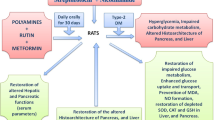
Abstract
Objectives
This study investigated the possible changes in glucose homeostasis, homeostatic model assessment, glucose transporter-4, and hepato-pancreatic histology in streptozotocin (STZ)-induced diabetic rats following intermittent fasting, exercise, starvation, and honey treatment.
Methods
In the non-diabetic phase, rats were administered an oral regimen of distilled water (10 mL/kg), IF, starvation, and honey (1 g/kg) for 4 weeks while in the diabetic phase, after STZ injections, interventions with IF, exercise, starvation, and honey treatment were administered for 4 weeks. This was followed by biochemical studies.
Results
In addition to increasing insulin resistance as shown by a high HOMA-IR score, STZ-induced diabetes decreased glucose transporter-4 (GLUT-4), increased HOMA-IR, HOMA-insulin sensitivity, and HOMA-beta cell function in the pancreas. Notably, IF and exercise greatly ameliorated these changes in diabetic rats unlike starvation and honey regimen. Streptozotocin and starvation substantially and partially destroyed hepato-pancreatic cells, according to histopathological studies, although these were moderately ameliorated by IF and exercise therapies, and less by honey treatment.
Conclusion
Our findings suggest that the IF and exercise and honey treatment to a smaller extent attenuate type 2 diabetes via enhancement of insulin sensitivity, reduction of sugar level, modulate GLUT-4, and hepatic-pancreatic-cell protection.









Similar content being viewed by others
Data availability
The corresponding author can provide all of the data used in this article upon request.
Code availability
Not applicable.
Abbreviations
- STZ:
-
Streptozotocin
- IF:
-
Intermittent fasting
- IS:
-
Insulin sensitivity
- IR:
-
Insulin resistance
- HOMA:
-
Homeostatic model assessment
- HOMA-IR:
-
Homeostatic model assessment of insulin resistance
- HOMA-IS:
-
Homeostatic model assessment of insulin sensitivity
- HOMA-B:
-
Homeostatic model assessment of beta cell
References
Srinivasan K, Ramarao P. Animal model in type 2 diabetes research: an overview. Indian J Med Res. 2007;125(3):451.
Chinnaboina GK, Sudhakar A, Verma R, Sharma P, Shrivastava B. A review on diabetes mellitus: current update on management and treatment. Asian Pac J Health Sci. 2018;53(3):67–82.
Bak E, Marcisz C, Krzeminska S, Dobrzyn-Matusiak D, Foltyn A, Drosdzol-Cop A. Does type 1 diabetes modify sexuality and mood of women and men? Int J Environ Res Public Health. 2018;15(5):958. https://doi.org/10.3390/ijerph15050958.
Talukder A, Hossain MZ. Prevalence of diabetes mellitus and its associated factors in Bangladesh: application of two-level logistic regression model. Sci Rep. 2020;10(1):10237. https://doi.org/10.1038/s41598-020-66084-9.
Gbadamosi MA, Tlou B. Prevalence of abnormal glucose metabolism among adults attending an outpatient department at a tertiary referral hospital in Swaziland: a cross-sectional study. BMC Public Health. 2020;20(1):392. https://doi.org/10.1186/s12889-020-08489-9.
Rachdaoui N. Insulin: the friend and the foe in the development of type 2 diabetes mellitus. Int J Mol Sci. 2020;21(5):1770. https://doi.org/10.3390/ijms21051770.
Wysham C, Shubrook J. Beta-cell failure in type 2 diabetes: mechanisms, markers, and clinical implications. Postgrad Med. 2020;132(8):676–86. https://doi.org/10.1080/00325481.2020.1771047.
Adeniyi O, Washington L, Glenn CJ, Franklin SG, Scott A, Aung M, Niranjan SJ, Jolly PE. The use of complementary and alternative medicine among hypertensive and type 2 diabetic patients in Western Jamaica: a mixed methods study. PLOS ONE. 2021;16(2):e0245163. https://doi.org/10.1371/journal.pone.0245163.
Albosta M, Bakke J. Intermittent fasting: is there a role in the treatment of diabetes? A review of the literature and guide for primary care physicians. Clin Diabetes Endocrinol. 2021;7(1):3. https://doi.org/10.1186/s40842-020-00116-1.
Grajower MM, Horne BD. Clinical management of intermittent fasting in patients with diabetes mellitus. Nutrients. 2019;11(4):873. https://doi.org/10.3390/nu11040873\.
Taylor R. Calorie restriction for long-term remission of type 2 diabetes. Clin Med (Lond). 2019;19(1):37–42. https://doi.org/10.7861/clinmedicine.19-1-37.
Henson J, Yates T, Edwardson CL, Khunti K, Talbot D, Gray LJ, Davies MJ. Sedentary time and markers of chronic low-grade inflammation in a high risk population. PLoS ONE. 2013;8(10):78350–007835.
Harvie M, Howell A. Potential benefits and harms of intermittent energy restriction and intermittent fasting amongst obese, overweight and normal weight subjects—a narrative review of human and animal evidence. Behav Sci. 2017;7(1):4.
Anton SD, Moehl K, Donahoo WT, Marosi K, Lee SA, Mainous III AG., ... and Mattson MP. Flip** the metabolic switch: understanding and applying the health benefits of fasting. Obesity. 2018,26(2), 254–268.
Stanford KI, Goodyear LJ. Exercise and type 2 diabetes: molecular mechanisms regulating glucose uptake in skeletal muscle. Adv Physiol Educ. 2014;38(4):308–14. https://doi.org/10.1152/advan.00080.2014.
Teixeira-Lemos E, Nunes S, Teixeira F, Reis F. Regular physical exercise training assists in preventing type 2 diabetes development: focus on its antioxidant and anti-inflammatory properties. Cardiovasc Diabetol. 2011;10(1):1–15.
Zheng J, Cheng J, Zheng S, Zhang L, Guo X, Zhang J, **ao X. Physical exercise and its protective effects on diabetic cardiomyopathy: what is the evidence? Front Endocrinol. 2018;9:729.
Agbonifo-Chijiokwu E, Nwangwa KE, Oyovwi MO, et al. Underlying biochemical effects of intermittent fasting, exercise and honey on streptozotocin-induced liver damage in rats. J Diabetes Metab Disord. 2023. https://doi.org/10.1007/s40200-022-01173-2.
Lemieux I. Reversing type 2 diabetes: the time for lifestyle medicine has come! Nutrients. 2020;12(7):1974. https://doi.org/10.3390/nu12071974.
Akanmu MA, Olowookere TA, Atunwa SA, Ibrahim BO, Lamidi OF, Adams PA, Ajimuda BO, Adeyemo LE. Neuropharmacological effects of Nigerian honey in mice. Afr J Tradit Complement Altern Med. 2011;8(3):230–49
Al Aamri ZM, Ali BH. Does honey have any salutary effect against streptozotocin - induced diabetes in rats? J Diab Metab Disord. 2017;16:1–6. https://doi.org/10.1186/s40200-016-0278-y.
Almasaudi SB, El-Shitany NA, Abbas AT, Abdel-dayem UA, Ali SS, Al Jaouni SK, Harakeh S. Antioxidant, anti-inflammatory, and antiulcer potential of manuka honey against gastric ulcer in rats. Oxidative Med Cell Longev. 2016;2016:1–10.
Moke EG, Omogbai EKI, Osagie-Eweka SDE, Uchendu AP, Omogbiya AI, Ben-Azu B, Eduviere AT, Edje KE, Umukoro EK, Anachuna KK, Asiwe JN, Ahante E, Oghoghovwe IJ. Co-administration of metformin and/or glibenclamide with losartan reverse NG-nitro-l-arginine-methyl ester-streptozotocin-induced hypertensive diabetes and haemodynamic sequelae in rats. Microvasc Res. 2023;147: 104497.
Erejuwa OO, Sulaiman SA, Wahab MSA, Sirajudeen KNS, Salleh MSM, Gurtu S. Glibenclamide or metformin combined with honey improves glycemic control in streptozotocin-induced diabetic rats. Int J Biol Sci. 2011;7(2):244–52. https://doi.org/10.7150/ijbs.7.244.
Szalai Z, Szász A, Nagy I, Puskás LG, Kupai K, Király A, Berkó AM, Pósa A, Strifler G, Baráth Z, Nagy LI, Szabó R, Pávó I, Murlasits Z, Gyöngyösi M, Varga C. Anti-inflammatory effect of recreational exercise in TNBS-induced colitis in rats: role of NOS/HO/MPO system. Oxidative Med Cell Longev. 2014;2014:925981. https://doi.org/10.1155/2014/925981.
Hsing AW, Gao YT, Chua S, Deng J, Stanczyk FZ. Insulin resistance and prostate cancer risk. JNCI J Nat Cancer Inst. 2003;95(1):67–71. https://doi.org/10.1093/jnci/95.1.67.
Park JM, Bong HY, Jeong HI, Kim YK, Kim JY, Kwon O. Postprandial hypoglycemic effect of mulberry leaf in Goto-Kakizaki rats and counterpart control Wistar rats. Nurs Res Pract. 2009;3(4):272. https://doi.org/10.4162/nrp.2009.3.4.272.
Abdulwahab DA, El-Missiry MA, Shabana S, Othman AI, Amer ME. Melatonin protects the heart and pancreas by improving glucose homeostasis, oxidative stress, inflammation and apoptosis in T2DM-induced rats. Heliyon. 2021;7(3):e06474. https://doi.org/10.1016/j.heliyon.2021.e06474.
Kim BH, Joo Y, Kim MS, Choe HK, Tong Q, Kwon O. Effects of intermittent fasting on the circulating levels and circadian rhythms of hormones. Endocrinol Metab (Seoul, Korea). 2021;36(4):745–56. https://doi.org/10.3803/EnM.2021.405.
Chijiokwu EA, Nwangwa EK, Oyovwi MO, Naiho AO, Emojevwe V, Ohwin EP, Ehiwarior PA, Ojugbeli ET, Nwabuoku US, Oghenetega OB, Ogheneyoma OO. Intermittent fasting and exercise therapy abates STZ-induced diabetotoxicity in rats through modulation of adipocytokines hormone, oxidative glucose metabolic, and glycolytic pathway. Physiol Rep. 2022;10(20): e15279.
Oza MJ, Kulkarni YA. Formononetin treatment in type 2 diabetic rats reduces insulin resistance and hyperglycemia. Front Pharmacol. 2018;9:739. https://doi.org/10.3389/fphar.2018.00739.
Mahmoud AM. Hematological alterations in diabetic rats - role of adipocytokines and effect of citrus flavonoids. EXCLI J. 2013;12:647–57.
Mahmoud AM, Ashour MB, Abdel-Moneim A, Ahmed OM. Hesperidin and naringin attenuate hyperglycemia-mediated oxidative stress and proinflammatory cytokine production in high fat fed/streptozotocin-induced type 2 diabetic rats. J Diab Complicat. 2012;26(6):483–90. https://doi.org/10.1016/j.jdiacomp.2012.06.001.Mahmoud,A.M.
Petersen MC, Shulman GI. Mechanisms of insulin action and insulin resistance. Physiol Rev. 2018;98(4):2133–223.
Beck-Nielsen H. Insulin resistens: organ manifestationer of cellulaere mekanismer. Ugeskr Laeger. 2002;164(16):2130–5.
Caliskan H, Akat F, Omercioglu G, Bastug G, Ficicilar H, Bastug M. Aerobic exercise has an anxiolytic effect on streptozotocin-induced diabetic rats. Acta Neurobiol Exp. 2020;80(3):245–55.
Caliskan H, Akat F, Tatar Y, Zaloglu N, Dursun AD, Bastug M, Ficicilar H. Effects of exercise training on anxiety in diabetic rats. Behav Brain Res. 2019;376:112084. https://doi.org/10.1016/j.bbr.2019.112084.
Munhoz AC, Vilas-Boas EA, Panveloski-Costa AC, Leite JSM, Lucena CF, Riva P, Emilio H, Carpinelli AR. Intermittent fasting for twelve weeks leads to increases in fat mass and hyperinsulinemia in young female Wistar rats. Nutrients. 2021;12:1029.
Adesoji F, Oluwakemi A. Differential effect of honey on selected variables in alloxan-induced and fructose-induced diabetic rats. African J Biomed Res. 2008;11(39):191–196
Meo SA, Ansari MJ, Sattar K, Chaudhary HU, Hajjar W, Alasiri S. Honey and diabetes mellitus: obstacles and challenges–road to be repaired. Saudi J Biol Sci. 2017;24(5):1030–3.
Jayaraman S, Devarajan N, Rajagopal P, Babu S, Ganesan SK, Veeraraghavan VP, Palanisamy CP, Cui B, Periyasamy V, Chandrasekar K. β-Sitosterol circumvents obesity induced inflammation and insulin resistance by down-regulating IKKβ/NF-κB and JNK signaling pathway in adipocytes of type 2 diabetic rats. Molecules. 2021;26(7):2101. https://doi.org/10.3390/molecules26072101.
Balamash KS, Alkreathy HM, Al-Gahdali EH, Khoja SO, Ahmad A. Comparative biochemical and histopathological studies on the efficacy of metformin and virgin olive oil against streptozotocin-induced diabetes in Sprague-Dawley rats. J Diab Res. 2018;2018:1–10. https://doi.org/10.1155/2018/4692197.
Wei S, Han R, Zhao J, Wang S, Huang M, Wang Y, Chen Y. Intermittent administration of a fasting-mimicking diet intervenes in diabetes progression, restores β cells and reconstructs gut microbiota in mice. Nutr Metab. 2018;15:1–12. https://doi.org/10.1186/s12986-018-0318-3.
Acknowledgements
The authors express their gratitude to the technical personnel of Delta State University’s Department of Physiology in Abraka, Nigeria.
Author information
Authors and Affiliations
Contributions
Conceptualization E.A.C., N.E.K., M.O.O.; data curation, writing original draft preparation M.O.O., B.A.B., A.O.N.; review and editing B.A.B., E.G.N., N.E.K., M.O.O.; supervision N.E.K.; validation N.E.K., E.V.; funding acquisition E.A.C., N.E.K., P.A.E., U.D.S. All authors have read and agreed to the publishing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The Ethical Review Committee of Delta State University gave their approval to perform this study on 09/11/2021 with the reference number REC/FBMS/DELSU/21/121. The Delta State University Ethical Review Committee (DSUERC) guarantees that all institutional guidelines and regulations are followed, as well as that all adverse events are reported to the DSUERC as soon as possible.
Consent to participate
Not applicable.
Consent for publication
All authors gave their consents for the article to be published.
Conflict of interest
The authors declare no competing interests.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chijiokwu, E.A., Nwangwa, E.K., Oyovwi, M.O. et al. Possible homeostatic, glucose uptake mechanisms and hepato-pancreatic histological effects of intermittent fasting, exercise, starvation, and honey in streptozotocin-induced diabetes in rats. Nutrire 48, 16 (2023). https://doi.org/10.1186/s41110-023-00204-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s41110-023-00204-z