Summary
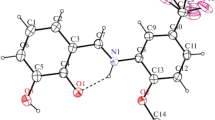
It has been established from H1 NMR spectra in CDCl3 with additions of Eu(FOD)3 as paramagnetic shift reagent that tavicone must be ascribed the structure of 7-(1,1,3a,5-tetramethyl-2-oxo-Δ 55 -hexahydroinden-4-ylmethoxy) coumarin. The stereochemistry of this compound has been determined.
Similar content being viewed by others
Literature cited
V. Yu. Bagirov, N. P. Kir'yalov, and V. I. Sheichenko, Khim. Prirodn. Soedin., 591 (1969).
V. I. Sheichenko, G. M. Nikolaev, N. I. Dubrovina, V. F. Bystrov, and G. P. Kugatova-Shemyakina, Izv. Akad. Nauk SSSR, Ser. Khim., No. 11 (1971).
E. W. Garbish, J. Amer. Chem. Soc.,86, 5561 (1964).
G. P. Newsoroff and S. Sternhell, Tetrahedron Lett.,58, 6117 (1968).
I. Ya. Slonim and A. Kh. Bulai, Usp. Khim.,17, No. 11, 1976 (1973).
A. J. Rafalski, Y. Barciszewski, and M. Wilwcorowski, Tetrahedron Lett., 2829 (1971).
Additional information
Institute of Botany, Academy of Sciences of the Azerbaidzhan SSR, Baku. All-Union Scientific-Research Institute of Medicinal Plants, Moscow. Translated from Khimiya Prirodnykh Soedinenii, No. 4, pp. 452–455, July–August, 1976.
Rights and permissions
About this article
Cite this article
Bagirov, V.Y., Sheichenko, V.I. The structure and stereochemistry of tavicone. Chem Nat Compd 12, 399–401 (1976). https://doi.org/10.1007/BF00564795
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00564795