008) Role of JNK translocation to mitochondria leading to inhibition of mitochondria bioenergetics in acetaminophen-induced liver injury. J Biol Chem 283(20):13565–13577. https://doi.org/10.1074/jbc.M708916200
Hassan R (2016) Possibilities and limitations of intravital imaging. EXCLI J 15:872–874. https://doi.org/10.17179/excli2016-863
Holland CH, Ramirez Flores RO, Myllys M et al (2022) Transcriptomic cross-species analysis of chronic liver disease reveals consistent regulation between humans and mice. Hepatol Commun 6(1):161–177. https://doi.org/10.1002/hep4.1797
Jaeschke H, McGill MR, Ramachandran A (2012) Oxidant stress, mitochondria, and cell death mechanisms in drug-induced liver injury: lessons learned from acetaminophen hepatotoxicity. Drug Metab Rev 44(1):88–106. https://doi.org/10.3109/03602532.2011.602688
Jaeschke H, Ramachandran A, Chao X, Ding WX (2019) Emerging and established modes of cell death during acetaminophen-induced liver injury. Arch Toxicol 93(12):3491–3502. https://doi.org/10.1007/s00204-019-02597-1
Jaeschke H, Adelusi OB, Akakpo JY et al (2021) Recommendations for the use of the acetaminophen hepatotoxicity model for mechanistic studies and how to avoid common pitfalls. Acta Pharm Sin B 11(12):3740–3755. https://doi.org/10.1016/j.apsb.2021.09.023
Kon K, Kim JS, Jaeschke H, Lemasters JJ (2004) Mitochondrial permeability transition in acetaminophen-induced necrosis and apoptosis of cultured mouse hepatocytes. Hepatology 40(5):1170–1179. https://doi.org/10.1002/hep.20437
Larson AM, Polson J, Fontana RJ et al (2005) Acetaminophen-induced acute liver failure: results of a United States multicenter, prospective study. Hepatology 42(6):1364–1372. https://doi.org/10.1002/hep.20948
McGill MR, Sharpe MR, Williams CD, Taha M, Curry SC, Jaeschke H (2012a) The mechanism underlying acetaminophen-induced hepatotoxicity in humans and mice involves mitochondrial damage and nuclear DNA fragmentation. J Clin Invest 122(4):1574–1583. https://doi.org/10.1172/jci59755
McGill MR, Williams CD, **e Y, Ramachandran A, Jaeschke H (2012b) Acetaminophen-induced liver injury in rats and mice: comparison of protein adducts, mitochondrial dysfunction, and oxidative stress in the mechanism of toxicity. Toxicol Appl Pharmacol 264(3):387–394. https://doi.org/10.1016/j.taap.2012.08.015
Mitchell JR, Jollow DJ, Potter WZ, Davis DC, Gillette JR, Brodie BB (1973) Acetaminophen-induced hepatic necrosis. I. Role of drug metabolism. J Pharmacol Exp Ther 187(1):185–194
Nelson SD (1990) Molecular mechanisms of the hepatotoxicity caused by acetaminophen. Semin Liver Dis 10(4):267–278. https://doi.org/10.1055/s-2008-1040482
Ramachandran A, Lebofsky M, Baines CP, Lemasters JJ, Jaeschke H (2011a) Cyclophilin D deficiency protects against acetaminophen-induced oxidant stress and liver injury. Free Radic Res 45(2):156–164. https://doi.org/10.3109/10715762.2010.520319
Ramachandran A, Lebofsky M, Weinman SA, Jaeschke H (2011b) The impact of partial manganese superoxide dismutase (SOD2)-deficiency on mitochondrial oxidant stress, DNA fragmentation and liver injury during acetaminophen hepatotoxicity. Toxicol Appl Pharmacol 251(3):226–233. https://doi.org/10.1016/j.taap.2011.01.004
Remetic J, Ghallab A, Hobloss Z et al (2022) Loss of bile salt export pump aggravates lipopolysaccharide-induced liver injury in mice due to impaired hepatic endotoxin clearance. Hepatology 75(5):1095–1109. https://doi.org/10.1002/hep.32289
Robichaud G, Garrard KP, Barry JA, Muddiman DC (2013) MSiReader: an open-source interface to view and analyze high resolving power MS imaging files on Matlab platform. J Am Soc Mass Spectrom 24(5):718–721. https://doi.org/10.1007/s13361-013-0607-z
Russomanno G, Sison-Young R, Livoti LA et al (2023) A systems approach reveals species differences in hepatic stress response capacity. Toxicol Sci 196(1):112–125. https://doi.org/10.1093/toxsci/kfad085
Saito C, Lemasters JJ, Jaeschke H (2010) c-Jun N-terminal kinase modulates oxidant stress and peroxynitrite formation independent of inducible nitric oxide synthase in acetaminophen hepatotoxicity. Toxicol Appl Pharmacol 246(1–2):8–17. https://doi.org/10.1016/j.taap.2010.04.015
Schuran FA, Lommetz C, Steudter A et al (2021) Aryl hydrocarbon receptor activity in hepatocytes sensitizes to hyperacute acetaminophen-induced hepatotoxicity in mice. Cell Mol Gastroenterol Hepatol 11(2):371–388. https://doi.org/10.1016/j.jcmgh.2020.09.002
Sezgin S, Hassan R, Zuhlke S et al (2018) Spatio-temporal visualization of the distribution of acetaminophen as well as its metabolites and adducts in mouse livers by MALDI MSI. Arch Toxicol 92(9):2963–2977. https://doi.org/10.1007/s00204-018-2271-3
Vucur M, Ghallab A, Schneider AT et al (2023) Sublethal necroptosis signaling promotes inflammation and liver cancer. Immunity 56(7):1578–1595. https://doi.org/10.1016/j.immuni.2023.05.017
Funding
A.G. was funded by the German Research Foundation (DFG)-Project number 517010379.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
Nothing to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hassan, R., Hobloss, Z., Myllys, M. et al. Acetaminophen overdose causes a breach of the blood–bile barrier in mice but not in rats. Arch Toxicol 98, 1533–1542 (2024). https://doi.org/10.1007/s00204-024-03705-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-024-03705-6