Abstract
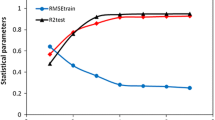
Support vector machine (SVM) was used to develop a nonlinear quantitative structure–activity relationship (QSAR) model for the prediction of the activities of the adenosine A2A receptor antagonists. Six molecular descriptors selected by the heuristic method (HM) in CODESSA were used as inputs for SVM. The results obtained by SVM were compared with those obtained by HM. The mean squared errors (MSEs) for the training set given by HM and SVM are 0.08 and 0.05, respectively, which shows the performance of SVM model is better than that of the HM model.



Similar content being viewed by others
References
Balaban AT (1982) Highly discriminating distance-based topological index. Chem Phys Lett 89:399–404
Bara-Jimenez W, Sherzai A, Dimitrova T, Favit A, Bibbiani F, Gillespie M, Morris MJ, Mouradian MM, Chase TN (2003) Adenosine A2A receptor antagonist treatment of Parkinson’s disease. Neurology 61:293–296
Basak SC, Balaban AT, Grunwald GD, Gute BD (2000) Topological indices: their nature and mutual relatedness. J Chem Inf Comput Sci 40:891–898
Burges CJC (1998) A tutorial on support vector machines for pattern recognition. Data Min Knowl Disc 2:1–47
Christianini N, Shawe-Taylor J (2000) An introduction to support vector machines and other kernel-based learning methods. Cambridge University Press, New York
Consonni V, Todeschini R, Pavan M, Gramatica P (2002) Structure/response correlations and similarity/diversity analysis by GETAWAY descriptors. 2. Application of the novel 3D molecular descriptors to QSAR/QSPR studies. J Chem Inf Comput Sci 42:693–705
Cortes C, Vapnik V (1995) Support-vector networks. Mach Learn 20:273–279
Cramer RD, Patterson DE, Bunce JD (1988) Comparative molecular field analysis (CoMFA). 1. Effect of shape on binding of steroids to carrier proteins. J Am Chem Soc 110:5959–5967
Crippen GM (1979) Distance geometry approach to rationalizing binding data. J Med Chem 22:988–997
Feigin A (2003) Nondopaminergic symptomatic therapies for Parkinson’s disease: turn on or turn off? Neurology 61:286–287
Fredholm BB, Abbracchio MP, Burnstock G, Daly JW, Harden TK, Jacobson KA, Leff P, Williams M (1994) Nomenclature and classification of purinoceptors. Pharmacol Rev 46:143–156
Gillespie RJ, Cliffe IA, Dawson CE, Dourish CT, Gaur S, Jordan AM, Knight AR, Lerpiniere J, Misra A, Pratt RM, Roffey J, Stratton GC, Upton R, Weiss SM, Williamson DS (2008) Antagonists of the human adenosine A2A receptor. Part 3: design and synthesis of pyrazolo[3,4-d]pyrimidines, pyrrolo[2,3-d]pyrimidines and 6-arylpurines. Bioorg Med Chem Lett 18:2924–2929
Gillespie RJ, Bamford SJ, Clay A, Gaur S, Haymes T, Jackson PS, Jordan AM, Klenke B, Leonardi S, Liu J, Mansell HL, Ng S, Saadi M, Simmonite H, Stratton GC, Todd RS, Williamson DS, Yule IA (2009) Antagonists of the human A2A receptor. Part 6: further optimization of pyrimidine-4-carboxamides. Bioorg Med Chem 17:6590–6605
Golbraikh A, Tropsha A (2002) Beware of q2!. J Mol Graph Model 20:269–276
Goodarzi M, Freitas MP, Jensen R (2009) Ant colony optimization as a feature selection method in the QSAR modeling of anti-HIV-1 activities of 3-(3,5-dimethylbenzyl)uracil derivatives using MLR, PLS and SVM regressions. Chemom Intell Lab Syst 98:123–129
Hansch C, Fujita T (1963) p-σ-π analysis. A method for the correlation of biological activity and chemical structure. J Am Chem Soc 86:1616–1626
Jackson JE (1991) A user’s guide to principal components. Wiley, New York
Jankovic J (2008) Parkinson’s disease: clinical features and diagnosis. J Neurol Neurosurg Psychiatry 79:368–376
Jenner P (2003) A2A antagonists as novel non-dopaminergic therapy for motor dysfunction in PD. Neurology 61:S32–S38
Jover J, Bosque R, Sales J (2004) Determination of Abraham solute parameters from molecular structure. J Chem Inf Comput Sci 44:1098–1106
Katritch V, Jaakola VP, Lane JP, Lin J, IJzerman AP, Yeager M, Kufareva I, Stevens RC, Abagyan R (2010) Structure-based discovery of novel chemotypes for adenosine A2A receptor antagonists. J Med Chem 53:1799–1809
Katritzky AR, Lobanov VS, Karelson M (1994) CODESSA: reference manual. University of Florida press, Gainesville
Katritzky AR, Lobanov VS, Karelson M (1995) QSPR: the correlation and quantitative prediction of chemical and physical properties from structure. Chem Soc Rev 24:279–287
Klebe G, Abraham U, Mietzner T (1994) Molecular similarity indices in a comparative analysis (CoMSIA) of drug molecules to correlate and predict their biological activity. J Med Chem 37:4130–4146
Kraim K, Khatmi D, Saihi Y, Ferkous F, Brahimi M (2009) Quantitative structure activity relationship for the computational prediction of α-glucosidase inhibitory. Chemom Intell Lab Syst 97:118–126
Leis J, Karelson M (2001) A QSPR model for the prediction of the gas-phase free energies of activation of rotation around the N–C(O) bond. Comput Chem 25:171–176
Li JZ, Liu HX, Yao XJ, Liu MC, Hu ZD, Fan BT (2007) Quantitative structure–activity relationship study of acyl ureas as inhibitors of human liver glycogen phosphorylase using least squares support vector machines. Chemom Intell Lab Syst 87:139–146
Luan F, Ma WP, Zhang XY, Zhang HX, Liu MC, Fan BT (2006) QSAR study of polychlorinated dibenzodioxins, dibenzofurans, and biphenyls using the heuristic method and support vector machine. QSAR Comb Sci 25:46–55
Ma WP, Luan F, Zhao CY, Zhang XY, Liu MC, Hu ZD, Fan BT (2006) QSAR prediction of the penetration of drugs across a polydimethylsiloxane membrane. QSAR Comb Sci 25:895–904
Melagraki G, Afantitis A, Sarimveis H, Koutentis PA, Markopoulos J, Igglessi-Markopoulou O (2007) Optimization of biaryl piperidine and 4-amino-2-biarylurea MCH1 receptor antagonists using QSAR modeling, classification techniques and virtual screening. J Comput Aided Mol Des 21:251–267
Mishra CB, Barodia SK, Prakash A, Kumar JBS, Luthra PM (2010) Novel 8-(furan-2-yl)-3-substituted thiazolo [5,4-e][1,2,4] triazolo[1,5-c] pyrimidine-2(3H)-thione derivatives as potential adenosine A2A receptor antagonists. Bioorg Med Chem 18:2491–2500
Nowaczyk A, Kulig K, Malawska B (2009) 1-(3-(4-Arylpiperazin-1-yl)-propyl)-pyrrolidin-2-one derivatives as 1-adrenoceptor antagonists: a QSAR study. QSAR Comb Sci 28:979–988
Pinna A (2009) Novel investigational adenosine A2A receptor antagonists for Parkinson’s disease. Expert Opin Investig Drugs 18:1619–1631
Qin S, Liu H, Wang J, Yao X, Liu M, Hu Z, Fan B (2007) Quantitative structure–activity relationship study on a series of novel ligands binding to central benzodiazepine receptor by using the combination of heuristic method and support vector machines. QSAR Comb Sci 26:443–451
Reunanen J (2003) Overfitting in making comparisons between variable selection methods. J Mach Learn Res 3:1371–1382
Rohrbaugh RH, Jurs PC (1987) Descriptions of molecular shape applied in studies of structure–activity and structure–property relationships. Anal Chim Acta 199:99–109
Roy PP, Roy K (2008) On some aspects of variable selection for partial least squares regression models. QSAR Comb Sci 27:302–313
Schefzick S, Kibbey C, Bradley MP (2004) Prediction of HPLC conditions using QSPR techniques: an effective tool to improve combinatorial library design. J Comb Chem 6:916–927
Si HZ, Wang T, Zhang KJ, Hu ZD, Fan BT (2006) QSAR study of 1,4-dihydropyridine calcium channel antagonists based on gene expression programming. Bioorg Med Chem 14:4834–4841
Sitkovsky M, Lukashev D, Deaglio S, Dwyer K, Robson SC, Ohta A (2008) Adenosine A2A receptor antagonists: blockade of adenosinergic effects and T regulatory cells. Br J Pharmacol 153:S457–S464
Stewart JPP (1989) MOPAC 6.0, Quantum Chemistry Program Exchange, QCPE, No. 455. Indiana University press, Bloomington
Strouf O (1986) Chemical pattern recognition. Wiley, New York
Takahashi RN, Pamplona FA, Prediger RD (2008) Adenosine receptor antagonists for cognitive dysfunction: a review of animal studies. Front Biosci 13:2614–2632
Vapnik V (1982) Estimation of dependencies based on empirical data. Springer, Berlin
Vapnik VN (1995) The nature of statistical learning theory. Springer, New York
Volpini R, Ben DD, Lambertucci C, Marucci G, Mishra RC, Ramadori AT, Klotz K-N, Trincavelli ML, Martini C, Cristalli G (2009) Adenosine A2A receptor antagonists: new 8-substituted 9-ethyladenines as tools for in vivo rat models ls of Parkinson’s Disease. ChemMedChem 4:1010–1019
Xu K, Bastia E, Schwarzschild M (2005) Therapeutic potential of adenosine A(2A) receptor antagonists in Parkinson’s disease. Pharmacol Ther 105:267–310
Zorko M, Majerle A, Sarlah D, Keber MM, Mohar B, Jerala R (2005) Combination of antimicrobial and endotoxin-neutralizing activities of novel oleoylamines. Antimicrob Agents Chemother 49:2307–2313
Acknowledgments
This study is supported by National Natural Science Foundation of P.R.China (No. 90912003, 60773108, and 90812001). The authors also thank the R Development Core Team for affording the free R2.4.1 software.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lu, P., Wei, X., Zhang, R. et al. Prediction of the binding affinities of adenosine A2A receptor antagonists based on the heuristic method and support vector machine. Med Chem Res 20, 1220–1228 (2011). https://doi.org/10.1007/s00044-010-9431-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-010-9431-1