Abstract
Introduction and hypothesis
The objective was to investigate the expression of endothelial nitric oxide synthase (eNOS) and phosphodiesterase (PDE) 5 in vaginal tissue of premenopausal women experiencing stress urinary incontinence (SUI) with and without sexual dysfunction.
Methods
Women presenting for treatment of SUI were screened using the Female Sexual Function Index (FSFI) and 10 were selected who met the criteria for female sexual dysfunction (FSD) and 10 asymptomatic controls. Vaginal tissue specimens were obtained from those premenopausal women aged ≥40 years who had had sexual activity ≥2 times every month for the last 6 months and who were scheduled to undergo surgery for SUI. FSD criteria was FSFI scores <18 and arousal domain scores <3. The control group had FSFI scores ≥26 and individual domain scores ≥4. The expressions of eNOS and PDE 5 were compared in the two groups using immunofluorescence staining and western blotting.
Results
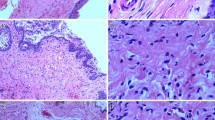
The mean total FSFI scores were 30.4 ± 2.6 and 15.3 ± 2.3 in the control and FSD groups respectively. In immunofluorescence staining, eNOS and PDE5 were localized in the vaginal epithelium. In western blotting, the expressions of eNOS and PDE5 were significantly lower in the FSD group than in the control group (p = 0.003 and p = 0.038 respectively).
Conclusions
eNOS and PDE5 in the vagina may play important roles in the pathophysiology of FSD.


Similar content being viewed by others
Abbreviations
- cGMP:
-
Cyclic guanosine monophosphate
- eNOS:
-
Endothelial nitric oxide synthase
- FSD:
-
Female sexual dysfunction
- FSFI:
-
Female Sexual Function Index
- NO:
-
Nitric oxide
- PBS:
-
Phosphate-buffered saline
- PDE:
-
Phosphodiesterase
- SUI:
-
Stress urinary incontinence
References
Witting K, Santtila P, Jern P, Varjonen M, Wager I, Hoglund M, et al. Evaluation of the female sexual function index in a population based sample from Finland. Arch Sex Behav. 2008;37:912–24. doi:10.1007/s10508-007-9287-8.
Song SH, Jeon H, Kim SW, Paick JS, Son H. The prevalence and risk factors of female sexual dysfunction in young Korean women: an internet-based survey. J Sex Med. 2008;5:1694–701. doi:10.1111/j.1743-6109.2008.00840.x.
Min K, O’Connell L, Munarriz R, Huang YH, Choi S, Kim N, et al. Experimental models for the investigation of female sexual function and dysfunction. Int J Impot Res. 2001;13:151–6.
Giuliano F, Rampin O, Allard J. Neurophysiology and pharmacology of female genital sexual response. J Sex Marital Ther. 2002;28 Suppl 1:101–21. doi:10.1080/00926230252851230.
Batra S, Al-Hijji J. Characterization of nitric oxide synthase activity in rabbit uterus and vagina: downregulation by estrogen. Life Sci. 1998;62:2093–100.
Giraldi A, Alm P, Werkstrom V, Myllymaki L, Wagner G, Andersson KE. Morphological and functional characterization of a rat vaginal smooth muscle sphincter. Int J Impot Res. 2002;14:271–82. doi:10.1038/sj.ijir.3900886.
D’Amati G, di Gioia CR, Bologna M, Giordano D, Giorgi M, Dolci S, et al. Type 5 phosphodiesterase expression in the human vagina. Urology. 2002;60:191–5.
Pace G, Palumbo P, Miconi G, Silvestri V, Cifone MG, Vicentini C. PDE-5 and NOS II mRNA expression in menopausal women: a molecular biology study. World J Urol. 2011;29:243–8. doi:10.1007/s00345-010-0517-7.
Bertin J, Dury AY, Ouellet J, Pelletier G, Labrie F. Localization of the androgen-synthesizing enzymes, androgen receptor, and sex steroids in the vagina: possible implications for the treatment of postmenopausal sexual dysfunction. J Sex Med. 2014;11:1949–61. doi:10.1111/jsm.12589.
Baldassarre M, Alvisi S, Berra M, Martelli V, Farina A, Righi A, et al. Changes in vaginal physiology of menopausal women with type 2 diabetes. J Sex Med. 2015;12:1346–55. doi:10.1111/jsm.12906.
Rosen R, Brown C, Heiman J, Leiblum S, Meston C, Shabsigh R, et al. The Female Sexual Function Index (FSFI): a multidimensional self-report instrument for the assessment of female sexual function. J Sex Marital Ther. 2000;26:191–208. doi:10.1080/009262300278597.
Kim HYSH, Park KS, Jeong SJ, Lee JY, Ryu SB. Development of the Korean-version of Female Sexual Function Index (FSFI). Korean J Androl. 2002;20:50–6.
Wiegel M, Meston C, Rosen R. The female sexual function index (FSFI): cross-validation and development of clinical cutoff scores. J Sex Marital Ther. 2005;31:1–20. doi:10.1080/00926230590475206.
Safarinejad MR. Female sexual dysfunction in a population-based study in Iran: prevalence and associated risk factors. Int J Impot Res. 2006;18:382–95. doi:10.1038/sj.ijir.3901440.
Allers KA, Richards N, Sultana S, Sudworth M, Dawkins T, Hawcock AB, et al. I. Slow oscillations in vaginal blood flow: alterations during sexual arousal in rodents and humans. J Sex Med. 2010;7:1074–87. doi:10.1111/j.1743-6109.2009.01465.x.
Kim SW, Jeong SJ, Munarriz R, Kim NN, Goldstein I, Traish AM. Role of the nitric oxide-cyclic GMP pathway in regulation of vaginal blood flow. Int J Impot Res. 2003;15:355–61. doi:10.1038/sj.ijir.3901038.
Kim NN, Stankovic M, Cushman TT, Goldstein I, Munarriz R, Traish AM. Streptozotocin-induced diabetes in the rat is associated with changes in vaginal hemodynamics, morphology and biochemical markers. BMC Physiol. 2006;6:4. doi:10.1186/1472-6793-6-4.
Mullershausen F, Russwurm M, Koesling D, Friebe A. In vivo reconstitution of the negative feedback in nitric oxide/cGMP signaling: role of phosphodiesterase type 5 phosphorylation. Mol Biol Cell. 2004;15:4023–30. doi:10.1091/mbc.E03-12-0890.
Lin G, **n ZC, Lue TF, Lin CS. Up and down-regulation of phosphodiesterase-5 as related to tachyphylaxis and priapism. J Urol. 2003;170:S15–18; discussion S19. doi:10.1097/01.ju.0000075500.11519.e8
Uckert S, Oelke M, Waldkirch E, Stief CG, Albrecht K, Troger HD, et al. Cyclic adenosine monophosphate and cyclic guanosine monophosphate-phosphodiesterase isoenzymes in human vagina: relation to nitric oxide synthase isoforms and vasoactive intestinal polypeptide-containing nerves. Urology. 2005;65:604–10. doi:10.1016/j.urology.2004.10.028.
Sun Q, Huang J, Yang DL, Cao XN, Zhou WL. Activation of beta-adrenergic receptors during sexual arousal facilitates vaginal lubrication by regulating vaginal epithelial Cl(−) secretion. J Sex Med. 2014;11:1936–48. doi:10.1111/jsm.12583.
Mayhan WG. Nitric oxide accounts for histamine-induced increases in macromolecular extravasation. Am J Physiol. 1994;266:H2369–73.
Game X, Roumiguie M, Bouali O, Allard J, Gourdy P, Mazerolles C, et al. Vaginal lubrication after cervicovaginal stimulation is facilitated by phosphodiesterase type 5 inhibition in ovariectomized mice. J Sex Med. 2013;10:1452–60. doi:10.1111/jsm.12127.
Aslan G, Koseoglu H, Sadik O, Gimen S, Cihan A, Esen A. Sexual function in women with urinary incontinence. Int J Impot Res. 2005;17:248–51. doi:10.1038/sj.ijir.3901296.
Su CC, Sun BY, Jiann BP. Association of urinary incontinence and sexual function in women. Int J Urol. 2015;22:109–13. doi:10.1111/iju.12610.
Musicki B, Liu T, Strong TD, Lagoda GA, Bivalacqua TJ, Burnett AL. Post-translational regulation of endothelial nitric oxide synthase (eNOS) by estrogens in the rat vagina. J Sex Med. 2010;7:1768–77. doi:10.1111/j.1743-6109.2010.01750.x.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Rights and permissions
About this article
Cite this article
Cho, K.J., Lee, KS., Choo, MS. et al. Expressions of vaginal endothelial nitric oxide synthase and phosphodiesterase 5 in female sexual dysfunction: a pilot study. Int Urogynecol J 28, 431–436 (2017). https://doi.org/10.1007/s00192-016-3159-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00192-016-3159-5