Summary
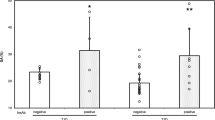
The aim of this study was to investigate the modifying effect of diabetes on the in vitro serum binding of gliclazide at therapeutic concentrations, and to try to determine the role of albumin glycation. Sera were obtained from 20 patients with diabetes and 20 matched nondiabetic controls. Gliclazide was added in vitro at a therapeutic concentration (5 mg/L). After equilibrium dialysis, gliclazide was assayed by high-performance liquid chromatography, while glycated albumin was assayed by affinity chromatography and laser-nephelemetry. The mean binding of gliclazide was lower in diabetics (82.6 ± 4.1%) than in controls (88.8 ± 4.8%) [p<O.OOl]. No correlation was observed between gliclazide binding and the percentage of albumin glycation. Sera were also obtained from the same diabetic patients 3 months later. There was no correlation between variation of glycated albumin and variation of gliclazide binding over 3 months. The protein binding of gliclazide was lower in diabetics than in matched controls. Furthermore, the decreased binding was not dependent upon the level of albumin glycation, and did not disappear when chronic hyperglycaernia was corrected.
Similar content being viewed by others
References
Koenig RJ, Cerami A. Non enzymatic glycosylation. Ann Rep Med Chem 1979; 14: 261–7
Ferner RE, Chaplin S. The relationship between the pharmacokinetics and pharmacodynamic effects of oral hypoglycaemic drugs. Clin Pharm 1987; 12: 379–401
Judis J. Binding of Sulfonylureas to serum proteins. J Pharm Sci. 1972; 1: 89–93
Tsuchiya S, Sakurai T, Sekiguchi SI. Nonenzymatic glucosylation of human serum albumin and its influence on binding capacity of sulfonylureas. Biochem Pharm 1984; 19: 2967–71
Kradjan WA, Kobayashi KA, Bauer LA, et al. Glipizide pharmacokinetics: effects of age, diabetes and multiple dosing. J Clin Pharmacol 1989; 29: 1121–7
National Diabetes Data Group. Diabetes 1979; 28: 1039–57
Charles BG, Ravenscroft PJ. Measurement of gliclazide in plasma by radial compression reversed-phase liquid chromatography. Clin Chem 1984; 30: 1789–91
Kimura M, Kobayashi K, Hata M, et al. Reverse-phase high-performance liquid chromatographic determination of gliclazide in human plasma. Chem Pharm Bull 1980; 28: 344–6
Kurz H, Trunk H, Weitz B. Evaluation of methods to determine protein-binding of drugs: Equilibrium dialysis, ultrafiltration, ultracentrifugation, gel filtration. Drug Res 1977; 7: 1373–680
John WG, Jones AE. Affinity chromatography: a precise method for glycosylated albumin estimation. Ann Clin Biochem 1985; 22: 79–83
Doucet J, Fresel J, Hue G, et al. Protein binding of digitoxin, valproate and phenytoin in sera from diabetics. Eur J Clin Pharm 1993; 45: 577–9
Garlick RL, Mazer JS. The principle site of nonenzymatic glycosylation of human serum albumin in vivo. J Biol Chem 1983; 258: 6142–6
Shaklai N, Garlick RL, Bunn HF. Nonenzymatic glycosylation of human serum albumin alters its conformation and function. J Biol Chem 1984; 259: 3812–7
Gatti G, Crema F, Attardo-Parinello G, et al. Serum binding of phenytoin and valproic acid in insulin-dependent diabetes mellitus. Therap Drug Monit 1987; 9:389–91
MacNamara PJ, Blouin RA, Brazzell RK. The protein binding of phenytoin, propranolol, diazepam and AL 01576 (an aldose-reductase inhibitor) in human and rat diabetic serum. Pharm Res 1988; 5: 261–5
Acknowledgements
We are grateful to Servier Laboratory (Neuilly, France) for donating gliclazide for this study.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Doucet, J., Fresel, J., Moore, N. et al. In Vitro Serum Binding of Gliclazide in Patients with Type I Diabetes Mellitus. Drug Invest 8, 219–224 (1994). https://doi.org/10.1007/BF03258481
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03258481