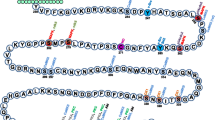
Abstract
Connexins (Cxs) are ubiquitous transmembrane proteins that possess both channel function (e.g., formations of gap junction and hemichannel) and non-channel properties (e.g., gene transcription and protein-protein interaction). Several factors have been identified to play a role in the regulation of Cxs, which include those acting intracellularly, as redox potential, pH, intramolecular interactions, and post-translational modifications (e.g., phosphorylation, S-nitrosylation) as well as those acting extracellularly, such as Ca2+ and Mg2+. The relationship between redox signaling and Cxs attracts considerable attention in recent years. There is ample evidence showing that redox signaling molecules (e.g., hydrogen peroxide (H2O2), nitric oxide (NO)) affect Cxs-based channel function while the opening of Cx channels also triggers the transfer of various redox-related metabolites (e.g., reactive oxygen species, glutathione, nicotinamide adenine dinucleotide, and NO). On the basis of these evidences, we propose the existence of redox-Cxs crosstalk. In this review, we briefly discuss the interaction between redox signaling and Cxs and the implications of the intersection in disease pathology and future therapeutic interventions.


Similar content being viewed by others
Abbreviations
- BTF3:
-
Basic transcription factor-3
- Cav-1:
-
Caveolin-1
- CBX:
-
Carbenoxolone
- Cx:
-
Connexin
- Cys:
-
Cysteine
- DTT:
-
Dithiothreitol
- EL:
-
Extracellular loop
- eNOS:
-
Endothelial nitric oxide synthase
- GJ:
-
Gap junction
- GSH:
-
Glutathione
- GSNO:
-
S-nitrosoglutathione
- H2O2 :
-
Hydrogen peroxide
- HC:
-
Hemichannel
- HM:
-
Hematopoietic microenvironment
- HSCs:
-
Hematopoietic stem cells
- HSC/P:
-
HSC and progenitor
- IL:
-
Intracellular loop
- IP3:
-
Inositol 1, 4, 5-trisphosphate
- NAD+ :
-
Nicotinamide adenine dinucleotide
- NADPH:
-
Nicotinamide adenine dinucleotide phosphate hydrogen
- NO:
-
Nitric oxide
- NOX2:
-
NADPH oxidase 2
- OS:
-
Oxidative stress
- PKG:
-
Protein kinase G
- ROS:
-
Reactive oxygen species
- SNO:
-
S-nitrosylation
- TM:
-
Transmembrane domain
- ZO-1:
-
Zonula occludens-1
References
Bonacquisti EE, Nguyen J (2019) Connexin 43 (Cx43) in cancer: Implications for therapeutic approaches via gap junctions. Cancer Lett 442:439–444
Abascal F, Zardoya R (2013) Evolutionary analyses of gap junction protein families. Biochim Biophys Acta 1828:4–14
Laird DW, Lampe PD (2018) Therapeutic strategies targeting connexins. Nat Rev Drug Discov 17:905–921
Söhl G, Willecke K (2004) Gap junctions and the connexin protein family. Cardiovasc Res 62:228–232
Oshima A (2014) Structure and closure of connexin gap junction channels. FEBS Lett 588:1230–1237
Bai D, Yue B, Aoyama H (2018) Crucial motifs and residues in the extracellular loops influence the formation and specificity of connexin docking. Biochim Biophys Acta Biomembr 1860:9–21
Alexander DB, Goldberg GS (2003) Transfer of biologically important molecules between cells through gap junction channels. Curr Med Chem 10:2045–2058
Anselmi F, Hernandez VH, Crispino G, Seydel A, Ortolano S, Roper SD, Kessaris N, Richardson W, Rickheit G, Filippov MA, Monyer H, Mammano F (2008) ATP release through connexin hemichannels and gap junction transfer of second messengers propagate Ca2+ signals across the inner ear. Proc Natl Acad Sci U S A 105:18770–18775
Hoorelbeke D, Decrock E, De Smet M, De Bock M, Descamps B, Van Haver V, Delvaeye T, Krysko DV, Vanhove C, Bultynck G et al (2020) Cx43 channels and signaling via IP(3)/Ca(2+), ATP, and ROS/NO propagate radiation-induced DNA damage to non-irradiated brain microvascular endothelial cells. Cell Death Dis 11:194
Slavi N, Rubinos C, Li L, Sellitto C, White TW, Mathias R, Srinivas M (2014) Connexin 46 (cx46) gap junctions provide a pathway for the delivery of glutathione to the lens nucleus. J Biol Chem 289:32694–32702
Kotini M, Barriga EH, Leslie J, Gentzel M, Rauschenberger V, Schambony A, Mayor R (2018) Gap junction protein Connexin-43 is a direct transcriptional regulator of N-cadherin in vivo. Nat Commun 9:3846
Sorgen PL, Duffy HS, Sahoo P, Coombs W, Delmar M, Spray DC (2004) Structural changes in the carboxyl terminus of the gap junction protein connexin43 indicates signaling between binding domains for c-Src and zonula occludens-1. J Biol Chem 279:54695–54701
Spagnol G, Trease AJ, Zheng L, Gutierrez M, Basu I, Sarmiento C, Moore G, Cervantes M, Sorgen PL (2018) Connexin43 carboxyl-terminal domain directly interacts with β-catenin. Int J Mol Sci 19. https://doi.org/10.3390/ijms19061562
Saidi Brikci-Nigassa A, Clement MJ, Ha-Duong T, Adjadj E, Ziani L, Pastre D, Curmi PA, Savarin P (2012) Phosphorylation controls the interaction of the connexin43 C-terminal domain with tubulin and microtubules. Biochemistry 51:4331–4342
Langlois S, Cowan KN, Shao Q, Cowan BJ, Laird DW (2008) Caveolin-1 and -2 interact with connexin43 and regulate gap junctional intercellular communication in keratinocytes. Mol Biol Cell 19:912–928
Retamal MA, Schalper KA, Shoji KF, Bennett MV, Sáez JC (2007) Opening of connexin 43 hemichannels is increased by lowering intracellular redox potential. Proc Natl Acad Sci U S A 104:8322–8327
Rozas-Villanueva MF, Casanello P, Retamal MA (2020) Role of ROS/RNS in preeclampsia: are connexins the missing piece? Int J Mol Sci 21. https://doi.org/10.3390/ijms21134698
Hua R, Zhang J, Riquelme MA, Jiang JX (2021) Connexin gap junctions and hemichannels link oxidative stress to skeletal physiology and pathology. Curr Osteoporos Rep 19:66–74
Skeberdis VA, Rimkute L, Skeberdyte A, Paulauskas N, Bukauskas FF (2011) pH-dependent modulation of connexin-based gap junctional uncouplers. J Physiol 589:3495–3506
Aasen T, Johnstone S, Vidal-Brime L, Lynn KS, Koval M (2018) Connexins: synthesis, post-translational modifications, and trafficking in health and disease. Int J Mol Sci 19. https://doi.org/10.3390/ijms19051296
Pinheiro AR, Paramos-de-Carvalho D, Certal M, Costa C, Magalhães-Cardoso MT, Ferreirinha F, Costa MA, Correia-de-Sá P (2013) Bradykinin-induced Ca2+ signaling in human subcutaneous fibroblasts involves ATP release via hemichannels leading to P2Y12 receptors activation. Cell Commun Signal 11:70
Tejada MG, Sudhakar S, Kim NK, Aoyama H, Shilton BH, Bai D (2018) Variants with increased negative electrostatic potential in the Cx50 gap junction pore increased unitary channel conductance and magnesium modulation. Biochem J 475:3315–3330
Zhou Y, Gao L, **a P, Zhao J, Li W, Zhou Y, Wei Q, Wu Q, Wu Q, Sun D, Gao K (2021) Glycyrrhetinic acid protects renal tubular cells against oxidative injury via reciprocal regulation of JNK-connexin 43-thioredoxin 1 signaling. Front Pharmacol 12:619567
Huang Y, Mao Z, Zhang Z, Obata F, Yang X, Zhang X, Huang Y, Mitsui T, Fan J, Takeda M, Yao J (2019) Connexin43 contributes to inflammasome activation and lipopolysaccharide-initiated acute renal injury via modulation of intracellular oxidative status. Antioxid Redox Signal 31:1194–1212
Retamal MA, Orellana VP, Arévalo NJ, Rojas CG, Arjona RJ, Alcaíno CA, González W, Canan JG, Moraga-Amaro R, Stehberg J, Reuss L, Altenberg GA (2019) Cx46 hemichannel modulation by nitric oxide: role of the fourth transmembrane helix cysteine and its possible involvement in cataract formation. Nitric Oxide 86:54–62
Bruzzone S, Guida L, Zocchi E, Franco L, De Flora A (2001) Connexin 43 hemi channels mediate Ca2+-regulated transmembrane NAD+ fluxes in intact cells. FASEB J 15:10–12
Retamal MA, Cortés CJ, Reuss L, Bennett MV, Sáez JC (2006) S-nitrosylation and permeation through connexin 43 hemichannels in astrocytes: induction by oxidant stress and reversal by reducing agents. Proc Natl Acad Sci U S A 103:4475–4480
Farnsworth NL, Walter RL, Hemmati A, Westacott MJ, Benninger RK (2016) Low level pro-inflammatory cytokines decrease connexin36 gap junction coupling in mouse and human islets through nitric oxide-mediated protein kinase Cδ. J Biol Chem 291:3184–3196
Lillo MA, Himelman E, Shirokova N, **e LH, Fraidenraich D, Contreras JE (2019) S-nitrosylation of connexin43 hemichannels elicits cardiac stress-induced arrhythmias in Duchenne muscular dystrophy mice. JCI Insight 4. https://doi.org/10.1172/jci.insight.130091
Barnett SD, Asif H, Anderson M, Buxton ILO (2021) Novel tocolytic strategy: modulating Cx43 activity by S-nitrosation. J Pharmacol Exp Ther 376:444–453
Vielma AZ, Boric MP, Gonzalez DR (2020) Apocynin treatment prevents cardiac connexin 43 hemichannels hyperactivity by reducing nitroso-redox stress in Mdx mice. Int J Mol Sci 21. https://doi.org/10.3390/ijms21155415
Lu C, McMahon DG (1997) Modulation of hybrid bass retinal gap junctional channel gating by nitric oxide. J Physiol 499(Pt 3):689–699
Patel LS, Mitchell CK, Dubinsky WP, O'Brien J (2006) Regulation of gap junction coupling through the neuronal connexin Cx35 by nitric oxide and cGMP. Cell Commun Adhes 13:41–54
Kameritsch P, Hoffmann A, Pohl U (2003) Opposing effects of nitric oxide on different connexins expressed in the vascular system. Cell Commun Adhes 10:305–309
McKinnon RL, Bolon ML, Wang HX, Swarbreck S, Kidder GM, Simon AM, Tyml K (2009) Reduction of electrical coupling between microvascular endothelial cells by NO depends on connexin37. Am J Physiol Heart Circ Physiol 297:H93–h101
Straub AC, Billaud M, Johnstone SR, Best AK, Yemen S, Dwyer ST, Looft-Wilson R, Lysiak JJ, Gaston B, Palmer L, Isakson BE (2011) Compartmentalized connexin 43 s-nitrosylation/denitrosylation regulates heterocellular communication in the vessel wall. Arterioscler Thromb Vasc Biol 31:399–407
Lin D, Takemoto DJ (2005) Oxidative activation of protein kinase Cgamma through the C1 domain. Effects on gap junctions J Biol Chem 280:13682–13693
Rouach N, Calvo CF, Duquennoy H, Glowinski J, Giaume C (2004) Hydrogen peroxide increases gap junctional communication and induces astrocyte toxicity: regulation by brain macrophages. Glia 45:28–38
Hu J, Engman L, Cotgreave IA (1995) Redox-active chalcogen-containing glutathione peroxidase mimetics and antioxidants inhibit tumour promoter-induced downregulation of gap junctional intercellular communication between WB-F344 liver epithelial cells. Carcinogenesis 16:1815–1824
Contreras JE, Sánchez HA, Eugenin EA, Speidel D, Theis M, Willecke K, Bukauskas FF, Bennett MV, Sáez JC (2002) Metabolic inhibition induces opening of unapposed connexin 43 gap junction hemichannels and reduces gap junctional communication in cortical astrocytes in culture. Proc Natl Acad Sci U S A 99:495–500
Anderson ME, Powrie F, Puri RN, Meister A (1985) Glutathione monoethyl ester: preparation, uptake by tissues, and conversion to glutathione. Arch Biochem Biophys 239:538–548
Retamal MA, Yin S, Altenberg GA, Reuss L (2009) Modulation of Cx46 hemichannels by nitric oxide. Am J Phys Cell Phys 296:C1356–C1363
Retamal MA, Fiori MC, Fernandez-Olivares A, Linsambarth S, Peña F, Quintana D, Stehberg J, Altenberg GA (1865) 4-Hydroxynonenal induces Cx46 hemichannel inhibition through its carbonylation. Biochim Biophys Acta Mol Cell Biol Lipids 2020:158705
Elias LA, Wang DD, Kriegstein AR (2007) Gap junction adhesion is necessary for radial migration in the neocortex. Nature 448:901–907
Theis M, Söhl G, Eiberger J, Willecke K (2005) Emerging complexities in identity and function of glial connexins. Trends Neurosci 28:188–195
Crespin S, Fromont G, Wager M, Levillain P, Cronier L, Monvoisin A, Defamie N, Mesnil M (2016) Expression of a gap junction protein, connexin43, in a large panel of human gliomas: new insights. Cancer Med 5:1742–1752
Sirnes S, Bruun J, Kolberg M, Kjenseth A, Lind GE, Svindland A, Brech A, Nesbakken A, Lothe RA, Leithe E, Rivedal E (2012) Connexin43 acts as a colorectal cancer tumor suppressor and predicts disease outcome. Int J Cancer 131:570–581
Aasen T, Sansano I, Montero M, Romagosa C, Temprana-Salvador J, Martínez-Marti A, Moliné T, Hernández-Losa J, Ramón y Cajal S (2019) Insight into the role and regulation of gap junction genes in lung cancer and identification of nuclear Cx43 as a putative biomarker of poor prognosis. Cancers (Basel) 11 https://doi.org/10.3390/cancers11030320
Gago-Fuentes R, Fernández-Puente P, Megias D, Carpintero-Fernández P, Mateos J, Acea B, Fonseca E, Blanco FJ, Mayan MD (2015) Proteomic analysis of connexin 43 reveals novel interactors related to osteoarthritis. Mol Cell Proteomics 14:1831–1845
Dang X, Doble BW, Kardami E (2003) The carboxy-tail of connexin-43 localizes to the nucleus and inhibits cell growth. Mol Cell Biochem 242:35–38
Kotova A, Timonina K, Zoidl GR (2020) Endocytosis of connexin 36 is mediated by interaction with caveolin-1. Int J Mol Sci 21. https://doi.org/10.3390/ijms21155401
Luanpitpong S, Talbott SJ, Rojanasakul Y, Nimmannit U, Pongrakhananon V, Wang L, Chanvorachote P (2010) Regulation of lung cancer cell migration and invasion by reactive oxygen species and caveolin-1. J Biol Chem 285:38832–38840
Taniguchi Ishikawa E, Gonzalez-Nieto D, Ghiaur G, Dunn SK, Ficker AM, Murali B, Madhu M, Gutstein DE, Fishman GI, Barrio LC, Cancelas JA (2012) Connexin-43 prevents hematopoietic stem cell senescence through transfer of reactive oxygen species to bone marrow stromal cells. Proc Natl Acad Sci U S A 109:9071–9076
Yuan D, Li X, Luo C, Li X, Cheng N, Ji H, Qiu R, Luo G, Chen C, Hei Z (2019) Inhibition of gap junction composed of Cx43 prevents against acute kidney injury following liver transplantation. Cell Death Dis 10:767
Gu Y, Huang F, Wang Y, Chen C, Wu S, Zhou S, Hei Z, Yuan D (2018) Connexin32 plays a crucial role in ROS-mediated endoplasmic reticulum stress apoptosis signaling pathway in ischemia reperfusion-induced acute kidney injury. J Transl Med 16:117
Chi Y, Zhang X, Zhang Z, Mitsui T, Kamiyama M, Takeda M, Yao J (2016) Connexin43 hemichannels contributes to the disassembly of cell junctions through modulation of intracellular oxidative status. Redox Biol 9:198–209
Shi W, Riquelme MA, Gu S, Jiang JX (2018) Connexin hemichannels mediate glutathione transport and protect lens fiber cells from oxidative stress. J Cell Sci 131. https://doi.org/10.1242/jcs.212506
Okuda H, Nishida K, Higashi Y, Nagasawa K (2013) NAD(+) influx through connexin hemichannels prevents poly(ADP-ribose) polymerase-mediated astrocyte death. Life Sci 92:808–814
Figueroa XF, Lillo MA, Gaete PS, Riquelme MA, Sáez JC (2013) Diffusion of nitric oxide across cell membranes of the vascular wall requires specific connexin-based channels. Neuropharmacology 75:471–478
Hill CE, Hickey H, Sandow SL (2000) Role of gap junctions in acetylcholine-induced vasodilation of proximal and distal arteries of the rat mesentery. J Auton Nerv Syst 81:122–127
Spees JL, Olson SD, Whitney MJ, Prockop DJ (2006) Mitochondrial transfer between cells can rescue aerobic respiration. Proc Natl Acad Sci U S A 103:1283–1288
Ren D, Zheng P, Zou S, Gong Y, Wang Y, Duan J, Deng J, Chen H, Feng J, Zhong C, Chen W (2021) GJA1-20K enhances mitochondria transfer from astrocytes to neurons via Cx43-TnTs after traumatic brain injury. Cell Mol Neurobiol. https://doi.org/10.1007/s10571-021-01070-x
Golan K, Singh AK, Kollet O, Bertagna M, Althoff MJ, Khatib-Massalha E, Petrovich-Kopitman E, Wellendorf AM, Massalha H, Levin-Zaidman S, Dadosh T, Bohan B, V. Gawali M, Dasgupta B, Lapidot T, Cancelas JA (2020) Bone marrow regeneration requires mitochondrial transfer from donor Cx43-expressing hematopoietic progenitors to stroma. Blood 136:2607–2619
Zhang DC, Chen R, Cai YH, Wang JJ, Yin C, Zou K (2020) Hyperactive reactive oxygen species impair function of porcine Sertoli cells via suppression of surface protein ITGB1 and connexin-43. Zool Res 41:203–207
Li H, Brodsky S, Kumari S, Valiunas V, Brink P, Kaide J, Nasjletti A, Goligorsky MS (2002) Paradoxical overexpression and translocation of connexin43 in homocysteine-treated endothelial cells. Am J Physiol Heart Circ Physiol 282:H2124–H2133
Boengler K, Dodoni G, Rodriguez-Sinovas A, Cabestrero A, Ruiz-Meana M, Gres P, Konietzka I, Lopez-Iglesias C, Garcia-Dorado D, Di Lisa F et al (2005) Connexin 43 in cardiomyocyte mitochondria and its increase by ischemic preconditioning. Cardiovasc Res 67:234–244
Kozoriz MG, Church J, Ozog MA, Naus CC, Krebs C (2010) Temporary sequestration of potassium by mitochondria in astrocytes. J Biol Chem 285:31107–31119
Miro-Casas E, Ruiz-Meana M, Agullo E, Stahlhofen S, Rodríguez-Sinovas A, Cabestrero A, Jorge I, Torre I, Vazquez J, Boengler K et al (2009) Connexin43 in cardiomyocyte mitochondria contributes to mitochondrial potassium uptake. Cardiovasc Res 83:747–756
Pecoraro M, Pala B, Di Marcantonio MC, Muraro R, Marzocco S, Pinto A, Mincione G, Popolo A (2020) Doxorubicin-induced oxidative and nitrosative stress: mitochondrial connexin 43 is at the crossroads. Int J Mol Med 46:1197–1209
Pecoraro M, Pinto A, Popolo A (2020) Trastuzumab-induced cardiotoxicity and role of mitochondrial connexin43 in the adaptive response. Toxicol in Vitro 67:104926
Availability of data and material
Not applicable
Code availability
Not applicable
Funding
This work was financially supported by the National Natural Science Foundation of China (Nos. 81974502, 81671293, and 81302750), Natural Science Foundation of Hunan Province (No. 2020JJ3061), and Hunan Provincial Department of Education Innovation Platform Open Fund Project (No. 17K100).
Author information
Authors and Affiliations
Contributions
XYM designed and revised the manuscript. KZ, QWG, and XYZ wrote the manuscript. QXX, XXY, and HHZ contributed to literature arrangement and the generation of figures and tables. All authors read and approved the final version of manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, K., Guan, QW., Zhou, XY. et al. The mutual interplay of redox signaling and connexins. J Mol Med 99, 933–941 (2021). https://doi.org/10.1007/s00109-021-02084-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-021-02084-0