Abstract
Background
Toxoplasma gondii is a neurotropic single-celled parasite that can infect mammals, including humans. Central nervous system infection with T. gondii infection can lead to Toxoplasma encephalitis. Toxoplasma infection can cause endoplasmic reticulum (ER) stress and unfolded protein response (UPR) activation, which ultimately can lead to apoptosis of host cells. The dense granule protein GRA3 has been identified as one of the secretory proteins that contribute to the virulence of T. gondii; however, the mechanism remains enigmatic.
Methods
The expression of the GRA3 gene in RH, ME49, Wh3, and Wh6 strains was determined using quantitative real-time polymerase chain reaction (qRT–PCR). pEGFP-GRA3Wh6 was constructed by inserting Chinese 1 Wh6 GRA3 (GRA3Wh6) cDNA into a plasmid encoding the enhanced GFP. Mouse neuro2a (N2a) cells were transfected with either pEGFP or pEGFP-GRA3Wh6 (GRA3Wh6) and incubated for 24–36 h. N2a cell apoptosis and ER stress-associated proteins were determined using flow cytometry and immunoblotting. Furthermore, N2a cells were pretreated with GSK2656157 (a PERK inhibitor) and Z-ATAD-FMK (a caspase-12 inhibitor) before GRA3Wh6 transfection, and the effect of the inhibitors on GRA3Wh6-induced ER stress and apoptosis were investigated.
Results
GRA3 gene expression was higher in the less virulent strains of type II ME49 and type Chinese 1 Wh6 strains compared with the virulent strains of type I RH strain and type Chinese 1 Wh3 strain. Transfection with GRA3Wh6 plasmid induced neuronal apoptosis and increased the expression of GRP78, p-PERK, cleaved caspase-12, cleaved caspase-3, and CHOP compared with the control vector. Pretreatment with GSK2656157 and Z-ATAD-FMK decreased apoptosis in N2a cells, and similarly, ER stress- and apoptosis-associated protein levels were significantly decreased.
Conclusion
GRA3 induces neural cell apoptosis via the ER stress signaling pathway, which could play a role in toxoplasmic encephalitis.
Graphical Abstract

Similar content being viewed by others
Background
Toxoplasma gondii is a common intracellular coccidian parasite that infects human beings and animals [1,2,3]. Most T. gondii infections are usually asymptomatic and result in a self-limiting disease in immunocompetent hosts [4]; however, in chronic immunocompromised individuals, in particular patients with HIV, T. gondii can cause severe and fatal tissue damage [4, 5]. In pregnant women, T. gondii can cause miscarriage or deleterious effects to infants or newborns [6,7,8]. Due to its preference for neural cells, Toxoplasma infection is responsible for neurological manifestations, including encephalitis, intracranial calcifications, and hydrocephalus [9,10,11].
Several conditions, including infectious and neurodegenerative diseases, are known to cause a build-up of misfolded proteins within the endoplasmic reticulum that interfere with the normal functioning of the endoplasmic reticulum (ER). This leads to ER stress [12, 13]. To alleviate the effect of the stress, ER-localized transmembrane signal proteins activate the unfolded protein response (UPR) to restore protein homeostasis [14, 15]. However, an unremitting UPR can activate UPR-mediated inflammatory and apoptotic pathways, resulting in cell death [16, 17].
Previous studies have demonstrated that the Chinese 1 Wh3 and type I RH strains can induce neural stem cell apoptosis via the ER stress-mediated apoptosis signaling pathway [18, 19]. Additionally, secretory proteins such as ROP18 and GRA15 have been demonstrated to induce apoptosis of neural cells [10, 19] and carcinoma JEG-3 cells [24]. Because the highest expression of GRA3 was found in the Wh6 strain compared with RH, ME49, and Wh3 strains, we adopted GRA3Wh6 for subsequent experiments.
Dense granule protein 3 (GRA3) gene expression among different isolates. GRA3 expression levels were compared between virulent RH and Wh3 strains and less virulent ME49 and Wh6 strains. The RT-qPCR was performed in triplicate and values were expressed as mean ± SD. *P < 0.05, ***P < 0.001, and ns, not statistically significant
Expression of GRA3Wh6 in N2a cells
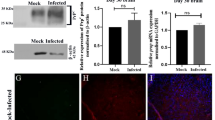
To determine whether GRA3Wh6 plasmid could be efficiently expressed in neural cells, we transfected pEGFP or pEGFP-GRA3Wh6 (GRA3Wh6) into the mouse neuroblastoma N2a cells and determined the expression of GRA3Wh6 protein. Twenty-four hours after transfection, GFP fluorescence was observed in both pEGFP- and pEGFP-GRA3Wh6-transfected N2a cells. The fluorescence signal in pEGFP-GRA3Wh6-transfected N2a cells was comparable to that in pEGFP-transfected cells (Fig. 2a, b). pEGFP and pEGFP-GRA3Wh6 fusion protein were blotted at 28 kDa and 58 kDa following a 24 h transfection (Fig. 2c). We noted that the observed molecular weight of GRA3 was ~ 30 kDa, which was similar to that of previous studies [21, 23].
Expression of dense granule protein 3 (GRA3Wh6). Mouse neuroblastoma (N2a) cells were transfected with either a control vector (pEGFP, plasmid encoding enhanced green fluorescent protein) or pEGFP-GRA3Wh6 fusion protein (GRA3Wh6) for a period of 24 h. Mock-transfected N2a cells served as a negative control. a The expression of green fluorescent protein (GFP) in pEGFP and pEGFP-GRA3Wh6 transfected N2a cells was observed using fluorescence microscopy. Scale-bar 20 µM. b The GFP intensity in pEGFP and pEGFP-GRA3Wh6-transfected N2a cells was measured using ImageJ. ns, not statistically significant. c The expression of GRA3Wh6 was confirmed by immunoblotting. Molecular weight (MW) of GFP = 28k Da, MW of pEGFP-GRA3Wh6 fusion protein = 58 kDa
Reduced cell viability and apoptosis of neuronal cells transfected with GRA3Wh6 in vitro
To investigate whether GRA3 could impact the survival of neural cells, the cell viability and rate of apoptosis in N2a cells transfected with either pEGFP or pEGFP-GRA3Wh6 (GRA3Wh6) were analyzed. The viability of mock-transfected N2a cells was 91.89% whereas staurosporine, which represented a positive control, decreased the viability of N2a cells to 49.9%. On the other hand, GRA3Wh6 showed a significant decrease in the number of viable N2a cells (P < 0.0001, 63.8% vs 80.7%) compared with the pEGFP-transfected cells (Fig. 3a). To further determine the rate of apoptosis, flow cytometry analysis using Annexin V-PE/7-AAD staining assay was performed. The results showed that GRA3Wh6-transfected N2a cells significantly promoted apoptosis (P < 0.0001, 44.8% vs 6.1%) compared with pEGFP-transfected N2a cells. The apoptosis rate in the mock-transfected cells was 5.2%, which was similar to that in pEGFP-transfected N2a cells (Fig. 3b).
Dense granule protein 3 (GRA3Wh6)-induced loss of cell viability and apoptosis. N2a cells were transfected with either a control vector (pEGFP) or pEGFP-GRA3Wh6 (GRA3Wh6) for a 24 h period. Mock-transfected N2a cells served as the negative control, and N2a cells treated with staurosporine (1 μM, 12 h) served as the positive control. a Cell viability was measured using the trypan blue staining cell viability assay. b Apoptosis of cells was determined using flow cytometry after staining with Annexin V-PE/7-AAD. The plots are from a representative measurement and the data were expressed as mean ± SD in three different assays (n = 3). ***P < 0.001
ER stress-mediated apoptosis induced by GRA3Wh6 in N2a cells
Our previous studies have identified certain effector proteins that can induce programmed cell death in mouse N2a and human choriocarcinoma JEG-3 cells via ER stress signaling pathways [10, 11, 20]. Here, we sought to determine whether the dense granule effector protein GRA3 activated ER stress-induced cell death. N2a cells were transfected with either pEGFP or pEGFP-GRA3Wh6 (GRA3Wh6), and the expression levels of ER stress-related proteins and apoptosis-associated proteins were assessed. Immunoblotting showed significantly increased ER stress-associated proteins, such as GRP78, an ER-associated molecular chaperone (P = 0.0002), and p-PERK (P = 0.0036) in GRA3Wh6-transfected cells compared with pEGFP-transfected cells. Consistent with the results obtained from ER stress-associated proteins, cells transfected with GRA3Wh6 showed elevated levels of apoptosis-mediated proteins such as C/EBP homologous protein (CHOP) (P = 0.0403), cleaved caspase-12 (P = 0.0010), and cleaved caspase-3 (P = 0.0196) compared with N2a cells transfected with pEGFP (Fig. 4). These results suggest that GRA3 of the Wh6 strain could induce ER stress-mediated apoptosis in neural cells.
Expression of apoptosis-associated proteins and Endoplasmic reticulum stress (ERS) proteins induced by GRA3wh6 N2a cells were transfected with either pEGFP or pEGFP-GRA3Wh6 (GRA3Wh6) for a 24 h period. The expression levels of ER stress- and apoptosis-associated proteins were then determined by immunoblotting. Mock-transfected cells served as the negative control. The represented values were normalized and expressed relative to β-actin levels. CHOP C/EBP homologous protein, GRP78 78-kDa glucose-regulated protein, PERK PKR-like ER kinase, P-PERK phosphorylated PERK. Data were expressed as mean ± SD on three different assays (n = 3). *P < 0.05, **P < 0.01, ***P < 0.001
Toxoplasma GRA3 elevated the expression of ER stress- and apoptosis-associated genes
Following immunoblotting, we performed RT-qPCR to further compare the mRNA expression levels of the associated apoptosis and ER stress genes between pEGFP or pEGFP-GRA3Wh6 (GRA3Wh6)-transfected N2a cells. The results showed that GRA3Wh6-transfected cells had elevated mRNA levels of GRP78 (t(2) = 2.800, P = 0.0488), PERK (t(2) = 7.186, P = 0.0020), CHOP (t(2) = 7.052, P = 0.0021), caspase-12 (t(2) = 21.090, P = 0.0003) and caspase-3 (t(2) = 4.066, P = 0.0153) compared with N2a cells transfected with pEGFP (Fig. 5).
Transcription levels of apoptosis-associated genes induced by GRA3wh6. N2a cells were transfected with either pEGFP or pEGFP-GRA3Wh6 (GRA3Wh6) plasmid for a 24 h period. mRNA expressions of the associated apoptosis and ER stress genes were measured using RT-qPCR. The represented values were normalized and expressed relative to β-actin levels. GRP78 78-kDa glucose-regulated protein; CHOP, C/EBP homologous protein, PERK PKR-like ER kinase. Data were expressed as mean ± SD on three different assays (n = 3). *P < 0.05, **P < 0.01, ***P < 0.001
Attenuation of GRA3-induced N2a cell apoptosis following pretreatment with GSK2656157 and Z-ATAD-FMK
Cell viability and apoptosis were analyzed in GRA3Wh6-transfected N2a cells pretreated with GSK2656157 (PERK inhibitor, 4 μM, 1.5 h) and Z-ATAD-FMK (caspase-12 inhibitor, 5 μM, 6 h). Mock-transfected N2a cells showed 90.6% viability and an 8.4% apoptosis rate. pEGFP-transfected cells showed 78.0% viability and 12.3% apoptosis. Treatment with tunicamycin decreased N2a cell viability to 50.1% and increased the apoptosis rate to 61%. N2a cells transfected with GRA3Wh6 significantly reduced cell viability (P < 0.0001, 62.3% vs 78.0%) and increased cell apoptosis (P < 0.0001, 37.7% vs 12.3%) when compared with N2a cells transfected with pEGFP. Our results showed that pretreatment with GSK2656157 and Z-ATAD-FMK significantly increased cell viability (P < 0.0001 and P < 0.0001) and significantly decreased apoptosis (P < 0.001 and P < 0.0001) in GRA3Wh6-transfected cells (Fig. 6a, b). Furthermore, we analyzed the related proteins expressions of ER stress-associated apoptosis in GRA3Wh6-transfected cells following pretreatment with GSK2656157 and Z-ATAD-FMK. Immunoblotting results revealed that GRA3 increased the expression of cleaved caspase-12 (t(2) = 5.236, P = 0.0110), cleaved caspase-3 (P = 0.0008), p-PERK (P < 0.0001) and CHOP (P = 0.0024) when compared with cells transfected with pEGFP. Compared with GRA3Wh6-transfected cells, the expression levels of p-PERK (P = 0.0008), cleaved caspase-12 (P = 0.0034), cleaved caspase-3 (P = 0.0003) and CHOP (P = 0.0051) were significantly decreased in GRA3Wh6-transfected cells pretreated with GSK2656157. Similarly, the protein expression levels of cleaved caspase-12 (P = 0.0169) and cleaved caspase-3 (P = 0.0284) were significantly decreased in Z-ATAD-FMK-pretreated GRA3Wh6-transfected N2a cells (Fig. 6c). Collectively, these results demonstrate that GRA3Wh6 induces ER stress-associated apoptosis via the PERK pathway.
Effects of PERK and caspase-12 inhibitors on loss of cell viability and apoptosis in pEGFP-GRA3wh6 transfected N2a cells. N2a cells were treated with or without GSK2656157 (4 μM) and Z-ATAD-FMK (ZAF, 5 μM) for 1.5 h and 6 h, respectively, and transfected with either pEGFP or pEGFP-GRA3Wh6 (GRA3Wh6) for 24 h. a Cell viability was measured using the trypan blue staining cell viability assay. b Apoptosis of cells was determined using flow cytometry after staining with Annexin V-PE/7-AAD. The plots are from a representative measurement and the data were expressed as mean ± SD on three different assays (n = 3). c The protein expression levels of ER stress- and apoptosis-related proteins were determined by immunoblotting. The represented values were normalized and expressed relative to β-actin levels. The data were expressed as mean ± SD on three different assays (n = 3). *P < 0.05, **P < 0.01, ***P < 0.001, and ns, not statistically significant. GRA3Wh6 + GSK2656157 represents N2a cells pretreated with GSK2656157, followed by transfection with GRA3Wh6 plasmid. GRA3Wh6 + ZAF represents N2a cells pretreated with ZAF, followed by transfection with GRA3Wh6 plasmid
Discussion
Toxoplasmosis is known to be one of the most common parasitic infections that infect warm-blooded animals, including humans. Nearly a third of the human population have been chronically infected with T. gondii [25]. T. gondii can infect neural cells, where it forms cysts that remain for a lifetime in the host, causing chronic subclinical neuroinflammation [3, 26].
During cell invasion, secretory proteins such as dense granule proteins and rhoptry proteins (ROPs) are released into the host nucleus and cause considerable harm [27]. Several studies have explored how parasite proteins such as ROP16, ROP18, GRA15, and GRA60 contribute to T. gondii virulence either by activating or subverting host defense mechanisms [10, 20, 28]. Craver and Knoll sought to investigate the importance of GRA3 in the virulence of T. gondii [23]. To achieve this, they created a GRA3-deficient type II parasite to begin a functional characterization of the GRA3 protein. The GRA3 locus was disrupted by homologous replacement with a chloramphenicol resistance gene in a type II strain. Both in vitro and in vivo tests were performed to assess parasite growth and bradyzoite development and to assess the function of T. gondii GRA3 during infection. The result revealed no differences in the cell culture growth rate or bradyzoite formation between wild-type and ∆GRA3. Moreover, this study showed that the GRA3 monoclonal antibody derived after an RH infection does not react with the type II GRA3 protein because of a single amino acid change, indicating a difference in antigenicity between the two proteins, which is consistent with our result.
An important finding from the present study was that the GRA3 expression levels in avirulent type II ME49 and Chinese 1 Wh6 strains were significantly higher (P < 0.05 and P < 0.001, respectively) than those in the virulent type I RH and Chinese I Wh3 strains (Fig. 1). This was consistent with our previous study, which revealed that the expression level of GRA3 in the Wh6 strain was significantly higher than that in the RH and Wh3 virulent strains [29, 30].
The ER is responsible for the production of cellular organic molecules, including proteins, sterols, carbohydrates, and lipids [31,32,33,34]. Its role in protein folding is critical for cell survival. Cellular disturbances such as infections and reactive oxygen species can interfere with the normal functions of the ER. These cellular disturbances cause ER stress [35]. To alleviate this stress, the ER-localized transmembrane signaling proteins, i.e., inositol-requiring protein 1 (IRE1)-α, PERK, and activating transcription factor 6 (ATF6), activate UPR to restore cellular homeostasis [14, 15]. However, an unremitting UPR can activate UPR-mediated inflammatory and apoptotic pathways, resulting in cell death [16, 17]. We, for the first time in this study, demonstrate that the ER-Toxoplasma GRA3 interaction activates downstream apoptotic cascades in T. gondii-infected mouse N2a cells via the ER stress pathway. Therefore, our results indicate that GRA3 triggers neuronal apoptosis, which is not beneficial to the survival and dissemination of the parasites. The high expression of GRA3 in the less virulent strain might contribute to its lower virulence. Consistently, the low expression of GRA3 in the highly virulent strain helps it avoid neuronal apoptosis as much as possible, which helps its parasitism and widespread dissemination and may be one of the reasons for its strong virulence.
The protein folding function of the ER requires the presence of Ca2+-dependent ER molecular chaperone proteins. GRP78, commonly known as Bip, is one of the most-studied ER chaperone proteins [36]. Aside from being critical for protein quality control and thus sensing and targeting misfolded and/or unfolded proteins for degradation, GRP78 controls the activation of ER-stress transducers and acts as an ER stress sensor [37]. Within the cell, GRP78 levels are kept relatively low; however, they are upregulated in response to stressors that alter ER and Ca2+ homeostasis [38]. In our study, we demonstrated that T. gondii GRA3Wh6 induced ER stress in N2a, which significantly increased GRP78 mRNA and protein levels after a 24 h transfection with GRA3Wh6 (Figs. 4, 5). This observation is similar to our previous study, which involved the transfection of carcinoma JEG-3 cells with Toxoplasma GRA15II. In that study, pEGFP-GRA15II increased the expression levels of GRP78. This suggests that the ER-GRA3 interaction induces ER stress, which in turn upregulates GRP78 expression levels in an attempt to restore homeostasis.
Caspase-12 plays a crucial role in ER stress-mediated cell death. Under ER stress conditions, pro-caspase-12 is cleaved, and the activated forms accumulate (Nakagawa et al.). Here, GRA3Wh6 was found to cleave pro-caspase-12 into active caspase-12, accelerating apoptosis. Having demonstrated that GRA3Wh6-induced ER stress activates caspase-12, we next examined the downstream targets of GRA3Wh6-induced apoptosis following caspase-12 activation. We observed that the activation of caspase-12 resulted in the activation of caspase-3, as demonstrated by the increased cleaved caspase-3 expression in GRA3Wh6-transfected mouse N2a cells. Our results showed that GRA3Wh6 induced the activation of caspase-12, which contributes to the pathogenesis of encephalitis during T. gondii infection. Moreover, pretreatment of N2a cells with the caspase-12 inhibitor Z-ATAD-FMK significantly decreased cleaved caspase-12 and cleaved caspase-3 protein expression levels; consequently, Z-ATAD-FMK downregulated apoptosis in GRA3Wh6 N2a cells.
As an important initiator of the unfolded protein response (UPR), PERK undergoes dimerization and autophosphorylation upon dissociation from GRP78. The kinase domain is then activated by phosphorylation of PERK, which then targets substrates such as eIF2a to activate the cascade [39]. The PERK signaling pathway is activated in response to excessive amounts of misfolded proteins in the ER and temporarily blocks protein translation, which results in neuronal cell death [40, 41]. Our results showed that GRA3Wh6 significantly activated ER stress and UPR, as observed by the significantly increased levels of phosphorylated PERK proteins following immunoblotting. Similarly, qPCR results showed that the mRNA levels of PERK were elevated in GRA3Wh6-infected N2a cells. Consistent with previous findings, our immunoblotting results showed that pretreatment of N2a cells with GSK2656157, a PERK inhibitor, significantly suppressed phosphorylated PERK expression. Trypan blue staining cell viability and Annexin V-PE/7-AAD apoptosis assays revealed that N2a cells that were pretreated with GSK26561157 also suppressed neuronal cell death 24 h after GRA3Wh6 transfection. Furthermore, inhibition of PERK downregulated CHOP, cleaved caspase-12, and cleaved caspase-3 expression. GSK2606414, a PERK inhibitor, was shown to have neuroprotective effects by rescuing the loss of dendritic development and number of synapses in neurons following traumatic brain injury and decreasing the expression of downstream targets such as phospho-eIF2a, ATF4, and CHOP [42, 43]. Therefore, initiation of UPR by a signal through the PERK pathway appears to play a crucial role in GRA3-mediated ER stress apoptosis.
CHOP plays a pathological role in ER stress-related diseases. During unremitting UPR, activation of PERK results in the phosphorylation of the eukaryotic translation initiation factor (eIF2), resulting in a general translational block. However, ATF4 (activating transcription factor 4) is translated, activating downstream targets such as C/EBP homologous protein (CHOP) [44]. PERK-ATF4-CHOP pathway activation during prolonged UPR induces apoptosis [45]. Our results showed that the mRNA and protein expression levels of CHOP in N2a cells were significantly increased 24 h after T. gondii GRA3Wh6 transfection, which translated into increased N2a cell death, as observed in cell viability and cell apoptosis flow cytometry assays. This further indicates that GRA3Wh6 induces ER stress and activates the PERK-ATF4-CHOP signaling pathway to induce apoptosis in neuronal cells.
Conclusion
In conclusion, we have come a long way in our understanding of this protozoan parasite and its interaction with host cells. Our study highlights the mechanism by which dense granule protein (GRA3) increases the virulence of T. gondii. GRA3Wh6 induces neuronal apoptosis via the endoplasmic reticulum stress-mediated apoptosis pathway. This study provides further understanding of the mechanisms by which T. gondii causes neuropathology.
Availability of data and materials
Data are available from the corresponding authors upon reasonable request.
Abbreviations
- GRA3:
-
Dense granule protein 3
- CAML:
-
Calcium modulating cyclophilin ligand
- ER:
-
Endoplasmic reticulum
- UPR:
-
Unfolded protein response
- HIV:
-
Human immunodeficiency virus
- N2a:
-
Neuro2a
- ROP:
-
Rhoptry protein
- cDNA:
-
Complementary DNA
- mRNA:
-
Messenger RNA
- DNA:
-
Deoxyribonucleic acid
- RNA:
-
Ribonucleic acid
- GRP78:
-
Glucose regulated protein 78
- PERK:
-
Protein kinase R (PKR)-like ER kinase
- CHOP:
-
C/EBP-homologous protein
- SDS–PAGE:
-
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis
References
Meng M, Zhou A, Lu G, Wang L, Zhao G, Han Y, et al. DNA prime and peptide boost immunization protocol encoding the Toxoplasma gondii GRA4 induces strong protective immunity in BALB/c mice. BMC Infect Dis. 2013;13:494.
Jones JL, Parise ME, Fiore AE. Neglected parasitic infections in the United States: toxoplasmosis. Am J Trop Med Hyg. 2014;90:794–9.
Parlog A, Schluter D, Dunay IR. Toxoplasma gondii-induced neuronal alterations. Parasite Immunol. 2015;37:159–70.
Robben J, Hertveldt K, Bosmans E, Volckaert G. Selection and identification of dense granule antigen GRA3 by Toxoplasma gondii whole genome phage display. J Biol Chem. 2002;277:17544–7.
Niedelman W, Gold DA, Rosowski EE, Sprokholt JK, Lim D, Farid Arenas A, et al. The rhoptry proteins ROP18 and ROP5 mediate Toxoplasma gondii evasion of the murine, but not the human, interferon-gamma response. PLoS Pathog. 2012;8:e1002784.
Dubey JP. Toxoplasmosis of animals and humans. Boca Raton: CRC Press; 2016.
Shwab EK, Saraf P, Zhu X-Q, Zhou D-H, McFerrin BM, Ajzenberg D, et al. Human impact on the diversity and virulence of the ubiquitous zoonotic parasite Toxoplasma gondii. Proc Natl Acad Sci. 2018;115:E6956–63.
Martin S. Congenital toxoplasmosis. Neonatal Netw. 2001;20:23–30.
Petersen E. Toxoplasmosis. Semin Fetal Neonatal Med. 2007;12:214–23.
Wan L, Gong L, Wang W, An R, Zheng M, Jiang Z, et al. T. gondii rhoptry protein ROP18 induces apoptosis of neural cells via endoplasmic reticulum stress pathway. Parasit Vectors. 2015;8:554.
An R, Tang Y, Chen L, Cai H, Lai D-H, Liu K, et al. Encephalitis is mediated by ROP18 of Toxoplasma gondii, a severe pathogen in AIDS patients. Proc Natl Acad Sci USA. 2018;115:E5344–52.
Inácio P, Zuzarte-Luís V, Ruivo MT, Falkard B, Nagaraj N, Rooijers K, et al. Parasite-induced ER stress response in hepatocytes facilitates plasmodium liver stage infection. EMBO Rep. 2015;16:955–64.
Hetz C, Mollereau B. Disturbance of endoplasmic reticulum proteostasis in neurodegenerative diseases. Nat Rev Neurosci. 2014;15:233–49.
Liu CY, Kaufman RJ. The unfolded protein response. J Cell Sci. 2003;116:1861–2.
Bravo R, Parra V, Gatica D, Rodriguez AE, Torrealba N, Paredes F, et al. Endoplasmic reticulum and the unfolded protein response: dynamics and metabolic integration. Int Rev Cell Mol Biol. 2013;301:215–90.
Sprenkle NT, Sims SG, Sánchez CL, Meares GP. Endoplasmic reticulum stress and inflammation in the central nervous system. Mol Neurodegener. 2017;12:42.
Bellezza I, Grottelli S, Mierla AL, Cacciatore I, Fornasari E, Roscini L, et al. Neuroinflammation and endoplasmic reticulum stress are coregulated by cyclo (His-Pro) to prevent LPS neurotoxicity. Int J Biochem Cell Biol. 2014;51:159–69.
Zhou J, Gan X, Wang Y, Zhang X, Ding X, Chen L, et al. Toxoplasma gondii prevalent in China induce weaker apoptosis of neural stem cells C17.2 via endoplasmic reticulum stress (ERS) signaling pathways. Parasites & Vectors. 2015;8:73.
Wang T, Zhou J, Gan X, Wang H, Ding X, Chen L, et al. Toxoplasma gondii induce apoptosis of neural stem cells via endoplasmic reticulum stress pathway. Parasitology. 2014;141:988–95.
Wei W, Zhang F, Chen H, Tang Y, **ng T, Luo Q, et al. Toxoplasma gondii dense granule protein 15 induces apoptosis in choriocarcinoma JEG-3 cells through endoplasmic reticulum stress. Parasit Vectors. 2018;11:251.
Kim JY, Ahn HJ, Ryu KJ, Nam HW. Interaction between parasitophorous vacuolar membrane-associated GRA3 and calcium modulating ligand of host cell endoplasmic reticulum in the parasitism of Toxoplasma gondii. Korean J Parasitol. 2008;46:209–16.
Holloway MP, Bram RJ. Co-localization of calcium-modulating cyclophilin ligand with intracellular calcium pools. J Biol Chem. 1998;273:16346–50.
Craver MP, Knoll LJ. Increased efficiency of homologous recombination in Toxoplasma gondii dense granule protein 3 demonstrates that GRA3 is not necessary in cell culture but does contribute to virulence. Mol Biochem Parasitol. 2007;153:149–57.
Cheng W, Wang C, Xu T, Liu F, Pappoe F, Luo Q, et al. Genoty** of polymorphic effectors of Toxoplasma gondii isolates from China. Parasit Vectors. 2017;10:580.
Halonen SK, Weiss LM. Toxoplasmosis. Handbook of clinical neurology. Amsterdam: Elsevier; 2013.
Weiss LM, Dubey JP. Toxoplasmosis: a history of clinical observations. Int J Parasitol. 2009;39:895–901.
Laliberté J, Carruthers VB. Host cell manipulation by the human pathogen Toxoplasma gondii. Cell Mol Life Sci. 2008;65:1900–15.
Nyonda MA, Hammoudi PM, Ye S, Maire J, Marq JB, Yamamoto M, et al. Toxoplasma gondii GRA60 is an effector protein that modulates host cell autonomous immunity and contributes to virulence. Cell Microbiol. 2021;23:e13278.
Cheng W, Liu F, Li M, Hu X, Chen H, Pappoe F, et al. Variation detection based on next-generation sequencing of type Chinese 1 strains of Toxoplasma gondii with different virulence from China. BMC Genomics. 2015;16:888.
Li M, Mo X-W, Wang L, Chen H, Luo Q-L, Wen H-Q, et al. Phylogeny and virulence divergency analyses of Toxoplasma gondii isolates from China. Parasit Vectors. 2014;7:133.
Reid DW, Nicchitta CV. Diversity and selectivity in mRNA translation on the endoplasmic reticulum. Nat Rev Mol Cell Biol. 2015;16:221–31.
Rapoport TA. Protein translocation across the eukaryotic endoplasmic reticulum and bacterial plasma membranes. Nature. 2007;450:663–9.
Braakman I, Hebert DN. Protein folding in the endoplasmic reticulum. Cold Spring Harb Perspect Biol. 2013;5:a013201.
Westrate LM, Lee JE, Prinz WA, Voeltz GK. Form follows function: the importance of endoplasmic reticulum shape. Annu Rev Biochem. 2015;84:791–811.
Zhang K, Kaufman RJ. From endoplasmic-reticulum stress to the inflammatory response. Nature. 2008;454:455–62.
Adams CJ, Kopp MC, Larburu N, Nowak PR, Ali MMU. Structure and molecular mechanism of ER stress signaling by the unfolded protein response signal activator IRE1. Front Mol Biosci. 2019. https://doi.org/10.3389/fmolb.2019.00011.
Wang M, Wey S, Zhang Y, Ye R, Lee AS. Role of the unfolded protein response regulator GRP78/BiP in development, cancer, and neurological disorders. Antioxid Redox Signal. 2009;11:2307–16.
Casas C. GRP78 at the centre of the stage in cancer and neuroprotection. Front Neurosci. 2017. https://doi.org/10.3389/fnins.2017.00177.
Bell MC, Meier SE, Ingram AL, Abisambra JF. PERK-opathies: an endoplasmic reticulum stress mechanism underlying neurodegeneration. Curr Alzheimer Res. 2016;13:150–63.
Radford H, Moreno JA, Verity N, Halliday M, Mallucci GR. PERK inhibition prevents tau-mediated neurodegeneration in a mouse model of frontotemporal dementia. Acta Neuropathol. 2015;130:633–42.
Meng C, Zhang J, Dang B, Li H, Shen H, Li X, et al. PERK pathway activation promotes intracerebral hemorrhage induced secondary brain injury by inducing neuronal apoptosis both in vivo and in vitro. Front Neurosci. 2018. https://doi.org/10.3389/fnins.2018.00111.
Sen T, Gupta R, Kaiser H, Sen N. Activation of PERK elicits memory impairment through inactivation of CREB and downregulation of PSD95 after traumatic brain injury. J Neurosci. 2017;37:5900–11.
Atkins C, Liu Q, Minthorn E, Zhang SY, Figueroa DJ, Moss K, et al. Characterization of a novel PERK kinase inhibitor with antitumor and antiangiogenic activity. Can Res. 2013;73:1993–2002.
Ron D, Walter P. Signal integration in the endoplasmic reticulum unfolded protein response. Nat Rev Mol Cell Biol. 2007;8:519–29.
Walter F, Schmid J, Düssmann H, Concannon CG, Prehn JHM. Imaging of single cell responses to ER stress indicates that the relative dynamics of IRE1/XBP1 and PERK/ATF4 signalling rather than a switch between signalling branches determine cell survival. Cell Death Differ. 2015;22:1502–16.
Acknowledgements
The authors would like to thank the members of our laboratories for their thoughtful discussions.
Funding
This study was financially supported by the National Natural Science Foundation of China (No. 82072300, No. 81902084, and No. 81871674).
Author information
Authors and Affiliations
Contributions
JD, JS: conceived and designed the experiments. CO: performed experiments, data analysis, writing—review & editing. MW, JW, FL: performed the experiments and data analysis. YC: review & editing. RA, HC, QL, LY: formal analysis. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
There are no competing interests with regard to the work reported in this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Obed, C., Wu, M., Chen, Y. et al. Toxoplasma gondii dense granule protein 3 promotes endoplasmic reticulum stress-induced apoptosis by activating the PERK pathway. Parasites Vectors 15, 276 (2022). https://doi.org/10.1186/s13071-022-05394-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13071-022-05394-5