Abstract
Background
About 80% of the roughly 7,000 known rare diseases are single gene disorders, about 85% of which are ultra-rare, affecting less than one in one million individuals. NGS technologies, in particular whole genome sequencing (WGS) in paediatric patients suffering from severe disorders of likely genetic origin improve the diagnostic yield allowing targeted, effective care and management. The aim of this study is to perform a systematic review and meta-analysis to assess the effectiveness of WGS, with respect to whole exome sequencing (WES) and/or usual care, for the diagnosis of suspected genetic disorders among the paediatric population.
Methods
A systematic review of the literature was conducted querying relevant electronic databases, including MEDLINE, EMBASE, ISI Web of Science, and Scopus from January 2010 to June 2022. A random-effect meta-analysis was run to inspect the diagnostic yield of different techniques. A network meta-analysis was also performed to directly assess the comparison between WGS and WES.
Results
Of the 4,927 initially retrieved articles, thirty-nine met the inclusion criteria. Overall results highlighted a significantly higher pooled diagnostic yield for WGS, 38.6% (95% CI: [32.6 – 45.0]), in respect to WES, 37.8% (95% CI: [32.9 – 42.9]) and usual care, 7.8% (95% CI: [4.4 – 13.2]). The meta-regression output suggested a higher diagnostic yield of the WGS compared to WES after controlling for the type of disease (monogenic vs non-monogenic), with a tendency to better diagnostic performances for Mendelian diseases. The network meta-analysis showed a higher diagnostic yield for WGS compared to WES (OR = 1.54, 95%CI: [1.11 – 2.12]).
Conclusions
Although whole genome sequencing for the paediatric population with suspected genetic disorders provided an accurate and early genetic diagnosis in a high proportion of cases, further research is needed for evaluating costs, effectiveness, and cost-effectiveness of WGS and achieving an informed decision-making process.
Trial Registration
This systematic review has not been registered.
Similar content being viewed by others
Introduction
Online Mendelian Inheritance in Man (OMIM), an updated catalogue of human genes and genetic phenotypes, contains over 16,000 genes [1, 2], and more than 9,300 Mendelian phenotypes, including more than 6,000 with a known molecular defect. In addition, there are more than 1,500 confirmed Mendelian phenotype or a phenotypic locus with an unknown molecular basis and more than 1,700 additional phenotypes of suspected Mendelian origin [3]. According to Orphanet [4] there are roughly 7,000 rare diseases (RD), 80% of which are thought to have a genetic cause, the majority of which are Mendelian/monogenic disorders [5, 6].
Non-monogenic genetic diseases, also known as complex genetic diseases, are conversely caused by a combination of genetic, environmental, and lifestyle factors. Unlike monogenic diseases that are caused by a single gene mutation, non-monogenic diseases involve multiple genes, each contributing a small effect, as well as environmental and lifestyle factors. According to the European definition, rare diseases are life-threatening or chronically debilitating conditions with a prevalence of less than one case in 2,000 individuals, while the US figure is less than one case in 1,500 people [7, 8]. Although rare individually, these disorders affect 264–446 million of people worldwide, and 17.8–30.3 million in Europe [7, 9]. About 50 to 60% of RDs affect children, 12% of which are congenital and 42% have an onset in the first two years of life [9, 10]. Patients suffering from RD share similar needs, suffer diagnostic delay, uncertainty in genetic counselling, and lack of proper clinical management and care, since an effective treatment is available only for about 400 diseases. This is also due to failure in the identification of the molecular defects underlying a large number of these diseases. Genetic testing confirms or rules out a suspected genetic condition, is diagnostic in a proportion of clinically unsolved cases and determines the individual chance of develo** or passing on a genetic disorder. Over the past decade, the development of next generation sequencing (NGS) technologies and bioinformatic pipelines to manage and analyse genomic data, jointly with an impressive reduction of sequencing costs, have led to a widespread implementation of genomic sequencing, most often whole exome sequencing (WES). These tools, which can identify the molecular defect causing Mendelian disorders [11], have shown to be effective and sustainable in genomic medicine. As a powerful tool, genomic medicine has the potential to improve outcomes and reduce costs in primary care settings [12]. The application of whole genome sequencing (WGS) and the whole exome sequencing (WES) in new-borns and children suffering from a severe disorder of likely genetic origin is expected to improve targeted, effective care and management [13, 14].
WGS and WES are increasingly used for diagnostic purposes on critically ill infants and children admitted to Neonatal Intensive Care Units (NICU) and Paediatric Intensive Care Units (PICU) with a suspected genetic disorder [14,15,16]. Traditional genetic testing allows to reach the diagnosis in around 20% of cases [14]. Thus, acutely ill neonates with suspected genetic diseases are often discharged or deceased before diagnosis. As a result, NICU treatment of genetic diseases is usually empirical, may lack efficacy, may be inappropriate, or even may cause adverse effects [17].
Currently, whole exome sequencing (WES) is more commonly used globally than whole genome sequencing (WGS) due to its easier data storage and processing [18], as well as its cost-saving benefits [19]. Nevertheless, despite the widespread adoption of whole exome sequencing (WES), previous research has demonstrated that whole genome sequencing (WGS) has the potential to yield a greater number of diagnoses than WES both in undiagnosed adults and suspected genetic diseases of the newborn. Particularly, over a large number of studies, the diagnostic yield attained by WES ranges between 25 and 50% while the WGS diagnostic yield is about 40–60% [20, 21]. Furthermore, a recent systematic review and meta-analysis showed a greater diagnostic yield for WGS (0.41, 95% CI 0.34–0.48, I2 = 44%) compared to WES (0.36, 95% CI 0.33–0.40, I2 = 83%), although not statistically significant, and usual care (UC) (0.10, 95% CI 0.08–0.12, I2 = 81%) [22]. In addition, another systematic review reported a diagnostic yield ranging from 3 to 79% for WES and between 17 and 73% for WGS [23].
Recent studies have also supported the clinical utility of WGS, compared to standard testing, in NICU highlighting a higher diagnostic yield, a sharp increase in changes in clinical management, and shortening of the time to diagnosis thanks to the PCR-free WGS approach[24]. Therefore, a wider use of WGS could change acute management and life outcomes in children with chronic diseases using stratified therapeutics [14].
Marshall et al. [25]. In addition, the translation of WGS into clinical settings has been hindered by the lack of access to technology, complex infrastructure, and expert personnel. At present, in a context of limited healthcare resources, it is necessary to retrieve evidence on how to integrate the WGS technology in the diagnostics, fulfilling both the criteria of clinical utility and cost-effectiveness [26].
The aim of this study was to perform a systematic review and meta-analysis to assess the effectiveness of WGS, with respect to WES and/or UC, for the diagnosis of suspected genetic disorders among the paediatric population.
Materials and methods
Search strategy and selection criteria
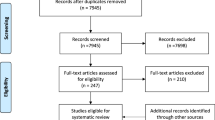
A systematic review of the literature was conducted querying relevant electronic databases, including MEDLINE, EMBASE, ISI Web of Science, and Scopus from January 2010 to June 2022 in order to retrieve peer-reviewed articles. The Population, Intervention, Comparison, Outcome (PICO) [27] framework was adopted to formulate the following research question: “Is implementing WGS for the care of the paediatric population effective?”. A comprehensive search strategy was created and implemented according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) [28] checklist. First, controlled descriptors and the relative key words were identified and verified in each scientific database. Afterwards, a Boolean search string, combining Medical Subject Headings (MeSH) and free-text words, such as “new born”, “infant”, “paediatric”, “paediatric”, “child”, “next-generation sequencing”, “whole genome sequencing”, “whole exome sequencing”, “genomic testing”, “panel test”, “diagnostic yield”, “effectiveness”, “appropriateness”, “efficacy”, “clinical efficacy”, “NICU”, “PICU”, “emergency”, was used. Full search strings for each database are detailed in the Supplementary Material. The search was completed by hand search in order to identify missing articles (i.e., snowball searching). Additional relevant articles were found by analysing bibliographic citations. The inclusion criteria for this systematic review were defined as follows: paediatric patients affected by severe life-threatening disorders of likely genetic origin undergoing WGS, and/or WES diagnostic test, either in an emergency setting (i.e., neonatal intensive care unit or NICU and paediatric intensive care unit or PICU) or in an outpatient setting. Where available in the included studies, UC was also considered. UC (e.g., chromosomal micro-array [CMA], targeted gene panel, array CGH, fluorescence in-situ hybridization, karyotype) was defined as sequencing methods not involving massively parallel sequencing and not allowing to screen simultaneously for mutations in hundreds of loci in genetically heterogeneous disorders, whole-genome screening for novel mutations, and sequence-based detection of novel pathogens that cause human diseases [29]. The inclusion was restricted to articles written in English and published between January 2010 and June 2022. The indicated timespan reflected the new sequencing technologies not available in older publications or being outdated owing to technological developments [30]. The search strategy was further restricted by availability of full texts published in peer-reviewed journals and by type of articles, which excluded non-primary literature, as commentary, books, thesis, and reviews. Assessment of the eligibility criteria was carried out independently by three authors; in the case of divergence, a fourth author was consulted. The primary outcome of our search was the diagnostic yield which was measured as the number of patients in which the genetic test suggested the definitive diagnosis out of the total number of patients undergoing the test. After the removal of duplicate articles, and according to the inclusion and exclusion criteria, three independent researchers performed the preliminary screening by evaluating the titles and abstracts. Then, the same subjects screened the full text of each study to determine the potential eligibility. In both screening phases, all disagreements were solved by a fourth author by discussing the inclusion and exclusion criteria of the article.
Data analysis
Data extraction was completed by three independent investigators. A pre-determined data extraction spreadsheet was designed including the following variables: study characteristics, country of the study, setting, sample size, age, intervention, comparator, indicators, and main findings. Methodological quality of studies evaluating diagnostic yield was assessed using the Quality Assessment of Diagnostic Accuracy studies (QUADAS-2) scale [29] as recommended by the Agency for Healthcare Research and Quality (AHRQ), the Cochrane Collaboration, and the National Institute for Health and Clinical Excellence (NICE). The use of QUADAS-2 implies four phases: (1) state the review question, (2) develop review specific guidance, (3) review the published flow diagram for the primary study or construction of a flow diagram if none is reported, and (4) judgement of bias and applicability. The scale includes four domains: (1) patient selection, (2) index test, (3) reference standard, and (4) flow and timing. The first part of each key domain regards bias and includes information used to support the risk of bias judgment, a set of signalling questions to help reviewers reach the judgements regarding bias, and judgment of risk of bias. For each signalling question, the investigator could select “yes,” “no,” or “unclear”. The risk of bias was judged as “low”, “high” or “unclear”. If all signalling questions for each domain answered “yes”, the risk of bias was judged “low”, while, if any signalling question answered “no”, the risk of bias was considered “high”. The term “unclear” was used whenever the risk of bias could not be assessed due to missing information. The second part of the first three domains regarded applicability. The applicability sections were similar to the bias sections except for the signalling questions. Concerns regarding applicability were rated as “low”, “high” or “unclear”, the latter definition being used when insufficient data were reported. Studies rated as “low” on all domains regarding bias or applicability received an overall judgment of “low risk of bias” or “low concern regarding applicability”. Studies rated as “high” in one or more domains were judged “at risk of bias” or as having “concern regarding applicability”. QUADAS-2 assessments were summarized through the relative tabular and graphical displays [30].
The Grading of Recommendations, Assessment, Development and Evaluations (GRADE) approach was adopted to assess the evidence quality for the outcome of interest across the included studies. The GRADE method categorizes the level of evidence quality into: high quality, further research is extremely unlikely to change the credibility of the pooled results; moderate quality, further research is likely to influence the credibility of pooled results and may change the estimate; low quality, further research is extremely likely to influence the credibility of pooled results and is likely to change the estimate; very low quality, the pooled results have extreme uncertainty [31]. The online GRADE profiler (GRADEpro) [32] was adopted to create evidence profiles and summary of findings tables. For the outcome of interest, the quality of evidence was downgraded if the risk of bias, inconsistency, indirectness, imprecision, and publication bias were assessed as having serious limitations [33]. The overall quality was rated as either “high”, “moderate”, “low”, or “very low”.
Quantitative data synthesis was performed by always kee** the diagnostic yield, in terms of proportion of cases detected out of the total, as outcome of reference. Firstly, the diagnostic yield was meta-analysed, by inspecting differences between diverse techniques (WES, WGS, and UC) via subgroup analyses: to this purpose, in view of the expected heterogeneity among studies [34], random-effects models were developed according to DerSimonian and Laird [35] and heterogeneity was inspected using the I2 statistic (threshold level for significant heterogeneity: ≥ 50%) and chi-squared test for homogeneity (significance level for heterogeneity: p < 0.1) [36]. Given the available number of studies, a meta-regression model was also built, in order to compare the techniques by adjusting for relevant covariates (ICU vs non-ICU setting, Mendelian vs non-Mendelian disease, publication before vs after 2017). Another meta-regression model was run stratifying by the value (i.e., low and high) of the diagnostic yield reported by the primary studies included in the revision. The cut-off value was set according to the pooled diagnostic yield estimated through the random-effects meta-analysis.
Secondly, a network meta-analysis was performed by considering all studies comparing at least two of the three techniques. A frequentist approach based on the Mantel–Haenszel method for binary data, as described by Efthimiou et al. [37], was adopted. Heterogeneity was quantified through the test of inconsistency (Cochran’s Q statistic), and the odds ratio was chosen as summary measure, as widely recommended for indirect comparisons of binary variables because of the symmetry and invariance of this measure to the coding of event and non-event [38,39,40].
All statistical analyses were carried out, and plots were drawn, using the statistical software R (version 4.0.5) [41]: specifically, the “meta” package (version 5.0.0) [42] was used for the meta-analysis of proportions and meta-regression, while the “netmeta” package (version 2.0–0) was used for the network meta-analysis [43]. Two-sided p-values < 0.05 were considered statistically significant.
Role of the funding source
The funding source had no involvement in study design; in the collection, analysis and interpretation of data; in the writing of the report; and in the decision to submit the article for publication.
Results
The database search resulted in 4,927 publications and 18 studies were retrieved through the snowball search method. After duplicates elimination, 3,955 titles and abstracts were screened. A total of 63 articles were identified for a full-text screening. After full-text examination, 24 papers were excluded since they did not fulfil the eligibility criteria. Thus, 39 [15, 16, 24, 82]. Further research rigorously assessing costs, effectiveness, cost-effectiveness, organizational impacts, ethical aspects of WGS in a health technology assessment perspective [83] in a transparent manner is mandatory to allow for a more informed decision-making process in this context.
Moreover, additional primary studies (preferably high-quality RCTs with larger samples) firstly evaluating the comparison between WGS and WES and then between WGS and standard genetic testing, are required to deeply investigate the differences in costs and diagnostic yield and to increase the level of quality evidence.
Conclusion
Whole genome sequencing for the paediatric population with suspected genetic disorders allows an accurate and early genetic diagnosis in a high proportion of cases. This provides understanding of the molecular mechanism underlying diseases, supports tailored treatments and accurate genetic counselling, while reduces the burden of unsolved cases that weigh on patients and their families [25] by putting end to the so called “diagnostic Odyssey” [84, 85]. The present review suggests the use of WGS in the diagnostic workup of ill paediatric patients with suspected genetic disorders strengthened by evidence at policy, program, and intervention levels. Our study also reinforces the use of methodologies capable of providing robust evidence for the formulation of health policy on funding, to overcome present hurdles in transitioning WGS from the research setting into routine clinical practice. However, there is a pressing issue of efficiently allocating limited healthcare resources for HTA agencies when it comes to WGS approaches. Overcoming this challenge will be critical to realizing the potential benefits of WGS for improving patient outcomes and reducing healthcare costs.
Availability of data and materials
Aggregated data and materials might be made available upon reasonable request via email to the corresponding author.
References
Amberger JS, Bocchini CA, Schiettecatte F, Scott AF, Hamosh A. OMIM.org: Online Mendelian Inheritance in Man (OMIM®), an online catalog of human genes and genetic disorders. Nucleic Acids Res. 2015;43:D789-98. https://doi.org/10.1093/nar/gku1205.
McKusick-Nathans Institute of Genetic Medicine JHU. Online Mendelian Inheritance in Man, OMIM®. 2021. https://omim.org. Accessed April 2, 2021.
Online Mendelian Inheritance in Man®. OMIM Entry Statistics. 2021. https://www.omim.org/statistics/entry. Accessed April 2, 2021 .
Orphanet. Orphanet Reports Series / Procedures. 2021.
Wright CF, FitzPatrick DR, Firth HV. Paediatric genomics: diagnosing rare disease in children. Nat Rev Genet. 2018;19:253–68. https://doi.org/10.1038/nrg.2017.116.
Rahit KMTH, Tarailo-Graovac M. Genetic Modifiers and Rare Mendelian Disease. Genes (Basel). 2020;11:239. https://doi.org/10.3390/genes11030239.
EURORDIS. About Rare Diseases. 2020. https://www.eurordis.org/about-rare-diseases. Accessed 2 April 2021.
Richter T, Nestler-Parr S, Babela R, Khan ZM, Tesoro T, Molsen E, et al. Rare Disease Terminology and Definitions—A Systematic Global Review: Report of the ISPOR Rare Disease Special Interest Group. Value Health. 2015;18:906–14. https://doi.org/10.1016/j.jval.2015.05.008.
Nguengang Wakap S, Lambert DM, Olry A, Rodwell C, Gueydan C, Lanneau V, et al. Estimating cumulative point prevalence of rare diseases: analysis of the Orphanet database. Eur J Hum Genet. 2020;28:165–73. https://doi.org/10.1038/s41431-019-0508-0.
Kole, Anna, Faurisson François, Mavris M. The Voice of 12,000 Patients. 2009.
Lu JT, Campeau PM, Lee BH. Genotype-Phenotype Correlation — Promiscuity in the Era of Next-Generation Sequencing. N Engl J Med. 2014;371:593–6. https://doi.org/10.1056/NEJMp1400788.
Radio FC, Ruzzeddu M, Bartuli A, Novelli A, Tartaglia M, Dallapiccola B. Cost-effectiveness of exome sequencing: an Italian pilot study on undiagnosed patients. New Genet Soc 2019;38. https://doi.org/10.1080/14636778.2019.1601008.
Rae W, Ward D, Mattocks C, Pengelly RJ, Eren E, Patel SV, et al. Clinical efficacy of a next-generation sequencing gene panel for primary immunodeficiency diagnostics. Clin Genet. 2018;93:647–55. https://doi.org/10.1111/cge.13163.
French CE, Delon I, Dolling H, Sanchis-Juan A, Shamardina O, Mégy K, et al. Whole genome sequencing reveals that genetic conditions are frequent in intensively ill children. Intensive Care Med. 2019;45:627–36. https://doi.org/10.1007/s00134-019-05552-x.
Meng L, Pammi M, Saronwala A, Magoulas P, Ghazi AR, Vetrini F, et al. Use of Exome Sequencing for Infants in Intensive Care Units. JAMA Pediatr. 2017;171:e173438. https://doi.org/10.1001/jamapediatrics.2017.3438.
Śmigiel R, Biela M, Szmyd K, Błoch M, Szmida E, Skiba P, et al. Rapid Whole-Exome Sequencing as a Diagnostic Tool in a Neonatal/Pediatric Intensive Care Unit. J Clin Med. 2020;9:2220. https://doi.org/10.3390/jcm9072220.
Saunders CJ, Miller NA, Soden SE, Dinwiddie DL, Noll A, Alnadi NA, et al. Rapid Whole-Genome Sequencing for Genetic Disease Diagnosis in Neonatal Intensive Care Units. Sci Transl Med. 2012;4:154ra135. https://doi.org/10.1126/scitranslmed.3004041.
Barbitoff YA, Polev DE, Glotov AS, Serebryakova EA, Shcherbakova IV, Kiselev AM, et al. Systematic dissection of biases in whole-exome and whole-genome sequencing reveals major determinants of coding sequence coverage. Sci Rep. 2020;10:2057. https://doi.org/10.1038/s41598-020-59026-y.
van Nimwegen KJM, van Soest RA, Veltman JA, Nelen MR, van der Wilt GJ, Vissers LELM, et al. Is the $1000 Genome as Near as We Think? A Cost Analysis of Next-Generation Sequencing. Clin Chem. 2016;62:1458–64. https://doi.org/10.1373/clinchem.2016.258632.
Radio FC, Ruzzeddu M, Bartuli A, Novelli A, Tartaglia M, Dallapiccola B. Cost-effectiveness of exome sequencing: an Italian pilot study on undiagnosed patients. New Genet Soc. 2019;38:249–63. https://doi.org/10.1080/14636778.2019.1601008.
Mattick JS, Dinger M, Schonrock N, Cowley M. Whole genome sequencing provides better diagnostic yield and future value than whole exome sequencing. Med J Aust. 2018;209:197–9. https://doi.org/10.5694/mja17.01176.
Clark MM, Stark Z, Farnaes L, Tan TY, White SM, Dimmock D, et al. Meta-analysis of the diagnostic and clinical utility of genome and exome sequencing and chromosomal microarray in children with suspected genetic diseases. NPJ Genom Med. 2018;3:16. https://doi.org/10.1038/s41525-018-0053-8.
Schwarze K, Buchanan J, Taylor JC, Wordsworth S. Are whole-exome and whole-genome sequencing approaches cost-effective? A systematic review of the literature. Genet Med. 2018;20:1122–30. https://doi.org/10.1038/gim.2017.247.
Sanford EF, Clark MM, Farnaes L, Williams MR, Perry JC, Ingulli EG, et al. Rapid Whole Genome Sequencing Has Clinical Utility in Children in the PICU*. Pediatr Crit Care Med. 2019;20:1007–20. https://doi.org/10.1097/PCC.0000000000002056.
Marshall CR, Bick D, Belmont JW, Taylor SL, Ashley E, Dimmock D, et al. The Medical Genome Initiative: moving whole-genome sequencing for rare disease diagnosis to the clinic. Genome Med. 2020;12:48. https://doi.org/10.1186/s13073-020-00748-z.
Daoud H, Luco SM, Li R, Bareke E, Beaulieu C, Jarinova O, et al. Next-generation sequencing for diagnosis of rare diseases in the neonatal intensive care unit. Can Med Assoc J. 2016;188:E254–60. https://doi.org/10.1503/cmaj.150823.
O’Connor, Denise Ann. Green, Sally Elizabeth. Higgins JP. Defining the review question and develo** criteria for including studies. In: Green, Sally Elizabeth. Higgins JP, editor. Cochrane Handbook for Systematic Reviews of Interventions. First: John Wiley & Sons, Ltd; 2008, 83–94.
Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. 2009;339:b2535–b2535. https://doi.org/10.1136/bmj.b2535.
Whiting PF. QUADAS-2: A Revised Tool for the Quality Assessment of Diagnostic Accuracy Studies. Ann Intern Med. 2011;155:529. https://doi.org/10.7326/0003-4819-155-8-201110180-00009.
University of Bristol. QUADAS-2: Background Document. 2020. https://www.bristol.ac.uk/media-library/sites/quadas/migrated/documents/background-doc.pdf. Accessed 15 March 2021.
Guyatt G, Oxman AD, Akl EA, Kunz R, Vist G, Brozek J, et al. GRADE guidelines: 1. Introduction—GRADE evidence profiles and summary of findings tables. J Clin Epidemiol. 2011;64:383–94. https://doi.org/10.1016/j.jclinepi.2010.04.026.
McMaster University and Evidence Prime Inc. GRADEpro GDT: GRADEpro Guideline Development Tool [Software]. 2020. https://gradepro.org. Accessed 18 March 2021.
Balshem H, Helfand M, Schünemann HJ, Oxman AD, Kunz R, Brozek J, et al. GRADE guidelines: 3. Rating the quality of evidence. J Clin Epidemiol. 2011;64:401–6. https://doi.org/10.1016/j.jclinepi.2010.07.015.
Borenstein M, Hedges L v, Higgins JPT, Rothstein HR. A basic introduction to fixed-effect and random-effects models for meta-analysis. Res Synth Methods. 2010;1:97–111. https://doi.org/10.1002/jrsm.12.
DerSimonian R, Laird N. Meta-analysis in clinical trials revisited. Contemp Clin Trials. 2015;45:139–45. https://doi.org/10.1016/j.cct.2015.09.002.
Higgins JPT. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557–60. https://doi.org/10.1136/bmj.327.7414.557.
Efthimiou O, Rücker G, Schwarzer G, Higgins JPT, Egger M, Salanti G. Network meta-analysis of rare events using the Mantel-Haenszel method. Stat Med. 2019;38:2992–3012. https://doi.org/10.1002/sim.8158.
Caldwell DM, Welton NJ, Dias S, Ades A. Selecting the best scale for measuring treatment effect in a network meta-analysis: a case study in childhood nocturnal enuresis. Res Synth Methods. 2012;3:126–41. https://doi.org/10.1002/jrsm.1040.
Coory M, Jordan S. Frequency of treatment-effect modification affecting indirect comparisons. Pharmacoeconomics. 2010;28:723–32. https://doi.org/10.2165/11535670-000000000-00000.
Eckermann S, Coory M, Willan AR. Indirect comparison: relative risk fallacies and odds solution. J Clin Epidemiol. 2009;62:1031–6. https://doi.org/10.1016/j.jclinepi.2008.10.013.
R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing 2021. https://www.r-project.org/ (Accessed 10 Feb 2022).
Balduzzi S, Rücker G, Schwarzer G. How to perform a meta-analysis with R: a practical tutorial. Evid Based Mental Health. 2019;22:153–60. https://doi.org/10.1136/ebmental-2019-300117.
Rücker G, Krahn U, König J, Efthimiou O, Davies A, Papakonstantinou T, et al. Package ‘netmeta’. 2022.
Zhu X, Petrovski S, **e P, Ruzzo EK, Lu Y-F, McSweeney KM, et al. Whole-exome sequencing in undiagnosed genetic diseases: interpreting 119 trios. Genet Med. 2015;17:774–81. https://doi.org/10.1038/gim.2014.191.
Zhu T, Gong X, Bei F, Ma L, Chen Y, Zhang Y, et al. Application of Next-Generation Sequencing for Genetic Diagnosis in Neonatal Intensive Care Units: Results of a Multicenter Study in China. Front Genet 2020;11. https://doi.org/10.3389/fgene.2020.565078.
Zhang L, Gao J, Liu H, Tian Y, Zhang X, Lei W, et al. Pathogenic variants identified by whole-exome sequencing in 43 patients with epilepsy. Hum Genomics. 2020;14:44. https://doi.org/10.1186/s40246-020-00294-0.
Willig LK, Petrikin JE, Smith LD, Saunders CJ, Thiffault I, Miller NA, et al. Whole-genome sequencing for identification of Mendelian disorders in critically ill infants: a retrospective analysis of diagnostic and clinical findings. Lancet Respir Med. 2015;3:377–87. https://doi.org/10.1016/S2213-2600(15)00139-3.
Vissers LELM, van Nimwegen KJM, Schieving JH, Kamsteeg E-J, Kleefstra T, Yntema HG, et al. A clinical utility study of exome sequencing versus conventional genetic testing in pediatric neurology. Genet Med. 2017;19:1055–63. https://doi.org/10.1038/gim.2017.1.
Valencia CA, Husami A, Holle J, Johnson JA, Qian Y, Mathur A, et al. Clinical impact and cost-effectiveness of whole exome sequencing as a diagnostic tool: a pediatric center’s experience. Front Pediatr 2015;3. https://doi.org/10.3389/fped.2015.00067.
Thevenon J, Duffourd Y, Masurel-Paulet A, Lefebvre M, Feillet F, el Chehadeh-Djebbar S, et al. Diagnostic odyssey in severe neurodevelopmental disorders: toward clinical whole-exome sequencing as a first-line diagnostic test. Clin Genet. 2016;89:700–7. https://doi.org/10.1111/cge.12732.
Taylor JC, Martin HC, Lise S, Broxholme J, Cazier J-B, Rimmer A, et al. Factors influencing success of clinical genome sequencing across a broad spectrum of disorders. Nat Genet. 2015;47:717–26. https://doi.org/10.1038/ng.3304.
Tan TY, Dillon OJ, Stark Z, Schofield D, Alam K, Shrestha R, et al. Diagnostic Impact and Cost-effectiveness of Whole-Exome Sequencing for Ambulant Children With Suspected Monogenic Conditions. JAMA Pediatr. 2017;171:855. https://doi.org/10.1001/jamapediatrics.2017.1755.
Stavropoulos DJ, Merico D, Jobling R, Bowdin S, Monfared N, Thiruvahindrapuram B, et al. Whole-genome sequencing expands diagnostic utility and improves clinical management in paediatric medicine. NPJ Genom Med. 2016;1:15012. https://doi.org/10.1038/npjgenmed.2015.12.
Stark Z, Tan TY, Chong B, Brett GR, Yap P, Walsh M, et al. A prospective evaluation of whole-exome sequencing as a first-tier molecular test in infants with suspected monogenic disorders. Genet Med. 2016;18:1090–6. https://doi.org/10.1038/gim.2016.1.
Scocchia A, Wigby KM, Masser-Frye D, del Campo M, Galarreta CI, Thorpe E, et al. Clinical whole genome sequencing as a first-tier test at a resource-limited dysmorphology clinic in Mexico. NPJ Genom Med. 2019;4:5. https://doi.org/10.1038/s41525-018-0076-1.
Retterer K, Juusola J, Cho MT, Vitazka P, Millan F, Gibellini F, et al. Clinical application of whole-exome sequencing across clinical indications. Genet Med. 2016;18:696–704. https://doi.org/10.1038/gim.2015.148.
Qi ZY, Duan J, He XY, Zhong Q-H, Zhang CY, **e YB, et al. [Clinical application of whole exome sequencing in monogenic hereditary disorders in critically ill newborns]. Zhongguo Dang Dai Er Ke Za Zhi. 2019;21:640–3.
Petrikin JE, Cakici JA, Clark MM, Willig LK, Sweeney NM, Farrow EG, et al. The NSIGHT1-randomized controlled trial: rapid whole-genome sequencing for accelerated etiologic diagnosis in critically ill infants. NPJ Genom Med. 2018;3:6. https://doi.org/10.1038/s41525-018-0045-8.
Monies D, Abouelhoda M, AlSayed M, Alhassnan Z, Alotaibi M, Kayyali H, et al. The landscape of genetic diseases in Saudi Arabia based on the first 1000 diagnostic panels and exomes. Hum Genet. 2017;136:921–39. https://doi.org/10.1007/s00439-017-1821-8.
Lionel AC, Costain G, Monfared N, Walker S, Reuter MS, Hosseini SM, et al. Improved diagnostic yield compared with targeted gene sequencing panels suggests a role for whole-genome sequencing as a first-tier genetic test. Genet Med. 2018;20:435–43. https://doi.org/10.1038/gim.2017.119.
Kingsmore SF, Cakici JA, Clark MM, Gaughran M, Feddock M, Batalov S, et al. A Randomized, Controlled Trial of the Analytic and Diagnostic Performance of Singleton and Trio, Rapid Genome and Exome Sequencing in Ill Infants. Am J HumGenet. 2019;105:719–33. https://doi.org/10.1016/j.ajhg.2019.08.009.
Farwell KD, Shahmirzadi L, El-Khechen D, Powis Z, Chao EC, Tippin Davis B, et al. Enhanced utility of family-centered diagnostic exome sequencing with inheritance model–based analysis: results from 500 unselected families with undiagnosed genetic conditions. Genet Med. 2015;17:578–86. https://doi.org/10.1038/gim.2014.154.
Farnaes L, Hildreth A, Sweeney NM, Clark MM, Chowdhury S, Nahas S, et al. Rapid whole-genome sequencing decreases infant morbidity and cost of hospitalization. NPJ Genom Med. 2018;3:10. https://doi.org/10.1038/s41525-018-0049-4.
Eldomery MK, Coban-Akdemir Z, Harel T, Rosenfeld JA, Gambin T, Stray-Pedersen A, et al. Lessons learned from additional research analyses of unsolved clinical exome cases. Genome Med. 2017;9:26. https://doi.org/10.1186/s13073-017-0412-6.
Du X, Gao X, Liu X, Shen L, Wang K, Fan Y, et al. Genetic Diagnostic Evaluation of Trio-Based Whole Exome Sequencing Among Children With Diagnosed or Suspected Autism Spectrum Disorder. Front Genet 2018;9. https://doi.org/10.3389/fgene.2018.00594.
Charng W-L, Karaca E, Coban Akdemir Z, Gambin T, Atik MM, Gu S, et al. Exome sequencing in mostly consanguineous Arab families with neurologic disease provides a high potential molecular diagnosis rate. BMC Med Genomics. 2016;9:42. https://doi.org/10.1186/s12920-016-0208-3.
Bick D, Fraser P, Gutzeit M, Harris J, Hambuch T, Helbling D, et al. Successful Application of Whole Genome Sequencing in a Medical Genetics Clinic. J Pediatr Genet. 2016;06:061–76. https://doi.org/10.1055/s-0036-1593968.
da Leite AJC, Pinto IP, Leijsten N, Ruiterkamp-Versteeg M, Pfundt R, de Leeuw N, et al. Diagnostic yield of patients with undiagnosed intellectual disability, global developmental delay and multiples congenital anomalies using karyotype, microarray analysis, whole exome sequencing from Central Brazil. PLoS One. 2022;17:e0266493. https://doi.org/10.1371/journal.pone.0266493.
Wu B, Kang W, Wang Y, Zhuang D, Chen L, Li L, et al. Application of Full-Spectrum Rapid Clinical Genome Sequencing Improves Diagnostic Rate and Clinical Outcomes in Critically Ill Infants in the China Neonatal Genomes Project*. Crit Care Med. 2021;49:1674–83. https://doi.org/10.1097/CCM.0000000000005052.
Sharma S, Repnikova E, Noel-MacDonnell JR, LePichon J. Diagnostic yield of genetic testing in 324 infants with hypotonia. Clin Genet. 2021;100:752–7. https://doi.org/10.1111/cge.14057.
Schon KR, Horvath R, Wei W, Calabrese C, Tucci A, Ibañez K, et al. Use of whole genome sequencing to determine genetic basis of suspected mitochondrial disorders: cohort study. BMJ 2021:e066288. https://doi.org/10.1136/bmj-2021-066288.
Scholz T, Blohm ME, Kortüm F, Bierhals T, Lessel D, van der Ven AT, et al. Whole-exome sequencing in critically Ill neonates and infants: diagnostic yield and predictability of monogenic diagnosis. Neonatology. 2021;118:454–61. https://doi.org/10.1159/000516890.
Ripen AM, Chear CT, Baharin MF, Nallusamy R, Chan KC, Kassim A, et al. A single-center pilot study in Malaysia on the clinical utility of whole-exome sequencing for inborn errors of immunity. Clin Exp Immunol. 2021;206:119–28. https://doi.org/10.1111/cei.13626.
Liu Y, Liu X, Qin D, Zhao Y, Cao X, Deng X, et al. Clinical utility of next-generation sequencing for developmental disorders in the rehabilitation department: experiences from a Single Chinese Center. J Mol Neurosci. 2021;71:845–53. https://doi.org/10.1007/s12031-020-01707-4.
Liu Y, Hao C, Li K, Hu X, Gao H, Zeng J, et al. Clinical application of whole exome sequencing for monogenic disorders in PICU of China. Front Genet 2021;12. https://doi.org/10.3389/fgene.2021.677699.
Krantz ID, Medne L, Weatherly JM, Wild KT, Biswas S, Devkota B, et al. Effect of Whole-Genome Sequencing on the Clinical Management of Acutely Ill Infants With Suspected Genetic Disease. JAMA Pediatr. 2021;175:1218. https://doi.org/10.1001/jamapediatrics.2021.3496.
Kose M, Isik E, Aykut A, Durmaz A, Kose E, Ersoy M, et al. The utility of next-generation sequencing technologies in diagnosis of Mendelian mitochondrial diseases and reflections on clinical spectrum. J Pediatr Endocrinol Metab. 2021;34:417–30. https://doi.org/10.1515/jpem-2020-0410.
Herman I, Lopez MA, Marafi D, Pehlivan D, Calame DG, Abid F, et al. Clinical exome sequencing in the diagnosis of pediatric neuromuscular disease. Muscle Nerve. 2021;63:304–10. https://doi.org/10.1002/mus.27112.
Dimmock D, Caylor S, Waldman B, Benson W, Ashburner C, Carmichael JL, et al. Project Baby Bear: Rapid precision care incorporating rWGS in 5 California children’s hospitals demonstrates improved clinical outcomes and reduced costs of care. Am J Hum Genet. 2021;108:1231–8. https://doi.org/10.1016/j.ajhg.2021.05.008.
Stavropoulos DJ, Merico D, Jobling R, Bowdin S, Monfared N, Thiruvahindrapuram B, et al. Whole-genome sequencing expands diagnostic utility and improves clinical management in paediatric medicine. NPJ Genom Med. 2016;1:1–9. https://doi.org/10.1038/npjgenmed.2015.12.
Brand A, Brand H, Schulte in den Bäumen T. The impact of genetics and genomics on public health. Eur J Hum Genet. 2008;16:5–13. https://doi.org/10.1038/sj.ejhg.5201942.
Mazzarotto F, Olivotto I, Walsh R. Advantages and perils of clinical whole-exome and whole-genome sequencing in cardiomyopathy. Cardiovasc Drugs Ther. 2020;34:241–53. https://doi.org/10.1007/s10557-020-06948-4.
EUnetHTA Joint Action 2 WP 8. HTA Core Model ® version 3.0. 2016. www.htacoremodel.info/BrowseModel.aspx . Accessed 10 April 2021.
Abolhassani H, Aghamohammadi A, Fang M, Rezaei N, Jiang C, Liu X, et al. Clinical implications of systematic phenoty** and exome sequencing in patients with primary antibody deficiency. Genet Med. 2019;21:243–51. https://doi.org/10.1038/s41436-018-0012-x.
Berberich AJ, Ho R, Hegele RA. Whole genome sequencing in the clinic: empowerment or too much information? Can Med Assoc J. 2018;190:E124–5. https://doi.org/10.1503/cmaj.180076.
Acknowledgements
The working group would like to thank Dr. Gianmarco Raspolini for his methodological support and Dr. Aurora Heidar Alizadeh for the final revision and proofreading of the manuscript.
Funding
This work was supported by the Italian Ministry of Health [grant number RF-2018 12366931]. The funding source had no involvement in study design; in the collection, analysis and interpretation of data; in the writing of the report; and in the decision to submit the article for publication.
Author information
Authors and Affiliations
Contributions
Mario Cesare Nurchis, Gianfranco Damiani, Bruno Dallapiccola, Marco Tartaglia and Giovanni Chillemi had the idea for and conceptualized the study, coordinated and supervised data collection, and critically reviewed the manuscript for important intellectual content. Mario Cesare Nurchis, Gerardo Altamura and Jacopo Garlasco analysed the data and designed the statistical analyses. Mario Cesare Nurchis, Gerardo Altamura, Jacopo Garlasco, Maria Teresa Riccardi, Enrico Silvio Bertini and Francesca Clementina Radio interpreted the analyses, visualized the results, and wrote the initial draft of the manuscript. All authors contributed intellectually to this study, and critically reviewed the scientific content of the manuscript. All authors approved the final manuscript as submitted and agreed to be accountable for all aspects of the work. Mario Cesare Nurchis is the guarantor of this work and, as such, had full access to all the data in the study, verified the data, and take responsibility for the integrity of the data and the accuracy of the data analysis. All authors had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
All authors declare that there is no competing interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1.
Aims and scope.
Additional file 2: Table S1.
Characteristics of the studies included in the systematic review andmeta-analysis, 2015–2022. Table S2. Results of the Quality Assessment of Diagnostic Accuracy Studies -2 (QUADAS-2) tool for studiesincluded in systematic review and meta-analysis, 2015–2022. Table S3. GRADE Evidence Profiles for studies included in the systematic review and meta-analysis, 2015–2022. Table S4. Summary of findings table for studies included in the systematic review and meta-analysis, 2015–2022. Table S5. Meta-regression analysis considering studies with lower diagnostic yield (i.e., inferior to the pooledestimate for each diagnostic technique). Table S6. Meta-regression analysis considering studies with higher diagnostic yield (i.e., superior to the pooled estimate for each diagnostic technique).
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Nurchis, M.C., Altamura, G., Riccardi, M.T. et al. Whole genome sequencing diagnostic yield for paediatric patients with suspected genetic disorders: systematic review, meta-analysis, and GRADE assessment. Arch Public Health 81, 93 (2023). https://doi.org/10.1186/s13690-023-01112-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13690-023-01112-4