Abstract
Key message
The role of BrKAO2 in leafy head formation was confirmed by using two allelic Chinese cabbage mutants.
Abstract
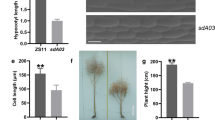
Chinese cabbage yield and quality are determined by leafy head formation. Cloning and characterising the key genes regulating leafy head formation are essential for its varietal improvement. We used an EMS-mutagenised population of the heading type ‘FT’ Chinese cabbage line and identified two allelic non-heading mutants, i.e. nhm3-1 and nhm3-2. Genetic analysis showed that the mutant trait was controlled by a single recessive gene. MutMap and Kompetitive Allele Specific PCR genoty** revealed that BraA05g012440.3C was the candidate gene, which encodes ent-kaurenoic acid oxidase 2 in gibberellin (GA) biosynthetic pathway. It was named BrKAO2. Two non-synonymous mutations in the second BrKAO2 exon, respectively, accounted for the mutant phenotypes of nhm3-1 and nhm3-2. BrKAO2 was expressed during all leaf development stages, and there were no significant differences between the wild type and mutants in terms of BrKAO2 expression. The mutant phenotypes were restored to the wild type via exogenous GA3 application. RNA-Seq was performed on wild-type ‘FT’, nhm3-1, and nhm3-1 + GA3 rosette leaves, and several key genes involved in GA biosynthesis, signal transduction, and leafy head development were identified. These findings indicate that BrKAO2 is responsible for the leafy head formation in nhm3 mutants.









Similar content being viewed by others
Data availability
All data generated and analysed in this study are available upon request. Transcriptome sequencing data have been deposited in the NCBI Gene Expression Omnibus (GEO) Database under accession number GSE183651. The sequence data of MutMap have been deposited in the NCBI Sequence Read Archive (SRA) repository under accession numbers SRR15803269, SRR15828094, and SRR15829494.
References
Byrne ME, Barley R, Curtis M, Arroyo JM, Dunham M, Hudson A, Martienssen RA (2000) Asymmetric leaves1 mediates leaf patterning and stem cell function in Arabidopsis. Nature 408:967–971
Chen ML, Fu XM, Liu JQ, Ye TT, Hou SY, Huang YQ, Yuan BF, Wu Y, Feng YQ (2012) Highly sensitive and quantitative profiling of acidic phytohormones using derivatization approach coupled with nano-LC–ESI–QTOF–MS analysis. J Chromatogr B 905:67–74
Cheng CX, Jiao C, Singer SD, Gao M, Xu XZ, Zhou YM, Li Z, Fei ZJ, Wang YJ, Wang XP (2015) Gibberellin-induced changes in the transcriptome of grapevine (Vitis labrusca × V. vinifera) cv. Kyoho flowers. BMC Genomics 16:128
Cheng F, Sun RF, Hou XL et al (2016) Subgenome parallel selection is associated with morphotype diversification and convergent crop domestication in Brassica rapa and Brassica oleracea. Nat Genet 48:1218–1224
Davidson SE, Elliott RC, Helliwell CA, Poole AT, Reid JB (2003) The pea gene NA encodes ent-kaurenoic acid oxidase. Plant Physiol 131:335–344
Daviere JM, Achard P (2013) Gibberellin Signaling in Plants. Development 140:1147–1151
Dill A, Thomas SG, Hu J, Steber CM, Sun TP (2004) The Arabidopsis F-box protein SLEEPY1 targets gibberellin signaling repressors for gibberellin-induced degradation. Plant Cell 16:1392–1405
Elliott RC, Ross JJ, Smith JJ, Lester DR, Reid JB (2001) Feed-forward regulation of gibberellin deactivation in pea. J Plant Growth Regul 20:87–94
Eriksson S, Böhlenius H, Moritz T, Nilsson O (2006) GA4 is the active gibberellin in the regulation of LEAFY transcription and Arabidopsis floral initiation. Plant Cell 18:2172–2181
Eshed Y, Baum SF, Perea JV, Bowman JL (2001) Establishment of polarity in lateral organs of plants. Curr Biol 11:1251–1260
Fahlgren N, Montgomery TA, Howell MD, Allen E, Dvorak SK, Alexander AL, Carrington JC (2006) Regulation of AUXIN RESPONSE FACTOR3 by TAS3 ta-siRNA affects developmental timing and patterning in Arabidopsis. Curr Biol 16:939–944
Fambrini M, Mariotti L, Parlanti S, Picciarelli P, Salvini M, Ceccarelli N, Pugliesi C (2011) The extreme dwarf phenotype of the GA-sensitive mutant of sunflower, dwarf2, is generated by a deletion in the ent-kaurenoic acid oxidase1 (HaKAO1) gene sequence. Plant Mol Biol 75:431–450
Fleet CM, Sun TP (2005) A DELLAcate balance: the role of gibberellin in plant morphogenesis. Curr Opin Plant Biol 8:77–85
Fu XD, Richards DE, Fleck B, **e DX, Burton N, Harberd NP (2004) The Arabidopsis mutant sleepy1gar2-1 protein promotes plant growth by increasing the affi nity of the SCFSLY1 E3 ubiquitin ligase for DELLA protein substrates. Plant Cell 16:1406–1418
Gao LW, Lyu SW, Tang J, Zhou DY, Bonnema G, **ao D, Hou XL, Zhang CW (2017) Genome-wide analysis of auxin transport genes identifies the hormone responsive patterns associated with leafy head formation in Chinese cabbage. Sci Rep 7:42229
Gao Y, Huang SN, Qu GY, Fu W, Zhang MD, Liu ZY, Feng H (2020) The mutation of ent-kaurene synthase, a key enzyme in the gibberellins biosynthesis, confers a non-heading phenotype to Chinese cabbage (Brassica rapa L. ssp. pekinensis). Hortic Res 7:178
Griffiths J, Murase K, Rieu I et al (2006) Genetic characterization and functional analysis of the GID1 gibberellin receptors in Arabidopsis. Plant Cell 18:3399–3414
Hartweck LM (2008) Gibberellin signaling. Planta 229:1–13
Hasson A, Plessis A, Blein T, Adroher B, Grigg SP, Tsiantis M, Boudaoud A, Damerval C, Laufs P (2011) Evolution and diverse roles of the CUP-SHAPED COTYLEDON genes in Arabidopsis leaf development. Plant Cell 23:54–68
Hay A, Tsiantis M (2010) KNOX genes: versatile regulators of plant development and diversity. Development 137:3153–3165
He YK, Xue WX, Sun YD, Yu XH, Liu PL (2000) Leafy head formation of the progenies of transgenic plants of Chinese cabbage with exogenous auxin genes. Cell Res 10:151–160
Hedden P, Phillips AL (2000) Gibberellin metabolism: New insights revealed by the genes. Trends Plant Sci 5:523–530
Hedden P, Thomas SG (2012) Gibberellin biosynthesis and its regulation. Biochem J 444:11–25
Helliwell CA, Chandler PM, Poole A, Dennis ES, Peacock WJ (2001) The CYP88A cytochrome P450, ent-kaurenoic acid oxidase, catalyzes three steps of the gibberellin biosynthesis pathway. PNAS 98:2065–2207
Huang SN, Liu ZY, Yao RP, Li DY, Zhang T, Li X, Hou L, Wang YH, Tang XY, Feng H (2016) Candidate gene prediction for a petal degeneration mutant, pdm, of the Chinese cabbage (Brassica campestris ssp. pekinensis) by using fine map** and transcriptome analysis. Mol Breed 36:26
Hunter C, Sun H, Poethig RS (2003) The Arabidopsis heterochronic gene ZIPPY is an ARGONAUTE family member. Curr Biol 13:1734–1739
Ito H, Kato T (1957) Studies on the head formation of Chinese cabbage: histological and physiological studies of head formation. J Jpn Soc Hortic Sci 26:154–204
Iwakawa H, Iwasaki M, Kojima S, Ueno Y, Soma T, Tanaka H, Semiarti E, Machida Y, Machida C (2007) Expression of the ASYMMETRIC LEAVES2 gene in the adaxial domain of Arabidopsis leaves represses cell proliferation in this domain and is critical for the development of properly expanded leaves. Plant J 51:173–184
Iwasaki M, Takahashi H, Iwakawa H et al (2013) Dual regulation of ETTIN (ARF3) gene expression by AS1-AS2, which maintains the DNA methylation level, is involved in stabilization of leaf adaxial–abaxial partitioning in Arabidopsis. Development 140:1958–1969
Jung CJ, Hur YY, Yu HJ, Noh JH, Park KS, Lee HJ (2014) Gibberellin application at pre-bloom in grapevines down-regulates the expressions of VvIAA9 and VvARF7, negative regulators of fruit set Initiation, during parthenocarpic fruit development. PLoS ONE 9:e95634
Kalve S, Vos DD, Beemster GTS (2014) Leaf development: a cellular perspective. Front Plant Sci 5:362
Kerstetter RA, Bollman K, Taylor RA, Bomblies K, Poethig RS (2001) KANADI regulates organ polarity in Arabidopsis. Nature 411:706–709
Khlestkina EK, Kumar U, Röder MS (2010) Ent-kaurenoic acid oxidase genes in wheat. Mol Breeding 25:251–258
Kim GT, Cho KH (2006) Recent advances in the genetic regulation of the shape of simple leaves. Physiol Plant 126:494–502
Kim D, Langmead B, Salzberg SL (2015) HISAT: a fast spliced aligner with low memory requirements. Nat Methods 12:357–360
Lei L, Fan ZR, Tang L, Chen F (2011) Molecular cloning and identification of tissue-specific expression of ent-kaurene oxidase gene in Momordica charantia. Afr J Biotechnol 10:15724–15730
Li H, Durbin R (2010) Fast and accurate long-read alignment with Burrows-Wheeler transform. Bioinformatics 26:589–595
Li YF, Fan Y, Jiao Y, Wu J, Zhang Z, Yu XL, Ma Y (2018) Transcriptome profiling of yellow leafy head development during the heading stage in Chinese cabbage (Brassica rapa subsp. pekinesis). Physiol Plantarum 165:800–813
Li J, Jiang JF, Qian Q et al (2011) Mutation of rice BC12/GDD1, which encodes a kinesin-like protein that binds to a GA biosynthesis gene promoter, leads to dwarfism with impaired cell elongation. Plant Cell 23:628–640
Li JR, Zhang XM, Lu Y et al (2019) Characterization of non-heading mutation in heading Chinese cabbage (Brassica rapa L. ssp. pekinensis). Front Plant Sci 10:112
Liang JL, Liu B, Wu J, Cheng F, Wang XW (2016) Genetic variation and divergence of genes involved in leaf adaxial–abaxial polarity establishment in Brassica rapa. Front Plant Sci 7:94
Lin W-C, Shuai B, Springer PS (2003) The Arabidopsis LATERAL ORGAN BOUNDARIES-Domain gene ASYMMETRIC LEAVES2 functions in the repression of KNOX gene expression and in adaxial–abaxial patterning. Plant Cell 15:2241–2252
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCt method. Methods 25:402–408
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15:550
Mao YF, Wu FJ, Yu X, Bai JJ, Zhong WL, He YK (2014) MicroRNA319a-targeted Brassica rapa ssp. pekinensis TCP genes modulate head shape in Chinese cabbage by differential cell division arrest in leaf regions. Plant Physiol 164:710–720
McConnell JR, Emery J, Eshed Y, Bao N, Bowan J, Barton MK (2001) Role of PHABULOSA and PHAVOLUTA in determining radial patterning in shoots. Nature 411:709–713
McKenna A, Hanna M, Banks E et al (2010) The genome analysis toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res 20:1297–1303
Olszewski N, Sun TP, Gubler F (2002) Gibberellin signaling: biosynthesis, catabolism, and response pathways. Plant Cell 14:S61–S80
Otsuga D, DeGuzman B, Prigge MJ, Drews GN, Clark SE (2001) REVOLUTA regulates meristem initiation at lateral positions. Plant J 25:223–236
Ou CQ, Jiang SL, Wang F, Wang ZG, Ma L, Li LW (2013) Cloning and expression analysis of ent-kaurenoic acid oxidase gene (PcKAO1) in Pear. Acta Hortic Sin 40:849–858
Regnault T, Daviere JM, Heintz D, Lange T, Achard P (2014) The gibberellin biosynthetic genes AtKAO1 and AtKAO2 have overlap** roles throughout Arabidopsis development. Plant J 80:462–474
Ren WQ, Wu FJ, Bai JJ, Li XR, Yang X, Xue WX, Liu H, He YK (2020) BcpLH organizes a specific subset of microRNAs to form a leafy head in Chinese cabbage (Brassica rapa ssp. pekinensis). Hortic Res 7:1
Ribeiro DM, Araújo WL, Fernie AR, Schippers JHM, Mueller-Roeber B (2012) Translatome and metabolome effects triggered by gibberellins during rosette growth in Arabidopsis. J Exp Bot 63:2769–2786
Rieu I, Ruiz-Rivero O, Fernandez-Garcia N et al (2008) The gibberellin biosynthetic genes AtGA20ox1 and AtGA20ox2 act, partially redundantly, to promote growth and development throughout the Arabidopsis life cycle. Plant J 53:488–504
Sakamoto T, Miura K, Itoh H et al (2004) An overview of gibberellin metabolism enzyme genes and their related mutants in rice. Plant Physiol 134:1642–1653
Shi ZY, Wang J, Wan XS, Shen GZ, Wang XQ, Zhan JL (2007) Over-expression of rice OsAGO7 gene induces upward curling of the leaf blade that enhanced erect-leaf habit. Planta 226:99–108
Sicard A, Thamm A, Marona C, Lee YW, Wahl V, Stinchcombe JR, Wright SI, Kappel C, Lenhard M (2014) Repeated evolutionary changes of leaf morphology caused by mutations to a homeobox gene. Curr Biol 24:1880–1886
Silverstone AL, Chang CW, Krol E, Sun TP (1997) Developmental regulation of the gibberellin biosynthetic gene GA1 in Arabidopsis thaliana. Plant J 12:9–19
Song J, Guo BJ, Song FW, Peng H, Yao Y, Zhang Y, Sun Q, Ni Z (2011) Genome-wide identification of gibberellins metabolic enzyme genes and expression profiling analysis during seed germination in maize. Gene 482:34–42
Sun TP, Gubler F (2004) Molecular mechanism of gibberellin of signaling in plants. Annu Rev Plant Biol 55:197–223
Thomas SG, Phillips AL, Hedden P (1999) Molecular cloning and functional expression of gibberellin 2-oxidases, multifunctional enzymes involved in gibberellin deactivation. PNAS 96:4698–4703
Vercruyssen L, Verkest A, Gonzalez N et al (2014) ANGUSTIFOLIA3 binds to SWI/SNF chromatin remodeling complexes to regulate transcription during Arabidopsis leaf development. Plant Cell 26:210–229
Wang F, Zhu D, Huang X, Li S, Gong Y, Yao Q, Fu X, Fan LM, Deng XW (2009) Biochemical insights on degradation of Arabidopsis DELLA proteins gained from a cell-free assay system. Plant Cell 21:2378–2390
Wang FD, Li HY, Zhang YH, Li JJ, Li LB, Liu LF, Wang LH, Wang CH, Gao JS (2013) MicroRNA expression analysis of rosette and folding leaves in Chinese cabbage using high-throughput Solexa sequencing. Gene 532:222–229
Wang YL, Wu FJ, Bai JJ, He YK (2014) BrpSPL9 (Brassica rapa ssp. pekinensis SPL9) controls the earliness of heading time in Chinese cabbage. Plant Biotechnol J 12:312–321
Wei CH, Zhu CY, Yang LP, Zhao W, Ma RX, Li H, Zhang Y, Ma JX, Yang JQ, Zhang X (2019) A point mutation resulting in a 13 bp deletion in the coding sequence of Cldf leads to a GA-deficient dwarf phenotype in watermelon. Hortic Res 6:132
Winkler RG, Helentjaris T (1995) The maize Dwarf3 gene encodes a cytochrome P450-mediated early step in gibberellin biosynthesis. Plant Cell 7:1307–1317
Yamaguchi S (2008) Gibberellin metabolism and its regulation. Annu Rev Plant Biol 59:225–251
Yamaguchi S, Kamiya Y (2000) Gibberellin biosynthesis: Its regulation by endogenous and environmental signals. Plant Cell Physiol 41:251–257
Yang YH, Zhang YD, Zhu Z, Zhao L, Chen T, Zhao QY, Wang CL (2010) Effects of gibberellic acid 3 and abscisic acid on growth, physiological characteristics and gene expression of GA20ox2 and GA3ox2 in different rice cultivars. Chin J Rice Sci 24:433–437
Yoshikawa M, Peragine A, Park MY, Poethig RS (2005) A pathway for the biogenesis of trans-acting siRNAs in Arabidopsis. Gene Dev 19:2164–2175
Yu XH, Peng JS, Feng XZ, Yang SX, Zheng ZR, Tang XR, Shen RJ, **lin Liu PL, He YK (2000) Cloning and structural and expressional characterization of BcpLH gene preferentially expressed in folding leaf of Chinese cabbage. Sci China Ser C 43:321–329
Yu X, Wang H, Zhong WL, Bai JJ, Liu PL, He YK (2013) QTL Map** of leafy heads by genome resequencing in the RIL population of Brassica rapa. PLoS ONE 8:e76059
Yu J, Gao LW, Liu WS, Song LX, **ao D, Liu TK, Hou XL, Zhang CW (2019) Transcription coactivator ANGUSTIFOLIA3 (AN3) regulates leafy head formation in Chinese cabbage. Front Plant Sci 10:520
Zgurski JM, Sharma R, Bolokoski DA, Schultz EA (2005) Asymmetric auxin response precedes asymmetric growth and differentiation of asymmetric leaf1 and asymmetric leaf2 Arabidopsis leaves. Plant Cell 17:77–91
Zhang X, Liu ZY, Wang P, Wang QS, Yang S, Feng H (2013) Fine map** of Br Wax1, a gene controlling cuticular wax biosynthesis in Chinese cabbage (Brassica rapa L. ssp. pekinensis). Mol Breeding 32:867–874
Zhang XL, Su YB, Liu YM, Fang ZY, Yang LM, Zhuang M, Zhang YY, Li ZS, Lv HY (2016a) Genetic analysis and QTL map** of traits related to head shape in cabbage (Brassica oleracea var. capitata L.). Sci Hortic 207:82–88
Zhang CW, Wei YP, **ao D, Gao LW, Lyu SW, Hou XL, Bouuema G (2016b) Transcriptomic and proteomic analyses provide new insights into the regulation mechanism of low-temperature-induced leafy head formation in Chinese cabbage. J Proteomics 144:1–10
Zhang YY, Liang JL, Cai X, Chen HX, Wu J, Lin RM, Cheng F, Wang XW (2021) Divergence of three BRX homoeologs in Brassica rapa and its effect on leaf morphology. Hortic Res 8:68
Acknowledgements
We would like to thank Editage (https://www.editage.cn) for English language editing.
Funding
This study was funded by the National Natural Science Foundation of China (31801854) and Liaoning Provincial Natural Science Foundation of China (2021-MS-231).
Author information
Authors and Affiliations
Contributions
HF and SH designed the experiments; SH and YG performed the research; MX, JX, RL, and SS participated in the research; CL and ZL provided technical help; YZ and XY participated in data analysis; and SH supervised this study and wrote the manuscript. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Richard G. F. Visser.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
122_2022_4126_MOESM2_ESM.tif
Fig. S2 Expression analysis of BrKAO2 and BrKAO1 in wild-type ‘FT’ and nhm3-1 and nhm3–2 mutants. BrKAO2 and BrKAO1 expression levels in wild-type ‘FT’ were used as references for relative expression. *significant differences in expression levels at P < 0.05 (TIF 648 kb)
Rights and permissions
About this article
Cite this article
Huang, S., Gao, Y., Xue, M. et al. BrKAO2 mutations disrupt leafy head formation in Chinese cabbage (Brassica rapa L. ssp. pekinensis). Theor Appl Genet 135, 2453–2468 (2022). https://doi.org/10.1007/s00122-022-04126-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-022-04126-8