Abstract
Introduction and Hypothesis
The aim of this article is to present a systematic literature review focused on microbiome diversity in women experiencing pelvic floor dysfunction.
Methods
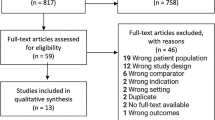
Utilizing PubMed/MedLine and Scopus, 25 pertinent studies were meticulously selected for this review.
Results
A key theme identified is the potential of microbiomes as diagnostic tools. The findings consistently highlight Lactobacillus as recurrent microbiota. Additionally, Gardnerella, Streptococcus, Prevotella, Aerococcus, Staphylococcus, Proteus, and Bifidobacterium species were frequently observed. This suggests the influential role of these microorganisms in sha** female urological and reproductive health. A deeper understanding of these predominant bacterial genera could offer invaluable insights into healthy physiological states and various disorders. The complex relationship between microbial compositions and diverse health conditions paves the way for novel diagnostic and therapeutic approaches. As we further explore the complexities of microbiomes, their role becomes increasingly crucial in transforming women's health care.
Conclusions
These findings emphasize the need for personalized care, integrating the microbiome into a comprehensive health assessment and treatment framework. This review lays the groundwork for future medical strategies where the microbiome is a pivotal element in both preventive and therapeutic care.


Similar content being viewed by others
References
Marchesi J, Adams D, Fava F, et al. The gut microbiota and host health: a new clinical frontier. Gut. 2015;65(2):330–9. https://doi.org/10.1136/gutjnl-2015-309990.
Champer M, Champer J, Brito I, Messer P, Hou J, Wright J, et al. The role of the vaginal microbiome in gynaecological cancer. BJOG. 2017;125(3):309–15. https://doi.org/10.1111/1471-0528.14631.
Zeng J, Zhang G, Chen C, et al. Alterations in urobiome in patients with bladder cancer and implications for clinical outcome: a single-institution study. Front Cell Infect Microbiol. 2020;10:555508. https://doi.org/10.3389/fcimb.2020.555508.
Walsh D, Hokenstad A, Chen J, et al. Postmenopause as a key factor in the composition of the endometrial cancer microbiome (ECbiome). Sci Rep. 2019;9(1):19213. https://doi.org/10.1038/s41598-019-55720-8.
Kawalec A, Zwołińska D. Emerging role of microbiome in the prevention of urinary tract infections in children. Int J Mol Sci. 2022;23(2):870. https://doi.org/10.3390/ijms23020870.
Hong S, Yang Y, Xu J, **a Q, Wang S, **ao Y. The renal pelvis urobiome in the unilateral kidney stone patients revealed by 2bRAD-M. J Trans Med. 2022;20(1):431. https://doi.org/10.1186/s12967-022-03639-6.
Jones J, Murphy CP, Sleator RD, Culligan EP. The urobiome, urinary tract infections, and the need for alternative therapeutics. Microb Pathog. 2021;161:105295. https://doi.org/10.1016/j.micpath.2021.105295.
Brubaker L, Gourdine J, Siddiqui N, Holland A, Halverson T, Limeria R et al. Forming consensus to advance urobiome research. mSystems. 2021;6(4)e0137120.. https://doi.org/10.1128/msystems.01371-20.
Colceriu M, Aldea P, Răchişan A, Clichici S, Sevastre-Berghian A, Mocan T. Vesicoureteral reflux and innate immune system: physiology, physiopathology, and clinical aspects. J Clin Med. 2023;12(6):2380. https://doi.org/10.3390/jcm12062380.
Torzewska A, Wiewiura P, Brodecka D, Szczerbiec D, Rozalski A. Potentially probiotic Lactobacillus strains derived from food intensify crystallization caused by Proteus mirabilis in urine. Probio Antimicrobial Proteins. 2020;13(2):441–52. https://doi.org/10.1007/s12602-020-09689-w.
Heidrich V, Inoue L, Asprino P, et al. Choice of 16s ribosomal RNA primers impacts male urinary microbiota profiling. Front Cell Infect Microbiol. 2022;12:862338. https://doi.org/10.3389/fcimb.2022.862338.
Hrbacek J, Tláskal V, Čermák P, Hanáček V, Zachoval R. Bladder microbiota are associated with clinical conditions that extend beyond the urinary tract. Microorganisms. 2022;10(5):874. https://doi.org/10.3390/microorganisms10050874.
Komesu YM, Richter HE, Dinwiddie DL, et al. Methodology for a vaginal and urinary microbiome study in women with mixed urinary incontinence. Int Urogynecol J. 2017;28(5):711–20. https://doi.org/10.1007/s00192-016-3165-7.
Okuyama Y, Okamoto T, Sasaki D, et al. The influence of gut microbiome on progression of overactive bladder symptoms: a community-based 3-year longitudinal study in Aomori, Japan. Int Urol Nephrol. 2022;54(1):9–16. https://doi.org/10.1007/s11255-021-03044-w.
Thomas-White KJ, Gao X, Lin H, . Urinary microbes and postoperative urinary tract infection risk in urogynecologic surgical patients. Int Urogynecol J. 2018;29(12):1797–805. https://doi.org/10.1007/s00192-018-3767-3.
Veit-Rubin N, De Tayrac R, Cartwright R, et al. Abnormal vaginal microbiome associated with vaginal mesh complications. Neurourol Urodyn. 2019;38(8):2255–63. https://doi.org/10.1002/nau.24129.
Chen CX, Carpenter JS, Gao X, et al. Associations between dysmenorrhea symptom-based phenotypes and vaginal microbiome: a pilot study. Nurs Res. 2021;70(4):248–55. https://doi.org/10.1097/NNR.0000000000000510.
Wei W, Zhang X, Tang H, Zeng L, Wu R. Microbiota composition and distribution along the female reproductive tract of women with endometriosis. Ann Clin Microbiol Antimicrob. 2020;19(1):15. https://doi.org/10.1186/s12941-020-00356-0.
Thomas-White KJ, Kliethermes S, Rickey L, et al. Evaluation of the urinary microbiota of women with uncomplicated stress urinary incontinence. Am J Obstet Gynecol. 2017;216(1):55.e1–16. https://doi.org/10.1016/j.ajog.2016.07.049.
Nickel JC, Stephens A, Landis JR, et al. Assessment of the lower urinary tract microbiota during symptom flare in women with urologic chronic pelvic pain syndrome: a MAPP network study. J Urol. 2016;195(2):356–62. https://doi.org/10.1016/j.juro.2015.09.075.
Braundmeier-Fleming A, Russell N, Yang W, et al. Stool-based biomarkers of interstitial cystitis/bladder pain syndrome. Sci Rep. 2016;6:26083. https://doi.org/10.1038/srep26083.
Okamoto T, Hatakeyama S, Imai A, et al. Altered gut microbiome associated with overactive bladder and daily urinary urgency. World J Urol. 2021;39(3):847–53. https://doi.org/10.1007/s00345-020-03243-7.
Li K, Chen C, Zeng J, et al. Interplay between bladder microbiota and overactive bladder symptom severity: a cross-sectional study. BMC Urol. 2022;22:39. https://doi.org/10.1186/s12894-022-00990-0.
Wu P, Chen Y, Zhao J, et al. Urinary microbiome and psychological factors in women with overactive bladder. Front Cell Infect Microbiol. 2017;7:488. https://doi.org/10.3389/fcimb.2017.00488.
Curtiss N, Balachandran A, Krska L, Peppiatt-Wildman C, Wildman S, Duckett J. A case controlled study examining the bladder microbiome in women with Overactive Bladder (OAB) and healthy controls. Eur J Obstet Gynecol Reprod Biol. 2017;214:31–5. https://doi.org/10.1016/j.ejogrb.2017.04.040.
Karstens L, Asquith M, Davin S, et al. Does the urinary microbiome play a role in urgency urinary incontinence and its severity? Front Cell Infect Microbiol. 2016;6:78. https://doi.org/10.3389/fcimb.2016.00078.
Chen Z, Phan MD, Bates LJ, et al. The urinary microbiome in patients with refractory urge incontinence and recurrent urinary tract infection. Int Urogynecol J. 2018;29(12):1775–82. https://doi.org/10.1007/s00192-018-3679-2.
Pearce MM, Hilt EE, Rosenfeld AB, et al. The female urinary microbiome: a comparison of women with and without urgency urinary incontinence. mBio. 2014;5(4):e01283–14. https://doi.org/10.1128/mBio.01283-14.
Nickel JC, Stephens-Shields AJ, Landis JR, et al. A culture-independent analysis of the microbiota of female interstitial cystitis/bladder pain syndrome participants in the MAPP research network. J Clin Med. 2019;8(3):415. https://doi.org/10.3390/jcm8030415.
Abernethy MG, Rosenfeld A, White JR, Mueller MG, Lewicky-Gaupp C, Kenton K. Urinary microbiome and cytokine levels in women with interstitial cystitis. Obstet Gynecol. 2017;129(3):500–6. https://doi.org/10.1097/AOG.0000000000001892.
Arya LA, Richter HE, Jelovsek E, et al. Metabolites and microbial composition of stool of women with fecal incontinence: study design and methods. Neurourol Urodyn. 2018;37(2):634–41. https://doi.org/10.1002/nau.23360.
Wessels JM, Domínguez MA, Leyland NA, Agarwal SK, Foster WG. Endometrial microbiota is more diverse in people with endometriosis than symptomatic controls. Sci Rep. 2021;11(1):18877. https://doi.org/10.1038/s41598-021-98380-3.
Price TK, Lin H, Gao X, et al. Bladder bacterial diversity differs in continent and incontinent women: a cross-sectional study. Am J Obstet Gynecol. 2020;223(5):729.e1–10. https://doi.org/10.1016/j.ajog.2020.04.033.
Chao X, Liu Y, Fan Q, Shi H, Wang S, Lang J. The role of the vaginal microbiome in distinguishing female chronic pelvic pain caused by endometriosis/adenomyosis. Ann Trans Med. 2021;9(9):771. https://doi.org/10.21037/atm-20-4586.
Li G, Li W, Song B, et al. Differences in the gut microbiome of women with and without hypoactive sexual desire disorder: case control study. J Med Internet Res. 2021;23(2):e25342. https://doi.org/10.2196/25342.
Pearce MM, Zilliox MJ, Rosenfeld AB, et al. The female urinary microbiome in urgency urinary incontinence. Am J Obstet Gynecol. 2015;213(3):347.e1–11. https://doi.org/10.1016/j.ajog.2015.07.009.
Nardos R, Leung ET, Dahl EM, et al. Network-based differences in the vaginal and bladder microbial communities between women with and without urgency urinary incontinence. Front Cell Infect Microbiol. 2022;12:759156. https://doi.org/10.3389/fcimb.2022.759156.
Author information
Authors and Affiliations
Contributions
G.B.: contributed to the conception and design of the study, acquisition and analysis of data, drafting of the article, and approved the final version to be published; P.K.: significantly contributed to the design of the study, interpretation of data, revising the article critically for important intellectual content, and final approval of the version to be published; T.M.: Contributed to the acquisition of data, analysis, drafting and revising the article, and final approval of the version to be published; D.B.: assisted in data acquisition, analysis, and interpretation, participated in drafting the article, and gave final approval of the version to be published; D.C.: involved in data analysis, interpretation, critical revision of the article for key intellectual content, and approved the final version to be published.
Corresponding author
Ethics declarations
Conflicts of Interest
None.
Additional information
Handling Editor: Annette Kuhn
Editor in Chief: Kaven Baessler
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Balaouras, G., Kostoulas, P., Mikos, T. et al. The Study of Microbiome of the Female Genital Area in Relation to Pelvic Floor Dysfunction: A Systematic Review. Int Urogynecol J (2024). https://doi.org/10.1007/s00192-024-05821-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00192-024-05821-4