Abstract
The phosphatidylinositol 3-kinase (PI3K) signalling pathway regulates cell survival, proliferation, migration, metabolism and other vital cellular life processes. In addition, activation of the PI3K signalling pathway is important for cancer development. As a result, a variety of PI3K inhibitors have been clinically developed to treat malignancies. Although several PI3K inhibitors have received approval from the Food and Drug Administration (FDA) for significant antitumour activity, frequent and severe adverse effects have greatly limited their clinical application. These toxicities are mostly on-target and immune-mediated; nevertheless, the underlying mechanisms are still unclear. Current management usually involves intervention through symptomatic treatment, with discontinuation if toxicity persists. Therefore, it is necessary to comprehensively understand these adverse events and ensure the clinical safety application of PI3K inhibitors by establishing the most effective management guidelines, appropriate intermittent dosing regimens and new combination administration. Here, the focus is on the development of PI3K inhibitors in cancer therapy, with particular emphasis on isoform-specific PI3K inhibitors. The most common adverse effects of PI3K inhibitors are also covered, as well as potential mechanisms and management approaches.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Excessive activation of the phosphatidylinositol 3-kinase (PI3K) signalling pathway is considered to be one of the hallmarks of human malignancies (Fruman et al. 2017). The pathway is activated through diverse genomic alterations, including the oncogenes phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha (PIK3CA) and phosphoinositide-3-kinase regulatory subunit 1 (PIK3R1), the tumour suppressor gene phosphatase and tensin homolog (PTEN), and other crucial genes, which hold promise as effective therapeutic targets (Janku et al. 2018). In addition, the PI3K axis plays a fundamental role in the survival, cell proliferation, metabolism and inflammation of several cellular processes (De Santis et al. 2019). Hence, it follows that the significance of the PI3K pathway has facilitated pharmacological intervention targeting PI3K.
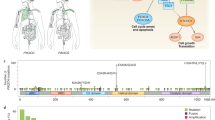
PI3K is a family of lipid kinases that have been divided into three classes (I, II, III) based on sequence homology and substrate specificity (Miller et al. 2019). There are few reports on the specific functions of class II and class III PI3Ks relative to class I PI3Ks. Class II PI3Ks consist of the p110-like catalytic subunit and are capable of regulating the internalization of receptors. Class III PI3Ks are key proteins for vesicle transport from the Golgi to the vacuole in budding yeast (Aksoy et al. 2018). Class I PI3Ks are closely related to cancer and have been studied in-depth. Focus is on the class I PI3Ks, a heterodimer composed of a regulatory subunit and a catalytic subunit, which are divided into class IA and class IB due to different coupled receptors. Class IA is activated by growth factor receptor tyrosine kinase (RTK) and class IB is activated by G-protein-coupled receptor (GPCR) (Fig. 1). Class IA PI3Ks are further subdivided into p110α, p110β, p110δ with regulatory subunit p85, and class IB only includes p110γ with regulatory subunit p87 or p101 (Bilanges et al. 2019). Isoform-specific functions are related to the expression levels in different tissues. p110α and p110β are widely distributed throughout tissues, while p110δ is highly expressed in haematopoietic cells, and p110γ is mainly expressed in leukocytes (Thorpe et al. 2015). Using gene targeting studies, p110α was identified as the crucial isoform involved in vascular remodelling (Vantler et al. 2015), and p110β was considered to play a major role in platelet physiology (Moore et al. 2019); p110δ and p110γ regulate diverse aspects of the function of T and B lymphocytes (De Henau et al. 2016; Horwitz et al. 2018). Therefore, the drug’s effectiveness may be increased by specific PI3K subtype inhibition, but this could also result in more severe adverse events. (Esposito et al. 2019).
Overview of the phosphatidylinositol 3-kinase (PI3K) signalling pathway. The PI3K signalling pathway is activated by G protein-coupled receptor (GPCR) or receptor tyrosine kinase (RTK). Class I PI3Ks activate phosphatidylinositol 4,5‑bisphosphate (PIP2) to generate phosphatidylinositol 3,4,5‑trisphosphate (PIP3), and PIP3 can be dephosphorylated by phosphatase and tensin homolog (PTEN) to form PIP2. PIP3 further induces the activation of the downstream protein kinases phosphoinositide-dependent kinase 1 (PDK1), protein kinase B (Akt), and mammalian target of rapamycin (mTOR) to regulate cell survival and proliferation. Class I PI3Ks are divided into class A and class B. The class A PI3Ks are further subdivided into catalytic subunit p110α, p110β, p110δ with p85 regulatory subunit, and class IB include p110γ with p87 or p101 regulatory subunit
PI3K inhibitors have shown desired therapeutic effects in various cancer treatments. Among these PI3K inhibitors, copanlisib, alpelisib, idelalisib, duvelisib and umbralisib have been approved by the Food and Drug Administration (FDA), although approvals/accelerated application indications of partial inhibitors (idelalisib, duvelisib and umbralisib) have been withdrawn (Meng et al. 2021). In addition, several PI3K inhibitors are undergoing clinical trials. Despite ongoing research on this target, unintended side effects such as hyperglycaemia, rash, diarrhoea/colitis, hepatotoxicity and hypertension continue to be a major barrier to the development of PI3K inhibitors (Hanker et al. 2019).
In this review, we provide a comprehensive summary of the PI3K inhibitors that have been approved or in clinical trials (Table 1). Representative adverse effects associated with PI3K inhibitors, management guidelines and the conceivable mechanisms that have been reported are also discussed.
The typical PI3K inhibitors approved or in clinical trials
Pan-PI3K inhibitors
Due to the lack of a thorough understanding of the structure of the PI3K protein and isoforms in the early stage, PI3K inhibitors targeting the four isoforms of class I PI3K were mainly developed. In addition to playing different roles in tumour proliferation, various isoforms are often associated with multiple physiological functions, such as glucose metabolism, inflammation and immunity. Thus, pan-PI3K inhibitors have inevitably increased safety risks. In particular, the high incidence of metabolic-related adverse events, such as hyperglycaemia, has led to limited clinical doses (De Santis et al. 2019; Janku et al. 2018).
Copanlisib
Copanlisib (Aliqopa™; BAY 80–6946; Bayer AG) is an intravenous pan-class I PI3K inhibitor with potent activity against all four isoforms (Markham 2017) and was approved by the FDA in May 2017 for the treatment of adult patients with relapsed follicular lymphoma (FL) who have received at least two prior systemic therapies. In addition, copanlisib shows greater efficacy and safety both as monotherapy and in combination in various clinical trials. In a phase II study, copanlisib as a single agent also demonstrated significant efficacy with relapsed or refractory indolent lymphoma (NCT01660451) (Dreyling et al. 2017b) and relapsed or refractory diffuse large B cell lymphoma (NCT02391116) (Lenz et al. 2020), and the median progression-free survival (PFS) was 11.2 months and 2.4 months, respectively. Copanlisib plus gemcitabine act synergistically when treating peripheral T cell lymphomas (NCT03052933); PFS was 6.9 months, and median overall survival (OS) was not reached (Yhim et al. 2021). In addition, copanlisib combined with rituximab improved PFS (21.5 months versus 13.8 months in the placebo plus rituximab group) in patients with relapsed indolent non-Hodgkin lymphoma in the CHRONOS-3 study (NCT02367040) (Matasar et al. 2021). Hyperglycaemia, diarrhoea and hypertension are the most frequent adverse events with copanlisib, but all of them are minor (Dreyling et al. 2020). Notably, although other PI3K inhibitors have remarkable safety concerns, copanlisib has a low incidence of severe toxicities may be due to the intravenous route of administration and intermittent dosing schedule (Killock 2021; Munoz et al. 2021).
Buparlisib
Buparlisib (NVP-BKM120; Novartis) is an oral pan-PI3K inhibitor that selectively inhibits all isoforms of class I PI3K (Maira et al. 2012). On one hand, buparlisib has shown improved PFS in the clinical development of combined treatment, but on the other hand, it is limited by extensive toxicities (van Dam 2019). The BELLE-2 (NCT01610284) and BELLE-3 (NCT01633060) trials reported that buparlisib plus fulvestrant was effective in hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative, advanced breast cancer, with a median PFS of 6.9 months and 3.9 months, respectively. However, the toxicities associated with this combination, such as increased alanine aminotransferase (ALT) and aspartate aminotransferase (AST), hyperglycaemia and rash, led to clinical trial discontinuation (Baselga et al. 2017; Di Leo et al. 2018). Other studies have shown that buparlisib failed to have sufficient antitumour activity as a single agent or even plus carboplatin/lomustine in patients with recurrent glioblastoma (Rosenthal et al. 2020; Wen et al. 2019). Although some clinical trials suggest that buparlisib showed modest activity in solid tumours, this may be due to the different tissue distributions and functions of the various subunits. To increase efficacy while reducing the toxicity of PI3K inhibitors, future development of PI3K inhibitors should concentrate on isoform-selective PI3K inhibitors (McPherson et al. 2020; Soulieres et al. 2017).
Isoform-specific PI3K inhibitors
The four isoforms of class I PI3Ks (α, β, δ, γ) have different tissue distributions and physiological functions, which affect the inhibitory effect of isoform-specific inhibitors on various tumours and the incidence of adverse effects (Table 2).
PI3Kα inhibitors
The PIK3CA gene encodes the p110α catalytic subunit of PI3K, and its mutation occurs at a high rate in endometrial, breast, bladder, cervical and colorectal cancers (Arafeh and Samuels 2019). The PIK3CA activation mutation is among the most common oncogenic mutations described in breast cancer to date (Arafeh and Samuels 2019).
p110α is a widely expressed PI3K isoform in vivo and a key intermediate in insulin-like growth factor-1 (IGF-1), insulin and leptin signalling, where it plays a key role in growth factor and metabolic signalling through highly selective recruitment and activation of the insulin receptor substrate (IRS) signalling complex (Hopkins et al. 2018). p110α is significantly expressed in endothelial cells and its activity is necessary for vascular development. Severe defects in angiogenic sprouting and vascular remodelling caused by generalized or endothelial cell-specific inactivation of p110α lead to embryo death in the second trimester (Araiz et al. 2019).
Alpelisib
Alpelisib (Piqray™; BYL719; Novartis) is an oral, highly selective inhibitor of PI3Kα, that received approval on May 2019 by the FDA and is indicated in combination with fulvestrant for the treatment of postmenopausal women, and men, with HR-positive, HER2-negative, PIK3CA-mutated, advanced or metastatic breast cancer (Markham 2019). The SOLAR-1 trial (NCT02437318) showed that the PFS of breast cancer patients treated with alpelisib plus fulvestrant was significantly longer than that of patients treated with placebo-fulvestrant (11 months versus 5.7 months), and the overall response rate was greater (26.6% versus 12.8%) (Andre et al. 2019). Furthermore, in a phase Ib clinical trial (NCT01623349), alpelisib in combination with olaparib was also tolerable and effective in patients with triple-negative breast cancer. The median OS of the enrolled patients was 11.8 months (Batalini et al. 2019). Currently, FDA-approved inhibitors include the pan-PI3K inhibitor copanlisib, PI3Kα inhibitor alpelisib, PI3Kγ/δ inhibitor duvelisib, PI3Kδ inhibitor idelalisib and umbralisib. In addition to alpelisib for the treatment of breast cancer, the approved indications for other inhibitors are haematological malignancies. Meanwhile, exploring the application of PI3K inhibitors in solid tumours has become a new hotspot.
However, with the launch of PI3K inhibitors, serious safety issues related to p110δ are increasingly exposed (Curigliano and Shah 2019). p110δ is preferentially expressed mainly in the haematopoietic system and affects immune cell development and function (Braun et al. 2021). On one hand, PI3Kδ inhibitors, thus, show significant efficacy in the treatment of haematological malignancies due to the regulation of immune cells (Xenou and Papakonstanti 2020). On the other hand, the immune-activating effects of PI3Kδ inhibitors have resulted in severe and lethal immune-related adverse reactions, such as hepatotoxicity, pneumonia, colitis and even intestinal perforation (Roskoski 2021). Furthermore, regulation of insulin signalling by p110α also contributes to PI3Kα inhibitor-related hyperglycaemia (Molinaro et al. 2019).
The PI3K inhibitors approved by the FDA based on single-arm trials have basically failed to show their due advantages. Although single-arm trials allow the evaluation of ORRs, they cannot accurately assess PFS and OS, and it is difficult to accurately characterize the efficacy and toxicity observed in patients. Approval of PI3K inhibitors will be significantly more difficult in the future due to serious safety issues. There are three recommendations made by ODAC: first, advocate careful dose selection through robust dose exploration in early randomized trials; second, avoid single-arm trials as a regulatory strategy in favour of randomized trials; third, comprehensively collect and analyse OS data to assess the effect of the drug on this “ultimate safety endpoint”. In brief, the above three points are achieved to determine a real and credible safety window of PI3K inhibitors, thereby ensuring the efficacy and safety of treatment.
Overall, first, on-target toxicities severely limit the development of PI3K inhibitors, and understanding the physiological functions of different isoforms of PI3K and their distribution in tissues is helpful for the clinical prediction of adverse effects. In addition to the need for corresponding adverse event management guidelines, it is also necessary to further clarify the mechanism of toxicities and find targeted intervention strategies. In addition, the side effects of PI3K inhibitors are closely related to their high-dose clinical application. Therefore, a large number of clinical studies are needed to determine the balance point of immunoregulation with PI3K inhibitors and to reduce side effects by optimizing low-dose and multiple administration. Future development of PI3K inhibitors requires exploring new intermittent delivery modalities or combination regimens to reduce the clinical dose of PI3K inhibitors while ensuring antitumour efficacy. Second, the lack of biomarkers also limits the clinical application of PI3K inhibitors. For example, stratification of breast tumours according to single and multiple copies of PIK3CA mutations resulted in distinct distributions of scores for PI3K signalling and cellular stemness (Madsen et al. 2021). This suggests that more genomic studies are still needed to accurately assess patient stratification for PI3K-targeted therapy and to identify a biomarker that can effectively predict patient susceptibility to PI3K inhibitors.
Data availability
The authors confirm that all the data are available within the text of the review.
Abbreviations
- Akt:
-
Protein kinase B
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- Bcl-2:
-
B cell lymphoma-2
- BTK:
-
Bruton's tyrosine kinase
- CCL:
-
C–C motif chemokine ligand
- CK1ε:
-
Casein kinase 1 epsilon
- CLL:
-
Chronic lymphocytic leukaemia
- FDA:
-
Food and Drug Administration
- FL:
-
Follicular lymphoma
- GPCR:
-
G-protein-coupled receptor
- HR:
-
Hormone receptor
- IGF-1:
-
Insulin-like growth factor-1
- IRS:
-
Insulin receptor substrate
- JNK:
-
C-Jun N-terminal kinase
- KRAS:
-
Kirsten rat sarcoma
- MEK:
-
Mitogen-activated protein kinase kinase
- HER2:
-
Human epidermal growth factor receptor 2
- ODAC:
-
Oncologic drug advisory committee
- PIP2:
-
Phosphatidylinositol 4,5‑bisphosphate
- PIP3:
-
Phosphatidylinositol 3,4,5-trisphosphate
- PTEN :
-
Phosphatase and tensin homolog
- PIK3CA :
-
Phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha
- PIK3R1 :
-
Phosphoinositide-3-kinase regulatory subunit 1
- PIK3CG :
-
Phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit gamma
- IGHV :
-
Immunoglobulin heavy chain variable region
- mTOR:
-
Mammalian target of rapamycin
- MZL:
-
Marginal zone lymphoma
- ORRs:
-
Objective response rates
- OS:
-
Overall survival
- PD-1:
-
Programmed death 1
- PD-L1:
-
Programmed cell death ligand 1
- PDX:
-
Patient-derived xenograft
- PFS:
-
Progression-free survival
- PI3K:
-
Phosphatidylinositol 3-kinase
- PJP:
-
Pneumocystis jirovecii Pneumonia
- RTK:
-
Receptor tyrosine kinase
- SLL:
-
Small lymphocytic lymphoma
- Th1:
-
T-helper type 1
- Tregs:
-
Regulatory T cells
- ULN:
-
Upper limit of normal
References
Aksoy E, Saveanu L, Manoury B (2018) The isoform selective roles of pi3ks in dendritic cell biology and function. Front Immunol 9:2574. https://doi.org/10.3389/fimmu.2018.02574
Albiges L, Chamming’s F, Duclos B et al (2012) Incidence and management of mTOR inhibitor-associated pneumonitis in patients with metastatic renal cell carcinoma. Ann Oncol 23(8):1943–1953. https://doi.org/10.1093/annonc/mds115
Anderson K, Ryan N, Alkhimovitch A, Siddiqui A, Oghumu S (2021) Inhibition of PI3K Isoform p110gamma increases both anti-tumor and immunosuppressive responses to aggressive murine head and neck squamous cell carcinoma with low immunogenicity. Cancers. https://doi.org/10.3390/cancers13050953
Andre F, Ciruelos E, Rubovszky G et al (2019) Alpelisib for PIK3CA-Mutated, Hormone Receptor-Positive Advanced Breast Cancer. N Engl J Med 380(20):1929–1940. https://doi.org/10.1056/NEJMoa1813904
Andre F, Ciruelos EM, Juric D et al (2021) Alpelisib plus fulvestrant for PIK3CA-mutated, hormone receptor-positive, human epidermal growth factor receptor-2-negative advanced breast cancer: final overall survival results from SOLAR-1. Ann Oncol 32(2):208–217. https://doi.org/10.1016/j.annonc.2020.11.011
Arafeh R, Samuels Y (2019) PIK3CA in cancer: The past 30 years. Semin Cancer Biol 59:36–49. https://doi.org/10.1016/j.semcancer.2019.02.002
Araiz C, Yan A, Bettedi L et al (2019) Enhanced beta-adrenergic signalling underlies an age-dependent beneficial metabolic effect of PI3K p110alpha inactivation in adipose tissue. Nat Commun 10(1):1546. https://doi.org/10.1038/s41467-019-09514-1
Barlaam B, Cosulich S, Degorce S et al (2015) Discovery of (R)-8-(1-(3,5-difluorophenylamino)ethyl)-N, N-dimethyl-2-morpholino-4-oxo-4H-chrom ene-6-carboxamide (AZD8186): a potent and selective inhibitor of PI3Kbeta and PI3Kdelta for the treatment of PTEN-deficient cancers. J Med Chem 58(2):943–962. https://doi.org/10.1021/jm501629p
Barr PM, Saylors GB, Spurgeon SE et al (2016) Phase 2 study of idelalisib and entospletinib: pneumonitis limits combination therapy in relapsed refractory CLL and NHL. Blood 127(20):2411–2415. https://doi.org/10.1182/blood-2015-12-683516
Barrientos JC (2016) Idelalisib for the treatment of indolent non-Hodgkin lymphoma: a review of its clinical potential. Onco Targets Ther 9:2945–2953. https://doi.org/10.2147/OTT.S102573
Baselga J, Im SA, Iwata H et al (2017) Buparlisib plus fulvestrant versus placebo plus fulvestrant in postmenopausal, hormone receptor-positive, HER2-negative, advanced breast cancer (BELLE-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol 18(7):904–916. https://doi.org/10.1016/S1470-2045(17)30376-5
Batalini F, **ong N, Tayob N et al (2022) Phase 1b Clinical Trial with Alpelisib plus Olaparib for Patients with Advanced Triple-Negative Breast Cancer. Clin Cancer Res 28(8):1493–1499. https://doi.org/10.1158/1078-0432.CCR-21-3045
Bilanges B, Posor Y, Vanhaesebroeck B (2019) PI3K isoforms in cell signalling and vesicle trafficking. Nat Rev Mol Cell Biol 20(9):515–534. https://doi.org/10.1038/s41580-019-0129-z
Blair HA (2018) Duvelisib: First Global Approval. Drugs 78(17):1847–1853. https://doi.org/10.1007/s40265-018-1013-4
Braun C, Schlaweck S, Daecke SN, Brossart P, Heine A (2021) The PI3Kdelta inhibitor idelalisib impairs the function of human dendritic cells. Cancer Immunol Immunother 70(12):3693–3700. https://doi.org/10.1007/s00262-021-02988-3
Cheson BD, O’Brien S, Ewer MS et al (2019) Optimal Management of Adverse Events From Copanlisib in the Treatment of Patients With Non-Hodgkin Lymphomas. Clin Lymphoma Myeloma Leuk 19(3):135–141. https://doi.org/10.1016/j.clml.2018.11.021
Choudhury AD, Higano CS, de Bono JS et al (2022) A Phase I Study Investigating AZD8186, a Potent and Selective Inhibitor of PI3Kbeta/delta, in Patients with Advanced Solid Tumors. Clin Cancer Res 28(11):2257–2269
Coutre SE, Barrientos JC, Brown JR et al (2015) Management of adverse events associated with idelalisib treatment: expert panel opinion. Leuk Lymphoma 56(10):2779–2786. https://doi.org/10.3109/10428194.2015.1022770
Cuneo A, Barosi G, Danesi R et al (2019) Management of adverse events associated with idelalisib treatment in chronic lymphocytic leukemia and follicular lymphoma: A multidisciplinary position paper. Hematol Oncol 37(1):3–14. https://doi.org/10.1002/hon.2540
Curigliano G, Shah RR (2019) Safety and Tolerability of Phosphatidylinositol-3-Kinase (PI3K) Inhibitors in Oncology. Drug Saf 42(2):247–262. https://doi.org/10.1007/s40264-018-0778-4
Davids MS, Fisher DC, Tyekucheva S et al (2021a) A phase 1b/2 study of duvelisib in combination with FCR (DFCR) for frontline therapy for younger CLL patients. Leukemia 35(4):1064–1072. https://doi.org/10.1038/s41375-020-01010-6
Davids MS, O’Connor OA, Jurczak W et al (2021b) Integrated safety analysis of umbralisib, a dual PI3Kdelta/CK1epsilon inhibitor, in relapsed/refractory lymphoid malignancies. Blood Adv 5(23):5332–5343. https://doi.org/10.1182/bloodadvances.2021005132
De Henau O, Rausch M, Winkler D et al (2016) Overcoming resistance to checkpoint blockade therapy by targeting PI3Kgamma in myeloid cells. Nature 539(7629):443–447. https://doi.org/10.1038/nature20554
De Santis MC, Gulluni F, Campa CC, Martini M, Hirsch E (2019) Targeting PI3K signaling in cancer: Challenges and advances. Biochim Biophys Acta Rev Cancer 1871(2):361–366. https://doi.org/10.1016/j.bbcan.2019.03.003
Dewan AK, Sowerby L, Jadeja S et al (2018) Pityriasis rubra pilaris-like erythroderma secondary to phosphoinositide 3-kinase inhibition. Clin Exp Dermatol 43(8):890–894. https://doi.org/10.1111/ced.13608
Dhillon S, Keam SJ (2021) Umbralisib: First Approval. Drugs 81(7):857–866. https://doi.org/10.1007/s40265-021-01504-2
Di Leo A, Johnston S, Lee KS et al (2018) Buparlisib plus fulvestrant in postmenopausal women with hormone-receptor-positive, HER2-negative, advanced breast cancer progressing on or after mTOR inhibition (BELLE-3): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol 19(1):87–100. https://doi.org/10.1016/S1470-2045(17)30688-5
Dreyling M, Morschhauser F, Bouabdallah K et al (2017a) Phase II study of copanlisib, a PI3K inhibitor, in relapsed or refractory, indolent or aggressive lymphoma. Ann Oncol 28(9):2169–2178. https://doi.org/10.1093/annonc/mdx289
Dreyling M, Santoro A, Mollica L et al (2017b) Phosphatidylinositol 3-Kinase Inhibition by Copanlisib in Relapsed or Refractory Indolent Lymphoma. J Clin Oncol 35(35):3898–3905. https://doi.org/10.1200/JCO.2017.75.4648
Dreyling M, Santoro A, Mollica L et al (2020) Long-term safety and efficacy of the PI3K inhibitor copanlisib in patients with relapsed or refractory indolent lymphoma: 2-year follow-up of the CHRONOS-1 study. Am J Hematol 95(4):362–371. https://doi.org/10.1002/ajh.25711
Eschweiler S, Ramirez-Suastegui C, Li Y et al (2022) Intermittent PI3Kdelta inhibition sustains anti-tumour immunity and curbs irAEs. Nature 605(7911):741–746. https://doi.org/10.1038/s41586-022-04685-2
Esposito A, Viale G, Curigliano G (2019) Safety, Tolerability, and Management of Toxic Effects of Phosphatidylinositol 3-Kinase Inhibitor Treatment in Patients With Cancer: A Review. JAMA Oncol 5(9):1347–1354. https://doi.org/10.1001/jamaoncol.2019.0034
Flinn IW, Hillmen P, Montillo M et al (2018) The phase 3 DUO trial: duvelisib vs ofatumumab in relapsed and refractory CLL/SLL. Blood 132(23):2446–2455. https://doi.org/10.1182/blood-2018-05-850461
Flinn IW, Cherry MA, Maris MB, Matous JV, Berdeja JG, Patel M (2019a) Combination trial of duvelisib (IPI-145) with rituximab or bendamustine/rituximab in patients with non-Hodgkin lymphoma or chronic lymphocytic leukemia. Am J Hematol 94(12):1325–1334. https://doi.org/10.1002/ajh.25634
Flinn IW, Miller CB, Ardeshna KM et al (2019b) DYNAMO: A Phase II Study of Duvelisib (IPI-145) in Patients With Refractory Indolent Non-Hodgkin Lymphoma. J Clin Oncol 37(11):912–922. https://doi.org/10.1200/JCO.18.00915
Fowler NH, Samaniego F, Jurczak W et al (2021) Umbralisib, a Dual PI3Kdelta/CK1epsilon Inhibitor in Patients With Relapsed or Refractory Indolent Lymphoma. J Clin Oncol 39(15):1609–1618. https://doi.org/10.1200/JCO.20.03433
Fruman DA, Chiu H, Hopkins BD, Bagrodia S, Cantley LC, Abraham RT (2017) The PI3K Pathway in Human Disease. Cell 170(4):605–635. https://doi.org/10.1016/j.cell.2017.07.029
Fujikawa T, Choi YH, Yang DJ et al (2019) P110beta in the ventromedial hypothalamus regulates glucose and energy metabolism. Exp Mol Med 51(4):1–9. https://doi.org/10.1038/s12276-019-0249-8
Furman RR, Sharman JP, Coutre SE et al (2014) Idelalisib and rituximab in relapsed chronic lymphocytic leukemia. N Engl J Med 370(11):997–1007. https://doi.org/10.1056/NEJMoa1315226
Goldman JW, Mendenhall MA, Rettinger SR (2016) Hyperglycemia Associated With Targeted Oncologic Treatment: Mechanisms and Management. Oncologist 21(11):1326–1336. https://doi.org/10.1634/theoncologist.2015-0519
Gopal AK, Kahl BS, de Vos S et al (2014) PI3Kdelta inhibition by idelalisib in patients with relapsed indolent lymphoma. N Engl J Med 370(11):1008–1018. https://doi.org/10.1056/NEJMoa1314583
Goulielmaki E, Bermudez-Brito M, Andreou M et al (2018) Pharmacological inactivation of the PI3K p110delta prevents breast tumour progression by targeting cancer cells and macrophages. Cell Death Dis 9(6):678. https://doi.org/10.1038/s41419-018-0717-4
Hancox U, Cosulich S, Hanson L et al (2015) Inhibition of PI3Kbeta signaling with AZD8186 inhibits growth of PTEN-deficient breast and prostate tumors alone and in combination with docetaxel. Mol Cancer Ther 14(1):48–58. https://doi.org/10.1158/1535-7163.MCT-14-0406
Hanker AB, Kaklamani V, Arteaga CL (2019) Challenges for the Clinical Development of PI3K Inhibitors: Strategies to Improve Their Impact in Solid Tumors. Cancer Discov 9(4):482–491. https://doi.org/10.1158/2159-8290.CD-18-1175
Haustraete E, Obert J, Diab S et al (2016) Idelalisib-related pneumonitis. Eur Respir J 47(4):1280–1283. https://doi.org/10.1183/13993003.01670-2015
He Y, Sun MM, Zhang GG et al (2021) Targeting PI3K/Akt signal transduction for cancer therapy. Signal Transduct Target Ther 6(1):425. https://doi.org/10.1038/s41392-021-00828-5
Hopkins BD, Pauli C, Du X et al (2018) Suppression of insulin feedback enhances the efficacy of PI3K inhibitors. Nature 560(7719):499–503. https://doi.org/10.1038/s41586-018-0343-4
Horwitz SM, Koch R, Porcu P et al (2018) Activity of the PI3K-delta, gamma inhibitor duvelisib in a phase 1 trial and preclinical models of T-cell lymphoma. Blood 131(8):888–898. https://doi.org/10.1182/blood-2017-08-802470
Huilaja L, Lindgren O, Soronen M, Siitonen T, Tasanen K (2018) A slowly developed severe cutaneous adverse reaction to idelalisib. J Eur Acad Dermatol Venereol 32(5):e192–e193. https://doi.org/10.1111/jdv.14700
Iannello A, Vitale N, Coma S et al (2021) Synergistic efficacy of the dual PI3K-delta/gamma inhibitor duvelisib with the Bcl-2 inhibitor venetoclax in Richter syndrome PDX models. Blood 137(24):3378–3389. https://doi.org/10.1182/blood.2020010187
Janku F, Yap TA, Meric-Bernstam F (2018) Targeting the PI3K pathway in cancer: are we making headway? Nat Rev Clin Oncol 15(5):273–291. https://doi.org/10.1038/nrclinonc.2018.28
Jiang M, He K, Qiu T et al (2020) Tumor-targeted delivery of silibinin and IPI-549 synergistically inhibit breast cancer by remodeling the microenvironment. Int J Pharm 581:119239
Juric D, Rodon J, Tabernero J et al (2018) Phosphatidylinositol 3-Kinase alpha-Selective Inhibition With Alpelisib (BYL719) in PIK3CA-Altered Solid Tumors: Results From the First-in-Human Study. J Clin Oncol 36(13):1291–1299. https://doi.org/10.1200/JCO.2017.72.7107
Juric D, Janku F, Rodon J et al (2019) Alpelisib plus fulvestrant in PIK3CA-altered and PIK3CA-wild-type estrogen receptor-positive advanced breast cancer: a phase 1b clinical trial. JAMA Oncol 5(2):e184475
Killock D (2021) Copanlisib safe and active in combination. Nat Rev Clin Oncol 18(6):322. https://doi.org/10.1038/s41571-021-00512-1
Lampson BL, Brown JR (2017) PI3Kdelta-selective and PI3Kalpha/delta-combinatorial inhibitors in clinical development for B-cell non-Hodgkin lymphoma. Expert Opin Investig Drugs 26(11):1267–1279. https://doi.org/10.1080/13543784.2017.1384815
Lampson BL, Kasar SN, Matos TR et al (2016) Idelalisib given front-line for treatment of chronic lymphocytic leukemia causes frequent immune-mediated hepatotoxicity. Blood 128(2):195–203. https://doi.org/10.1182/blood-2016-03-707133
Lampson BL, Kim HT, Davids MS et al (2019) Efficacy results of a phase 2 trial of first-line idelalisib plus ofatumumab in chronic lymphocytic leukemia. Blood Adv 3(7):1167–1174. https://doi.org/10.1182/bloodadvances.2018030221
Lenz G, Hawkes E, Verhoef G et al (2020) Single-agent activity of phosphatidylinositol 3-kinase inhibition with copanlisib in patients with molecularly defined relapsed or refractory diffuse large B-cell lymphoma. Leukemia 34(8):2184–2197. https://doi.org/10.1038/s41375-020-0743-y
Li M, Sala V, De Santis MC et al (2018) Phosphoinositide 3-kinase gamma inhibition protects from anthracycline cardiotoxicity and reduces tumor growth. Circulation 138(7):696–711. https://doi.org/10.1161/CIRCULATIONAHA.117.030352
Li JY, Ren KK, Zhang WJ et al (2019) Human amniotic mesenchymal stem cells and their paracrine factors promote wound healing by inhibiting heat stress-induced skin cell apoptosis and enhancing their proliferation through activating PI3K/AKT signaling pathway. Stem Cell Res Ther 10(1):247. https://doi.org/10.1186/s13287-019-1366-y
Liu R, Huang J, Ge Y et al (2020) Inhibition of Phosphatidylinositol 3-Kinase gamma by IPI-549 Attenuates Abdominal Aortic Aneurysm Formation in Mice. Eur J Vasc Endovasc Surg 60(2):254–263. https://doi.org/10.1016/j.ejvs.2020.03.042
Louie CY, DiMaio MA, Matsukuma KE, Coutre SE, Berry GJ, Longacre TA (2015) Idelalisib-associated Enterocolitis: Clinicopathologic Features and Distinction From Other Enterocolitides. Am J Surg Pathol 39(12):1653–1660. https://doi.org/10.1097/PAS.0000000000000525
Lynch JT, Polanska UM, Delpuech O et al (2017) Inhibiting PI3Kbeta with AZD8186 Regulates Key Metabolic Pathways in PTEN-Null Tumors. Clin Cancer Res 23(24):7584–7595. https://doi.org/10.1158/1078-0432.CCR-17-0676
Madsen RR, Erickson EC, Rueda OM et al (2021) Positive correlation between transcriptomic stemness and PI3K/AKT/mTOR signaling scores in breast cancer, and a counterintuitive relationship with PIK3CA genotype. PLoS Genet 17(11):e1009876
Maharaj K, Powers JJ, Achille A et al (2020) The dual PI3Kdelta/CK1epsilon inhibitor umbralisib exhibits unique immunomodulatory effects on CLL T cells. Blood Adv 4(13):3072–3084. https://doi.org/10.1182/bloodadvances.2020001800
Maira SM, Pecchi S, Huang A et al (2012) Identification and characterization of NVP-BKM120, an orally available pan-class I PI3-kinase inhibitor. Mol Cancer Ther 11(2):317–328. https://doi.org/10.1158/1535-7163.MCT-11-0474
Mao N, Zhang Z, Lee YS et al (2021) Defining the therapeutic selective dependencies for distinct subtypes of PI3K pathway-altered prostate cancers. Nat Commun 12(1):5053. https://doi.org/10.1038/s41467-021-25341-9
Markham A (2014) Idelalisib: first global approval. Drugs 74(14):1701–1707. https://doi.org/10.1007/s40265-014-0285-6
Markham A (2017) Copanlisib: First Global Approval. Drugs 77(18):2057–2062. https://doi.org/10.1007/s40265-017-0838-6
Markham A (2019) Alpelisib: First Global Approval. Drugs 79(11):1249–1253. https://doi.org/10.1007/s40265-019-01161-6
Marques RB, Aghai A, de Ridder CMA et al (2015) High Efficacy of Combination Therapy Using PI3K/AKT Inhibitors with Androgen Deprivation in Prostate Cancer Preclinical Models. Eur Urol 67(6):1177–1185. https://doi.org/10.1016/j.eururo.2014.08.053
Marques M, Tranchant R, Risa-Ebri B et al (2020) Combined MEK and PI3K/p110beta Inhibition as a Novel Targeted Therapy for Malignant Mesothelioma Displaying Sarcomatoid Features. Cancer Res 80(4):843–856. https://doi.org/10.1158/0008-5472.CAN-19-1633
Matasar MJ, Capra M, Ozcan M et al (2021) Copanlisib plus rituximab versus placebo plus rituximab in patients with relapsed indolent non-Hodgkin lymphoma (CHRONOS-3): a double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Oncol 22(5):678–689. https://doi.org/10.1016/S1470-2045(21)00145-5
Mateo J, Ganji G, Lemech C et al (2017) A First-Time-in-Human Study of GSK2636771, a Phosphoinositide 3 Kinase Beta-Selective Inhibitor, in Patients with Advanced Solid Tumors. Clin Cancer Res 23(19):5981–5992. https://doi.org/10.1158/1078-0432.CCR-17-0725
McPherson V, Reardon B, Bhayankara A et al (2020) A phase 2 trial of buparlisib in patients with platinum-resistant metastatic urothelial carcinoma. Cancer 126(20):4532–4544. https://doi.org/10.1002/cncr.33071
Meeting of the oncologic drugs advisory committee (2022) U.S. Food and Drug Administration. https://www.fda.gov/advisory-committees/advisory-committee-calendar/updated-information-april-21-22-2022-meeting-oncologic-drugs-advisory-committee-meeting-announcement. Accessed 21 April 2022.
Meng D, He W, Zhang Y et al (2021) Development of PI3K inhibitors: Advances in clinical trials and new strategies (Review). Pharmacol Res 173:105900
Miller MS, Thompson PE, Gabelli SB (2019) Structural Determinants of Isoform Selectivity in PI3K Inhibitors. Biomolecules. https://doi.org/10.3390/biom9030082
Mishra R, Patel H, Alanazi S, Kilroy MK, Garrett JT (2021) PI3K inhibitors in cancer: clinical implications and adverse effects. Int J Mol Sci. https://doi.org/10.3390/ijms22073464
Molinaro A, Becattini B, Mazzoli A et al (2019) Insulin-Driven PI3K-AKT Signaling in the Hepatocyte Is Mediated by Redundant PI3Kalpha and PI3Kbeta Activities and Is Promoted by RAS. Cell Metab 29(6):1400-1409.e5. https://doi.org/10.1016/j.cmet.2019.03.010
Moore SF, Smith NR, Blair TA, Durrant TN, Hers I (2019) Critical roles for the phosphatidylinositide 3-kinase isoforms p110beta and p110gamma in thrombopoietin-mediated priming of platelet function. Sci Rep 9(1):1468. https://doi.org/10.1038/s41598-018-37012-9
Morschhauser F, Dyer MJS, Walter HS et al (2021) Phase 1b study of tirabrutinib in combination with idelalisib or entospletinib in previously treated B-cell lymphoma. Leukemia 35(7):2108–2113. https://doi.org/10.1038/s41375-020-01108-x
Munoz J, Follows GA, Nastoupil LJ (2021) Copanlisib for the Treatment of Malignant Lymphoma: Clinical Experience and Future Perspectives. Target Oncol 16(3):295–308. https://doi.org/10.1007/s11523-021-00802-9
Nair KS, Cheson B (2016) The role of idelalisib in the treatment of relapsed and refractory chronic lymphocytic leukemia. Ther Adv Hematol 7(2):69–84. https://doi.org/10.1177/2040620715625966
Nakanishi Y, Walter K, Spoerke JM et al (2016) Activating Mutations in PIK3CB Confer Resistance to PI3K Inhibition and Define a Novel Oncogenic Role for p110beta. Cancer Res 76(5):1193–1203. https://doi.org/10.1158/0008-5472.CAN-15-2201
Nastoupil LJ, Lunning MA, Vose JM et al (2019) Tolerability and activity of ublituximab, umbralisib, and ibrutinib in patients with chronic lymphocytic leukaemia and non-Hodgkin lymphoma: a phase 1 dose escalation and expansion trial. Lancet Haematol 6(2):e100–e109. https://doi.org/10.1016/S2352-3026(18)30216-3
Nunnery SE, Mayer IA (2019) Management of toxicity to isoform alpha-specific PI3K inhibitors. Ann Oncol 30(Suppl 10):x21–x26. https://doi.org/10.1093/annonc/mdz440
Nurnberg B, Beer-Hammer S (2019) Function, regulation and biological roles of PI3Kgamma variants. Biomolecules. https://doi.org/10.3390/biom9090427
Perrotta M, Lembo G, Carnevale D (2016) The multifaceted roles of pi3kgamma in hypertension, vascular biology, and inflammation. Int J Mol Sci. https://doi.org/10.3390/ijms17111858
Pridham KJ, Le L, Guo S et al (2018) PIK3CB/p110beta is a selective survival factor for glioblastoma. Neuro Oncol 20(4):494–505. https://doi.org/10.1093/neuonc/nox181
Roeker LE, Feldman TA, Soumerai JD et al (2022) Adding Umbralisib and Ublituximab (U2) to Ibrutinib in Patients with CLL: A Phase 2 Study of an MRD-driven Approach. Clin Cancer Res. https://doi.org/10.1158/1078-0432.CCR-22-0964
Rohrbacher L, Brauchle B, Ogrinc Wagner A, von Bergwelt-Baildon M, Bucklein VL, Subklewe M (2021) The PI3K partial differential-Selective Inhibitor Idelalisib Induces T- and NK-Cell Dysfunction Independently of B-Cell Malignancy-Associated Immunosuppression. Front Immunol 12:608625
Rosenthal M, Clement PM, Campone M et al (2020) Buparlisib plus carboplatin or lomustine in patients with recurrent glioblastoma: a phase Ib/II, open-label, multicentre, randomised study. ESMO Open. https://doi.org/10.1136/esmoopen-2020-000672
Roskoski R Jr (2021) Properties of FDA-approved small molecule phosphatidylinositol 3-kinase inhibitors prescribed for the treatment of malignancies. Pharmacol Res 168:105579
Rugo HS, Andre F, Yamashita T et al (2020) Time course and management of key adverse events during the randomized phase III SOLAR-1 study of PI3K inhibitor alpelisib plus fulvestrant in patients with HR-positive advanced breast cancer. Ann Oncol 31(8):1001–1010. https://doi.org/10.1016/j.annonc.2020.05.001
Rugo HS, Lerebours F, Ciruelos E et al (2021) Alpelisib plus fulvestrant in PIK3CA-mutated, hormone receptor-positive advanced breast cancer after a CDK4/6 inhibitor (BYLieve): one cohort of a phase 2, multicentre, open-label, non-comparative study. Lancet Oncol 22(4):489–498. https://doi.org/10.1016/S1470-2045(21)00034-6
Sarker D, Dawson NA, Aparicio AM et al (2021) A Phase I, Open-Label, Dose-Finding Study of GSK2636771, a PI3Kbeta Inhibitor, Administered with Enzalutamide in Patients with Metastatic Castration-Resistant Prostate Cancer. Clin Cancer Res 27(19):5248–5257. https://doi.org/10.1158/1078-0432.CCR-21-1115
Schwartz S, Wongvipat J, Trigwell CB et al (2015) Feedback suppression of PI3Kalpha signaling in PTEN-mutated tumors is relieved by selective inhibition of PI3Kbeta. Cancer Cell 27(1):109–122. https://doi.org/10.1016/j.ccell.2014.11.008
Sharma P, Abramson VG, O’Dea A et al (2021) Clinical and Biomarker Results from Phase I/II Study of PI3K Inhibitor Alpelisib plus Nab-paclitaxel in HER2-Negative Metastatic Breast Cancer. Clin Cancer Res 27(14):3896–3904. https://doi.org/10.1158/1078-0432.CCR-20-4879
Sharman JP, Coutre SE, Furman RR et al (2019) Final Results of a Randomized, Phase III Study of Rituximab With or Without Idelalisib Followed by Open-Label Idelalisib in Patients With Relapsed Chronic Lymphocytic Leukemia. J Clin Oncol 37(16):1391–1402. https://doi.org/10.1200/JCO.18.01460
Soulieres D, Faivre S, Mesia R et al (2017) Buparlisib and paclitaxel in patients with platinum-pretreated recurrent or metastatic squamous cell carcinoma of the head and neck (BERIL-1): a randomised, double-blind, placebo-controlled phase 2 trial. Lancet Oncol 18(3):323–335. https://doi.org/10.1016/S1470-2045(17)30064-5
Thorpe LM, Yuzugullu H, Zhao JJ (2015) PI3K in cancer: divergent roles of isoforms, modes of activation and therapeutic targeting. Nat Rev Cancer 15(1):7–24. https://doi.org/10.1038/nrc3860
Tomowiak C, Poulain S, Herbaux C et al (2021) Obinutuzumab and idelalisib in symptomatic patients with relapsed/refractory Waldenstrom macroglobulinemia. Blood Adv 5(9):2438–2446. https://doi.org/10.1182/bloodadvances.2020003895
Torres C, Mancinelli G, Cordoba-Chacon J et al (2019) p110gamma deficiency protects against pancreatic carcinogenesis yet predisposes to diet-induced hepatotoxicity. Proc Natl Acad Sci U S A 116(29):14724–14733. https://doi.org/10.1073/pnas.1813012116
Uno JK, Rao KN, Matsuoka K et al (2010) Altered macrophage function contributes to colitis in mice defective in the phosphoinositide-3 kinase subunit p110delta. Gastroenterology 139(5):1642–1653
van Dam PA (2019) The use of buparlisib as a radiosentisiser: What about toxicity? Eur J Cancer 119:194–195. https://doi.org/10.1016/j.ejca.2019.06.026
Vantler M, Jesus J, Leppanen O et al (2015) Class IA Phosphatidylinositol 3-Kinase Isoform p110alpha Mediates Vascular Remodeling. Arterioscler Thromb Vasc Biol 35(6):1434–1444. https://doi.org/10.1161/ATVBAHA.114.304887
Varshney P, Saini N (2018) PI3K/AKT/mTOR activation and autophagy inhibition plays a key role in increased cholesterol during IL-17A mediated inflammatory response in psoriasis. Biochim Biophys Acta Mol Basis Dis 1864:1795–1803
Weidner AS, Panarelli NC, Geyer JT et al (2015) Idelalisib-associated Colitis: Histologic Findings in 14 Patients. Am J Surg Pathol 39(12):1661–1667. https://doi.org/10.1097/PAS.0000000000000522
Wen PY, Touat M, Alexander BM et al (2019) Buparlisib in Patients With Recurrent Glioblastoma Harboring Phosphatidylinositol 3-Kinase Pathway Activation: An Open-Label, Multicenter, Multi-Arm. Phase II Trial J Clin Oncol 37(9):741–750. https://doi.org/10.1200/JCO.18.01207
Wu X, Sun Q, He S et al (2022) Ropivacaine inhibits wound healing by suppressing the proliferation and migration of keratinocytes via the PI3K/AKT/mTOR Pathway. BMC Anesthesiol 22(1):106. https://doi.org/10.1186/s12871-022-01646-0
Xenou L, Papakonstanti EA (2020) p110delta PI3K as a therapeutic target of solid tumours. Clin Sci (lond) 134(12):1377–1397. https://doi.org/10.1042/CS20190772
Yang Q, Modi P, Newcomb T, Queva C, Gandhi V (2015) Idelalisib: First-in-Class PI3K Delta Inhibitor for the Treatment of Chronic Lymphocytic Leukemia, Small Lymphocytic Leukemia, and Follicular Lymphoma. Clin Cancer Res 21(7):1537–1542. https://doi.org/10.1158/1078-0432.CCR-14-2034
Yhim HY, Kim T, Kim SJ et al (2021) Combination treatment of copanlisib and gemcitabine in relapsed/refractory PTCL (COSMOS): an open-label phase I/II trial. Ann Oncol 32(4):552–559. https://doi.org/10.1016/j.annonc.2020.12.009
Zhang J, Gao X, Schmit F et al (2017) CRKL Mediates p110beta-Dependent PI3K Signaling in PTEN-Deficient Cancer Cells. Cell Rep 20(3):549–557. https://doi.org/10.1016/j.celrep.2017.06.054
Zhang X, Shen L, Liu Q, Hou L, Huang L (2019) Inhibiting PI3 kinase-gamma in both myeloid and plasma cells remodels the suppressive tumor microenvironment in desmoplastic tumors. J Control Release 309:173–180. https://doi.org/10.1016/j.jconrel.2019.07.039
Zhang B, Luk C, Valadares J, Aronis C, Foukas LC (2021) Dominant Role of PI3K p110alpha over p110beta in Insulin and beta-Adrenergic Receptor Signalling. Int J Mol Sci. https://doi.org/10.3390/ijms222312813
Zhao HF, Wang J, Jiang HR, Chen ZP, To SS (2016) PI3K p110beta isoform synergizes with JNK in the regulation of glioblastoma cell proliferation and migration through Akt and FAK inhibition. J Exp Clin Cancer Res 35:78. https://doi.org/10.1186/s13046-016-0356-5
Zhao X, Lwin T, Silva A et al (2017) Unification of de novo and acquired ibrutinib resistance in mantle cell lymphoma. Nat Commun 8:14920. https://doi.org/10.1038/ncomms14920
Zhou L, Chu C, Teng F et al (2019) Innate lymphoid cells support regulatory T cells in the intestine through interleukin-2. Nature 568(7752):405–409. https://doi.org/10.1038/s41586-019-1082-x
Acknowledgements
This work was supported by the Fund of the National Natural Science Foundation of China (82274018 to **aochun Yang).
Author information
Authors and Affiliations
Contributions
The idea for the article was proposed by **aochun Yang and Hao Yan; the literature search was done by Miaomiao Yu, Jiajia Chen and Zhifei Xu; the draft of the manuscript was written by Miaomiao Yu; all authors participated in the revision work. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yu, M., Chen, J., Xu, Z. et al. Development and safety of PI3K inhibitors in cancer. Arch Toxicol 97, 635–650 (2023). https://doi.org/10.1007/s00204-023-03440-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-023-03440-4