Abstract
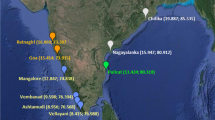
Restriction enzyme analysis of mitochondrial DNA (mtDNA) was used to test for genetic homogeneity of orange roughy (Hoplostethus atlanticus) in the southern hemisphere. Two hundred and eighty-six orange roughy specimens were collected from seven general localities: the Great Australian Bight; South Australia (off southeastern Kangaroo Island); the west coast of Tasmania; the east coast of Tasmania; New South Wales; New Zealand and South Africa. Mitochondrial DNA was extracted from develo** ovary tissue and analysed with 10 six-base enzymes and 3 four-base enzymes. Both forms of analysis revealed a low level of genetic diversity in this species. The six-base enzyme study found no evidence of reproductively isolated populations of orange roughy in southeastern Australian waters. However, an analysis of 107 fish with 3 four-base enzymes identified at least partial genetic separation of the New South Wales (NSW) sample of orange roughy from South Australian (off southeastern Kangaroo Island) and Tasmanian samples. This finding supports biological evidence for the presence of a distinct subpopulation of orange roughy in NSW waters. The four-base study also provided evidence of the presence of genetically distinct samples of orange roughy occurring in the same localities off southeastern Kangaroo Island from consecutive years. Additional sampling and the use of a greater number of four-base enzymes may be needed to determine if any genetic structuring exists among orange roughy south of New South Wales.
Similar content being viewed by others
Literature cited
Avise, J. C., Arnold, J., Ball, R. M., Bermingham, E., Lamb, T., Neigel, J. E., Reeb, C. A., Saunders, N. C. (1987a). Intraspecific phylogeography: The mitochondrial DNA bridge between population genetics and systematics A. Rev. Ecol. Syst. 18: 489–552
Avise, J. C., Reeb, C. A., Saunders, N. C. (1987b). Geographic population structure and species differences in mitochondrial DNA of mouthbrooding marine catfishes (Ariidae) and demersal spawning toadfishes (Batrachoididae). Evolution 41:991–1002
Bell, J. D. (1989). Reproductive cycle and fecundity of orange roughy, Hoplostethus atlanticus, in southeastern Australia. Report of the Bureau of Rural Resources Department of Primary Industry and Energy, Canberra
Bell, J. D., Lyle, J. M., Bulman, C. M., Graham, K. J., Newton, G. M., Smith, D. C. (1992). Spatial variation in reproduction, and occurrence of non-reproductive adults, in orange roughy, Hoplostethus atlanticus Collett (Trachichthyidae), from south-eastern Australia. J. Fish Biol. 40: 107–122
Birky, C. W., Fuerst, P., Maruyama, T. (1989). Organelle gene diversity under migration, mutation, and drift: equilibrium expectations, approach to equilibrium, effects of heteroplasmic cells, and comparison to nuclear genes. Genetics, Austin, Tex. 121: 613–627
Black, M., Dixon, P. I. (1989). Population structure of orange roughy (Hoplostethus atlanticus) in Australian waters. Centre for Marine Science, University of New South Wales, Sydney (unpublished report)
Brasher, D. J., Ovenden, J. R., Booth, J. D., White, R. W. G. (1992). Genetic subdivision of Australian and New Zealand populations of Jasus verreauxi (Decapoda, Palinuridae) — preliminary evidence from the mitochondrial genome. N. Z. Jl mar. Freshwat. Res. 26: 53–58
Brown, W. M., George, M., Wilson, A. C. (1979). Rapid evolution of animal mitochondrial DNA. Proc. natn. Acad. Sci. U.S.A. 76: 1967–1971
Bucklin, A., Rienecker, M. M., Mooers, N. K. (1989). Genetic tracers of zooplankton transport in coastal filaments off northern California. J. geophys. Res. 94: 8277–8288
Bulman, C. M., Wayte, S. E., Elliott, N. G. (1993). Orange roughy survey, 1988 and 1989; abundance indices. Rep. mar. Lab. C.S.I.R.O. 215: (in press)
Chapman, R. W., Powers, D. A. (1984). A method for the rapid isolation of mitochondrial DNA from fishes. Tech. Rep. Univ. Md Sea Grant Progm. UM-SGTS 84-05: 1–11
Dahle, G. (1991). Cod, Gadus morhua L., populations identified by mitochondrial DNA. J. Fish Biol. 38: 295–303
Edmonds, J. S., Caputi, N., Morita, M. (1991). Stock discrimination by trace element analysis of otoliths of orange roughy (Hoplostethus atlanticus) a deep-water marine teleost. Aust. J. mar. Freshwat. Res. 42: 383–389
Elliott, N. G., Ward, R. D. (1993). Enzyme variation in orange roughy (Hoplostethus atlanticus) samples from southern Australian and New Zealand waters. Aust. J. mar. Freshwat. Res. (in press)
Fenton, G. E., Short, S. A., Ritz, D. A. (1991). Age determination of orange roughy, Hoplostethus atlanticus (Pisces: Trachichthyidae) using 210Pb:226Ra disequilibria. Mar. Biol. 109: 187–202
Hamilton, L. J. (1990). Temperature inversions at intermediate depths in the Antarctic intermediate waters of the south-western Pacific. Aust. J. mar. Freshwat. Res. 41: 325–351
Heath, R. A. (1985) A review of the physical oceanography of the seas around New Zealand-1982. N. Z. Jl mar. Freshwat. Res. 1: 79–124
Kessler, L. G., Avise, J. C. (1984). Systematic relationships among waterfowl (Anatidae) inferred from restriction endonuclease analysis of mitochondrial DNA. Syst. Zool. 33: 370–380
Lester, R. J. G. (1990). Reappraisal of the use of parasites for fish stock identification. Aust. J. mar. Freshwat. Res. 41: 855–864
Lester, R. J. G., Sewell, K. B., Barnes, A., Evans, K. (1988). Stock discrimination of orange roughy Hoplostethus atlanticus, by parasite analysis. Mar. Biol. 99: 137–143
Lyle, J., Kitchener, J., Riley, S. (1989). Orange roughy bonanza off Tasmania. Aust. Fish. 48: 20–24
Lyle, J., Kitchener, J., Riley, S. (1990). St. Helens roughy site — 1990 season. Aust. Fish. 49: 27–29
Lynch, M., Crease, T. J. (1990). The analysis of population survey data on DNA sequence variation. Molec. Biol. Evolut. 7: 377–394
Mace, P. M., Fenaughty, J. M., Coburn, R. P., Doonan, I. J. (1990). Growth and productivity of orange roughy (Hoplostethus atlanticus) on the North Chatham Rise. N. Z. Jl mar. Freshwat. Res. 24: 105–119
Nei, M., **, L. (1989). Variance of the average numbers of nucleotide substitutions within and between populations. Molec. Biol. Evolut. 6: 290–300
Nei, M., Li, W. H. (1979). Mathematical model for studying genetic variation in terms of restriction endonucleases. Proc. natn. Acad. Sci. U.S.A. 76: 5269–5273
Nei, M., Tajima, F. (1983) Maximum likelihood estimation of the number of nucleotide substitutions from restriction sites data. Genetics, Austin, Tex. 105: 207–217
Newton, G. (1988). Orange roughy research in the Great Australian Bight. Aust. Fish. 47: 7–8
Newton, G., Burnell, S. (1989). ‘Roughy rush’ in the Great Australian Bight. Aust. Fish. 48: 10–13
Newton, G., Burnell, S., Riley, S. (1990). Spawning roughy in the G.A.B. — a new find. Aust. Fish. 49: 24–26
Ovenden, J. R. (1990). Mitochondrial DNA and marine stock assessment: a review. Aust. J. mar. Freshwat. Res. 41: 835–853
Ovenden, J. R., Brasher, D. J., White, R. W. G. (1992a). Mitochondrial DNA analyses of the red rock lobster Jasus edwardsii supports an apparent absence of population subdivision throughout Australasia. Mar. Biol 113: 319–326
Ovenden, J. R., Bywater, R., White, R. W. G. (1992b). A program for the estimation of restriction endonuclease site position from restriction fragment size and number — an aid for mtDNA analysis. J. Hered. 83: 240–241
Ovenden, J. R., Smolenski, A. J., White, R. W. G. (1989). Mitochondrial DNA restriction site variation in Tasmanian populations of orange roughy (Hoplostethus atlanticus) a deep water marine teleost. Aust. J. mar. Freshwat. Res. 40: 1–9
Palumbi, S. R., Wilson, A. C. (1990). Mitochondrial DNA diversity in the sea urchins Strongylocentrotus purpuratus and S. droebachiensis. Evolution 44: 403–415
Pankhurst, N. W., Conroy, A. M. (1987). Size-fecundity relationships in the orange roughy Hoplostethus atlanticus. N. Z. Jl mar. Freshwat. Res. 21: 295–300
Pankhurst, N. W., McMillan, P. J., Tracey, D. M. (1987). Seasonal reproductive cycles in three commercially exploited fishes from the slope waters off New Zealand. J. Fish Biol. 30: 193–211
Robertson, D. A., Grimes, P. J., McMillan, P. J. (1984). Orange roughy on Chatham Rise; results of a trawl survey August–September 1982. Occ. Publs Fish. Res. Div. N. Z. Minist. Agric. Fish. 46: 1–27
Roff, D. A., Bentzen, P. (1989). The statistical analysis of mitochondrial DNA polymorphisms: x 2 and the problem of small samples. Molec. Biol. Evolut. 6: 539–545
Sambrook, J., Fritsch, E. F., Maniatis, T. (1989). Molecular cloning: a laboratory manual. 2nd ed. Cold Spring Harbor Press, New York
Smith, P. J. (1986). Genetic similarity between samples of the orange roughy Hoplostethus atlanticus from the Tasman Sea, Southwest Pacific Ocean and North-east Atlantic Ocean. Mar. Biol. 91: 173–180
Smith, P. J., Francis, R. I. C. C., McVeagh, M. (1991). Loss of genetic diversity due to fishing pressure. Fish. Res. 10: 309–316
Takahata, N., Palumbi, S. R. (1985). Extranuclear differentiation and gene flow in the finite island model. Genetics, Austin, Tex. 109: 441–457
White, B. A., Shaklee, J. B. (1991). Need for replicated electrophoretic analyses in multiagency genetic stock identification (GSI) programs: examples from a pink salmon (Oncorhynchus gorbuscha) GSI fisheries study. Can J. Fish. aquat. Sciences 48: 1396–1407
Wilson, A. C., Cann, R. L., Carr, S. M., George, M., Gyllensten, U. B., Helm-Bychowski, K. M., Higuchi, R. G., Palumbi, S. R., Prager, E. M., Sage, R. D., Stoneking, M. (1985). Mitochondrial DNA and two perspectives on evolutionary genetics. Biol. J. Linn. Soc. 26: 375–400
Wirgin, I. I., Proenca, R., Grossfield, J. (1988). Mitochondrial DNA diversity among populations of striped bass in the Southeastern United States. Can. J. Zool. 67: 891–907
Author information
Authors and Affiliations
Additional information
Communicated by G. F. Humphrey, Sydney
Rights and permissions
About this article
Cite this article
Smolenski, A.J., Ovenden, J.R. & White, R.W.G. Evidence of stock separation in southern hemisphere organge roughy (Hoplostethus atlanticus, Trachichthyidae) from restriction-enzyme analysis of mitochondrial DNA. Marine Biology 116, 219–230 (1993). https://doi.org/10.1007/BF00350011
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00350011