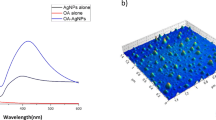
Abstract
Acanthamoeba castellanii is the causative agent of fatal encephalitis and blinding keratitis. Current therapies remain a challenge, hence there is a need to search for new therapeutics. Here, we tested embelin (EMB) and silver nanoparticles doped with embelin (EMB–AgNPs) against A. castellanii. Using amoebicidal assays, the results revealed that both compounds inhibited the viability of Acanthamoeba, having an IC50 of 27.16 ± 0.63 and 13.63 ± 1.08 μM, respectively, while causing minimal cytotoxicity against HaCaT cells in vitro. The findings suggest that both samples induced apoptosis through the mitochondria-mediated pathway. Differentially expressed genes analysis showed that 652 genes were uniquely expressed in treated versus untreated cells, out of which 191 were significantly regulated in the negative control vs. conjugate. Combining the analysis, seven genes (ARIH1, RAP1, H3, SDR16C5, GST, SRX1, and PFN) were highlighted as the most significant (Log2 (FC) value ± 4) for the molecular mode of action in vitro. The KEGG analysis linked most of the genes to apoptosis, the oxidative stress signaling pathway, cytochrome P450, Rap1, and the oxytocin signaling pathways. In summary, this study provides a thorough framework for develo** therapeutic agents against microbial infections using EMB and EMB–AgNPs.
Graphical abstract














Similar content being viewed by others
Availability of data and materials
Data will be provided upon request on case-to-case basis.
Abbreviations
- A. castellanii :
-
Acanthamoeba castellanii
- AgNO3 :
-
Silver nitrate
- AK:
-
Acanthamoeba Keratitis
- ATCC:
-
American Type Culture Collection
- BBB:
-
Blood–brain barrier
- BF:
-
Biological process
- CC:
-
Cellular component
- CNS:
-
Central nervous system
- CPR:
-
NADPH-cytochrome c reductase
- DCF:
-
Dichlorofluorescein
- DCFDA:
-
2′,7′-Dichlorodihydro-fluorescein diacetate
- DEGs:
-
Differentially expressed genes
- DEU:
-
Differential exon usage
- DMSO:
-
Dimethyl sulfoxide
- EMB:
-
Embelin
- EMB–AgNPs:
-
Silver nanoparticles coated with embelin
- FBS:
-
Fetal bovine serum
- FE-SEM:
-
Field emission-scanning electron microscopy
- FPKM:
-
Fragments per kilobases per million
- GAE:
-
Granulomatous amoebic encephalitis
- GO:
-
Gene ontology
- H2O2 :
-
Hydrogen peroxide
- HaCaT:
-
Human keratinocyte cell
- HCS:
-
HiSeq Control Software
- KEGG:
-
Kyoto Encyclopedia of Genes and Genomes
- LDH:
-
Lactate dehydrogenase
- MF:
-
Molecular function
- NGS:
-
Next-generation sequencing
- NPs:
-
Nanoparticles
- PBS:
-
Phosphate-buffered saline
- PCD:
-
Programmed cell death
- PCR:
-
Polymerase chain reaction
- Pen-strep:
-
Penicillin Streptomycin
- PYG:
-
Proteose peptone, yeast extract and glucose
- RCC1:
-
Regulator of chromosome condensation
- ROS:
-
Reactive oxygen species
- RPMI-1640:
-
Roswell Park Memorial Institute 1640 medium
- SDS:
-
Sodium dodecyl sulfate
- TEM:
-
Transmission electron microscope
- UV–Vis:
-
Ultraviolet–visible
- ΔΨm:
-
Mitochondrial membrane potential
References
Ahmed U, Anwar A, Ong SK, Anwar A, Khan NA (2022a) Applications of medicinal chemistry for drug discovery against Acanthamoeba infections. Med Res Rev 42(1):462–512
Ahmed U, Ho KY, Simon SE, Saad SM, Ong SK, Anwar A, Tan KO, Sridewi N, Khan KM, Khan NA, Anwar A (2022b) Potential anti-acanthamoebic effects through inhibition of CYP51 by novel quinazolinones. Acta Trop 231:106440
Alsam S, Sissons J, Jayasekera S, Khan NA (2005) Extracellular proteases of Acanthamoeba castellanii (encephalitis isolate belonging to T1 genotype) contribute to increased permeability in an in-vitro model of the human blood–brain barrier. J Infect 51(2):150–156
Anwar A, Siddiqui R, Hussain MA, Ahmed D, Shah MR, Khan NA (2018) Silver nanoparticle conjugation affects antiacanthamoebic activities of amphotericin B, nystatin, and fluconazole. Parasitol Res 117:265–271
Anwar A, Abdalla SA, Aslam Z, Shah MR, Siddiqui R, Khan NA (2019a) Oleic acid–conjugated silver nanoparticles as efficient antiamoebic agent against Acanthamoeba castellanii. Parasitol Res 118(7):2295–2304
Anwar A, Masri A, Rao K, Rajendran K, Khan NA, Shah MR, Siddiqui R (2019b) Antimicrobial activities of green synthesized gums-stabilized nanoparticles loaded with flavonoids. Sci Rep 9:1–12
Anwar A, Numan A, Siddiqui R, Khalid M, Khan NA (2019c) Cobalt nanoparticles as novel nanotherapeutics against Acanthamoeba castellanii. Parasites Vectors 12(1):1–10
Anwar A, Ting EL, Anwar A, Ul Ain N, Faizi S, Shah MR, Khan NA, Siddiqui R (2020) Antiamoebic activity of plant-based natural products and their conjugated silver nanoparticles against Acanthamoeba castellanii (ATCC 50492). AMB Express 10(1):1–10
Austin A, Lietman T, Rose-Nussbaumer J (2017) Update on the management of infectious keratitis. Ophthalmology 124(11):1678–1689
Baig AM (2015) Pathogenesis of amoebic encephalitis: are the amoebae being credited to an ‘inside job’done by the host immune response? Acta Trop 148:72–76
Baig AM, Lalani S, Khan NA (2017) Apoptosis in Acanthamoeba castellanii belonging to the T4 genotype. J Basic Microbiol 9999:1–6.
Davey RJ, Moens PD (2020) Profilin: many facets of a small protein. Biophys Rev 12:827–849
Dhadde SB, Nagakannan P, Roopesh M, Kumar SA, Thippeswamy BS, Veerapur VP, Badami S (2016) Effect of embelin against 3-nitropropionic acid-induced Huntington’s disease in rats. Biomed Pharmacother 77:52–58
Ding Z, Bae YH, Roy P (2012) Molecular insights on context-specific role of profilin-1 in cell migration. Cell Adh Migr 6(5):442–534
Elmore S (2007) Apoptosis: a review of programmed cell death. Toxicol Pathol 35(4):495–516
Elsheikha HM, Siddiqui R, Khan NA (2020) Drug discovery against Acanthamoeba infections: present knowledge and unmet needs. Pathogens 9(5):405
Fleury C, Mignotte B, Vayssiere JL (2002) Mitochondrial reactive oxygen species in cell death signalling. Biochimie 84:131–141
Goldstein G, Scheid M, Hammerling U, Schlesinger DH, Niall HD, Boyse EA (1975) Isolation of a polypeptide that has lymphocyte-differentiating properties and is probably represented universally in living cells. Proc Natl Acad Sci 72(1):11–15
Hayes JD, Strange RC (2000) Glutathione S-transferase polymorphisms and their biological consequences. Pharmacology 61:154–166
Heredero-Bermejo I, Martin CS, de Carranza JS, Copa-Patiño JL, Pérez-Serrano J (2012) Acanthamoeba castellanii: in-vitro UAH-T17c3 trophozoite growth study in different culture media. Parasitol Res 110(6):2563–2567
Heredero-Bermejo I, Copa-Patiño JL, Soliveri J, Fuentes-Paniagua E, de la Mata FJ, Gomez R, Perez-Serrano J (2015) Evaluation of the activity of new cationic carbosilane dendrimers on trophozoites and cysts of Acanthamoeba polyphaga. Parasitol Res 114(2):473–486
Hu R, Zhu K, Li Y, Yao K, Zhang R, Wang H, Yang W, Liu Z (2011) Embelin induces apoptosis through down-regulation of XIAP in human leukemia cells. Med Oncol 28(4):1584–1588
Jeyamogan S, Khan NA, Anwar A, Shah MR, Siddiqui R (2018) Cytotoxic effects of benzodioxane, naphthalene diimide, porphyrin and acetamol derivatives on HeLa cells. SAGE Open Med 6:1–11
Jha BK, Jung HJ, Seo I, Suh SI, Suh MH, Baek WK (2015) Juglone induces cell death of Acanthamoeba through increased production of reactive oxygen species. Exp Parasitol 159:100–106
Joshi DC, Bakowska JC (2011) Determination of mitochondrial membrane potential and reactive oxygen species in live rat cortical neurons. J vis Exp 51:2704
Kim MG, Su’udi M, Park SR, Hwang DJ, Bae SC (2010) Working mechanism of peroxiredoxins (Prxs) and sulphiredoxin1 (Srx1) in Arabidopsis thaliana. J Life Sci 20(12):1777–1783
Kim D, Langmead B, Salzberg SL (2015) HISAT: a fast spliced aligner with low memory requirements. Nat Methods 12(4):357–360
Ko JH, Lee SG, Yang WM, Um JY, Sethi G, Mishra S, Shanmugam MK, Ahn KS (2018) The application of embelin for cancer prevention and therapy. Molecules 23(3):621
Koutsogiannis Z, MacLeod ET, Maciver SK (2019) G418 induces programmed cell death in Acanthamoeba through the elevation of intracellular calcium and cytochrome c translocation. Parasitol Res 118(2):641–651
Krishnan K, Moens PD (2009) Structure and functions of profilins. Biophys Rev 1(2):71–81
Kundap UP, Bhuvanendran S, Kumari Y, Othman I, Shaikh M (2017) Plant derived phytocompound, embelin in CNS disorders: a systematic review. Front Pharmacol 8:76
Levine B, Kroemer G (2008) Autophagy in the pathogenesis of disease. Cell 132(1):27–42
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15(12):1–21
Loyola A, Almouzni G (2007) Marking histone H3 variants: how, when and why? Trends Biochem Sci 32(9):425–433
Mani A, Gelmann EP (2005) The ubiquitin-proteasome pathway and its role in cancer. J Clin Oncol 23(21):4776–4789
Marciano-Cabral F, Cabral G (2003) Acanthamoeba spp. as agents of disease in humans. Clin Microbiol Rev 16(2):273–307
Masri A, Khan NA, Zoqratt MZ, Ayub Q, Anwar A, Rao K, Shah MR, Siddiqui R (2021) Transcriptome analysis of Escherichia coli K1 after therapy with hesperidin conjugated with silver nanoparticles. BMC Microbiol 21(1):1–1
Misra BB (2014) An updated snapshot of recent advances in transcriptomics and genomics of phytomedicinals. J Postdoc Res 2:1–5
Moon EK, Choi H-S, Kong H-H, Quan F-S (2018) Polyhexamethylene biguanide and chloroquine induce programmed cell death in Acanthamoeba castellanii. Exp Parasitol 191:31–35
Mortazavi A, Williams BA, McCue K, Schaeffer L, Wold B (2008) Map** and quantifying mammalian transcriptomes by RNA-Seq. Nat Methods 5(7):621–628
Moyer BD, Allan BB, Balch WE (2001) Rab1 interaction with a GM130 effector complex regulates COPII vesicle cis-Golgi tethering. Traffic 2(4):268–276
Myung J, Kim KB, Crews CM (2001) The ubiquitin-proteasome pathway and proteasome inhibitors. Med Res Rev 21(4):245–273
Nebert DW, Vasiliou V (2004) Analysis of the glutathione S-transferase (GST) gene family. Hum Genom 1(6):1–5
Nur-E-Kamal A, Gross SR, Pan Z, Balklava Z, Ma J, Liu L (2004) Nuclear translocation of cytochrome c during apoptosis. J Biol Chem 279(24):24911–24914
Orrenius S, Gogvadze V, Zhivotovsky B (2007) Mitochondrial oxidative stress: implications for cell death. Ann Rev Pharmacol Toxicol 47:143–183
Sebastián-Pérez V, Sifaoui I, Reyes-Batlle M, Domínguez-De Barros A, López-Arencibia A, Campillo NE, Piñero JE, Lorenzo-Morales J, Gil C (2021) Discovery of amoebicidal compounds by combining computational and experimental approaches. Antimicrob Agents Chemother 65(2):e01749-e1820
Shirole RL, Shirole NL, Saraf MN (2015) Embelia ribes ameliorates lipopolysaccharide-induced acute respiratory distress syndrome. J Ethnopharmacol 168:356–363
Siddiqui R, Khan NA (2012) Biology and pathogenesis of Acanthamoeba. Parasit Vectors 5:1–3
Siddiqui R, Rajendran K, Abdella B, Ayub Q, Lim SY, Khan NA (2020) Naegleria fowleri: differential genetic expression following treatment with hesperidin conjugated with silver nanoparticles using RNA-Seq. Parasitol Res 119:2351–2358
Sifaoui I, Rodríguez-Expósito RL, Reyes-Batlle M, Rizo-Liendo A, Piñero JE, Bazzocchi IL, Lorenzo-Morales J, Jiménez IA (2019) Ursolic acid derivatives as potential agents against Acanthamoeba Spp. Pathogens 8(3):130
Szatmári Z, Sass M (2014) The autophagic roles of Rab small GTPases and their upstream regulators: a review. Autophagy 10(7):1154–1166
Trapnell C, Williams BA, Pertea G, Mortazavi A, Kwan G, Van Baren MJ, Salzberg SL, Wold BJ, Pachter L (2010) Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol 28(5):511–515
Trocha LK, Stobienia O (2007) Response of Acanthamoeba castellanii mitochondria to oxidative stress. Acta Biochim Pol 54(4):797–803
Turner NA, Russell AD, Furr JR, Lloyd D (2000) Emergence of resistance to biocides during differentiation of Acanthamoeba castellanii. J Antimicrob Chemother 46(1):27–34
Turner NA, Russell AD, Furr JR, Lloyd D (2004) Resistance, biguanide sorption and biguanide-induced pentose leakage during encystment of Acanthamoeba castellanii. J Appl Microbiol 96(6):1287–1295
Visentin C, Musso L, Broggini L, Bonato F, Russo R, Moriconi C, Bolognesi M, Miranda E, Dallavalle S, Passarella D, Ricagno S (2020) Embelin as lead compound for new neuroserpin polymerization inhibitors. Life 10(7):111
Visvesvara GS, Moura H, Schuster FL (2007) Pathogenic and opportunistic free-living amoebae: Acanthamoeba spp., Balamuthia mandrillaris, Naegleria fowleri, and Sappinia diploidea. FEMS Immunol Med Microbiol 50(1):1–26
Wang C, Zhu J, Liu M, Yang Q, Wu J, Li Z (2018) De novo sequencing and transcriptome assembly of Arisaema heterophyllum Blume and identification of genes involved in isoflavonoid biosynthesis. Sci Rep 8(1):1–2
Wang YJ, Chen CH, Chen JW, Lin WC (2021) Commensals serve as natural barriers to mammalian cells during Acanthamoeba castellanii Invasion. Microbiol Spectr 9(3):e0051221
Webster D, Umar I, Kolyvas G, Bilbao J, Guiot MC, Duplisea K, Qvarnstrom Y, Visvesvara GS (2012) Treatment of granulomatous amoebic encephalitis with voriconazole and miltefosine in an immunocompetent soldier. Am J Trop Med Hyg 87(4):715–718
Weng L, Shen S, Wu S, Yin X, Liu B, Shang M, Zou X, Mao A (2020) Identification of critical genes and proteins for stent restenosis induced by esophageal benign hyperplasia in esophageal cancer. Front Genet 11:563954
Wingett SW, Andrews S (2018) FastQ screen: a tool for multi-genome map** and quality control. F1000Res 7:1338
Wu D, Qiao K, Feng M, Fu Y, Cai J, Deng Y, Tachibana H, Cheng X (2018) Apoptosis of Acanthamoeba castellanii trophozoites induced by oleic acid. J Eukaryot Microbiol 65(2):191–199
Xue Z, Ge Z, Zhang K, Sun R, Yang J, Han R, Peng M, Li Y, Li W, Zhang D, Hao J (2014) Embelin suppresses dendritic cell functions and limits autoimmune encephalomyelitis through the TGF-β/β-catenin and STAT3 signalling pathways. Mol Neurobiol 49(2):1087–1101
Young MD, Wakefield MJ, Smyth GK, Oshlack A (2010) Gene ontology analysis for RNA-seq: accounting for selection bias. Genome Biol 11(2):1–2
Acknowledgements
This research work was supported by Sunway University, Malaysia, Monash University, Malaysia, and the H. E. J. Research Institute of Chemistry, International Center for Chemical and Biological Sciences, University of Karachi.
Funding
This research work was supported by Sunway University, Malaysia (Individual Research Grant, GRTIN-IRG-10-2022) and Sindh Higher Education Commission (SHEC), Pakistan vide letter No. NO.DD/SHEC/1-14/2014, Project code SHEC/SRSP/Med-3/15/2021-21. Ruqaiyyah Siddiqui and Naveed Ahmed Khan were supported by the Air Force Office of Scientific Research (AFOSR), USA.
Author information
Authors and Affiliations
Contributions
AA and NAK conceptualized the study. AA, RS, BSA, and NAK planned the study. UA and AA conducted the experiments and wrote the first draft of the manuscript. S-KO wrote the parts of NGS, KMK, RS, BSA, and MFS corrected the manuscript. NAK and AA supervised the entire study. All authors approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
No studies were conducted on animals and/or humans.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ahmed, U., Ong, SK., Khan, K.M. et al. Effect of embelin on inhibition of cell growth and induction of apoptosis in Acanthamoeba castellanii. Arch Microbiol 205, 360 (2023). https://doi.org/10.1007/s00203-023-03698-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-023-03698-3