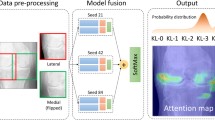
Abstract
Background
For knee osteoarthritis, the commonly used radiology severity criteria Kellgren–Lawrence lead to variability among surgeons. Most existing diagnosis models require preprocessed radiographs and specific equipment.
Methods
All enrolled patients diagnosed with KOA who met the criteria were obtained from **** Hospital. This study included 2579 images shot from posterior–anterior X-rays of 2,378 patients. We used RefineDet to train and validate this deep learning-based diagnostic model. After develo** the model, 823 images of 697 patients were enrolled as the test set. The whole test set was assessed by up to 5 surgeons and this diagnostic model. To evaluate the model’s performance we compared the results of the model with the KOA severity diagnoses of surgeons based on K-L scales.
Results
Compared to the diagnoses of surgeons, the model achieved an overall accuracy of 0.977. Its sensitivity (recall) for K-L 0 to 4 was 1.0, 0.972, 0.979, 0.983 and 0.989, respectively; for these diagnoses, the specificity of this model was 0.992, 0.997, 0.994, 0.991 and 0.995. The precision and F1-score were 0.5 and 0.667 for K-L 0, 0.914 and 0.930 for K-L 1, 0.978 and 0.971 for K-L 2, 0.981 and 0.974 for K-L 3, and 0.988 and 0.985 for K-L 4, respectively. All K-L scales perform AUC > 0.90. The quadratic weighted Kappa coefficient between the diagnostic model and surgeons was 0.815 (P < 0.01, 95% CI 0.727–0.903). The performance of the model is comparable to the clinical diagnosis of KOA. This model improved the efficiency and avoided cumbersome image preprocessing.
Conclusion
The deep learning-based diagnostic model can be used to assess the severity of KOA in portable devices according to the Kellgren–Lawrence scale. On the premise of improving diagnostic efficiency, the results are highly reliable and reproducible.
Similar content being viewed by others
Introduction
Knee osteoarthritis (KOA) is a degenerative joint disease with a prevalence ranging from 4 to 12% [1,2,3]. Currently, for orthopaedic surgeons, knee weight-bearing standing X-ray radiographs as a standard method for evaluating KOA remain the most common radiology examination method due to their safety, popularity and low cost [4,5,6]. Currently, accurate KOA diagnosis and assessment are highly based on radiographic evidence [7, 8].
Currently, the Kellgren–Lawrence scale (K-L) is most commonly used to diagnose and determine the severity of KOA based on joint space narrowing, osteophytes, sclerosis, and definite bony deformities on X-rays in radiology examinations [9, 10]. However, the classification criteria of the K-L scales are subjective [11]. In clinical use, different doctors or the same doctor at different times may often obtain similar results rather than identical results on the same X-ray. Many clinical studies involving KOA diagnosis have ensured reliability by increasing the number of repeated diagnoses [12,13,14,15,16]. Moreover, the total numbers of X-ray examinations are much higher in large hospitals, which is a heavy burden on radiologists and surgeons. Therefore, many rapid diagnosis and assessment models have been developed in collaboration with image analysis. The models can identify images via digital processing techniques to make the artificial intelligence process more accurate and cost-effective [13]. The technology includes knee joint recognition and image processing based on deep learning [14]. Swiecicki et al. [15] developed a diagnostic model based on the two-stage Faster R-CNN model to assess the severity of KOA from both posterior–anterior (PA) and lateral (LAT) views. Tiulpin et al. [16] also developed a diagnostic model based on the ResNet34 model to detect KOA from original PA views of knees. Similarly, Norman et al. [17] developed a KOA diagnostic model based on DenseNet, which uses the feature more effectively in deep technology. Current studies show that the existing diagnostic models can achieve satisfactory accuracy [15,16,17]. However, those models rely on preprocessed, highly optimized digital images in specific software and hardware, which may not be feasible in most clinical scenarios and affects the actual use value in some ways [18,19,20,32]. Second, although the study shows that this model has good reliability and reproducibility in radiology assessment, the specific diagnosis still needs other evidence, such as the patient’s clinical manifestations, laboratory tests, and other imaging findings [12]. Third, in the process of use, due to the restrictions of the algorithm, there may be some photos in which the entire knee joints cannot be identified, and the diagnostic model does not display a diagnosis if the algorithm detects implants or no object. Therefore, there may be a subset that is not identified as implants or knee joints, which requires rephotographing or further diagnosis by surgeons. Fifth, compared to similar studies, our sample size is relatively small and from a single hospital, and outcomes from a larger cohort and multiple countries may be different. Sixth, we propose the deep-learning-based RefineDet for object detection and classification, but the outcomes may be different based on other algorithms.
Conclusion
The deep learning-based diagnostic model can be used to assess the severity of KOA in portable devices. On the premise of improving the diagnostic efficiency, the results are highly reliable and reproducible.
Availability of data and materials
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- KOA:
-
Knee osteoarthritis
- K-L:
-
Kellgren–Lawrence
- PA X-ray:
-
Posterior–anterior view standing bearing X-ray
- AUC:
-
Area under curve
- LAT:
-
Lateral
- ARM:
-
Anchor refinement module
- ODM:
-
Object detection module
- TCB:
-
Transfer connection block
- IoU:
-
Intersection over Union
- TKA:
-
Total knee arthroplasty
- UKA:
-
Unicompartmental knee arthroplasty
- I-F:
-
Internal fixation
- E–F:
-
External fixation
- PFA:
-
Patellofemoral arthroplasty
- CHD:
-
Coronary heart disease
References
Mandl LA. Osteoarthritis year in review 2018: clinical. Osteoarthr Cartil. 2019;27:359–64.
Batushansky A, Zhu S, Komaravolu RK, South S, Mehta-D’souza P, Griffin TM. Fundamentals of OA. An initiative of osteoarthritis and cartilage. obesity and metabolic factors in OA. Osteoarthr Cartil. 2022;30:501–15.
Zhang W, Moskowitz RW, Nuki G, Abramson S, Altman RD, Arden N, et al. OARSI recommendations for the management of hip and knee osteoarthritis, Part II: OARSI evidence-based, expert consensus guidelines. Osteoarthr Cartil. 2008;16:137–62.
Conaghan PG, Porcheret M, Kingsbury SR, Gammon A, Soni A, Hurley M, et al. Impact and therapy of osteoarthritis: the arthritis care OA Nation 2012 survey. Clin Rheumatol. 2015;34:1581–8.
Disease-modifying treatments for osteoarthritis (DMOADs) of the knee and hip: lessons learned from failures and opportunities for the future - PubMed [Internet]. [cited 2022 Nov 6]. Available from: https://pubmed.ncbi.nlm.nih.gov/27492463/
Cross M, Smith E, Hoy D, Nolte S, Ackerman I, Fransen M, et al. The global burden of hip and knee osteoarthritis: estimates from the global burden of disease 2010 study. Ann Rheum Dis. 2014;73:1323–30.
Lee LS, Chan PK, Wen C, Fung WC, Cheung A, Chan VWK, et al. Artificial intelligence in diagnosis of knee osteoarthritis and prediction of arthroplasty outcomes: a review. Arthroplasty. 2022;4:16.
Oei EHG, Hirvasniemi J, van Zadelhoff TA, van der Heijden RA. Osteoarthritis year in review 2021: imaging. Osteoarthr Cartil. 2022;30:226–36.
Kellgren JH, Lawrence JS. Radiological assessment of osteo-arthrosis. Ann Rheum Dis. 1957;16:494–502.
Harlaar J, Macri EM, Wesseling M. Osteoarthritis year in review 2021: mechanics. Osteoarthritis Cartilage. 2022;30:663–70.
Wright RW, MARS Group. Osteoarthritis Classification Scales: Interobserver Reliability and Arthroscopic Correlation. J Bone Joint Surg Am. 2014;96:1145–51.
Culvenor AG, Engen CN, Øiestad BE, Engebretsen L, Risberg MA. Defining the presence of radiographic knee osteoarthritis: a comparison between the Kellgren and Lawrence system and OARSI atlas criteria. Knee Surg Sports Traumatol Arthrosc. 2015;23:3532–9.
Nich C, Behr J, Crenn V, Normand N, Mouchère H, d’Assignies G. Applications of artificial intelligence and machine learning for the hip and knee surgeon: current state and implications for the future. Int Orthop. 2022;46:937–44.
Kurmis AP, Ianunzio JR. Artificial intelligence in orthopedic surgery: evolution, current state and future directions. Arthroplasty. 2022;4:9.
Swiecicki A, Li N, O’Donnell J, Said N, Yang J, Mather RC, et al. Deep learning-based algorithm for assessment of knee osteoarthritis severity in radiographs matches performance of radiologists. Comput Biol Med. 2021;133:104334.
Tiulpin A, Thevenot J, Rahtu E, Lehenkari P, Saarakkala S. Automatic knee osteoarthritis diagnosis from plain radiographs: a deep learning-based approach. Sci Rep. 2018;8:1727.
Norman B, Pedoia V, Noworolski A, Link TM, Majumdar S. Applying densely connected convolutional neural networks for staging osteoarthritis severity from plain radiographs. J Digit Imaging. 2019;32:471–7.
Mun SK, Wong KH, Lo S-CB, Li Y, Bayarsaikhan S. Artificial intelligence for the future radiology diagnostic service. Front Mol Biosci. 2020;7:614258.
Purnomo G, Yeo SJ, Liow MHL. Artificial intelligence in arthroplasty. Arthroplasty. 2021;3:1–7.
Chang GH, Park LK, Le NA, Jhun RS, Surendran T, Lai J, et al. Subchondral bone length in knee osteoarthritis: a deep learning-derived imaging measure and its association with radiographic and clinical outcomes. Arthritis Rheumatol. 2021;73:2240–8.
Antony J, McGuinness K, Connor NEO, Moran K. Quantifying Radiographic Knee Osteoarthritis Severity using Deep Convolutional Neural Networks [Internet]. ar**v; 2016 [cited 2022 Nov 6]. Available from: http://arxiv.org/abs/1609.02469
Khan S, Azam B, Yao Y, Chen W. Deep collaborative network with alpha matte for precise knee tissue segmentation from MRI. Comput Methods Programs Biomed. 2022;222:106963.
Bayramoglu N, Nieminen MT, Saarakkala S. Automated detection of patellofemoral osteoarthritis from knee lateral view radiographs using deep learning: data from the Multicenter Osteoarthritis Study (MOST). Osteoarthr Cartil. 2021;29:1432–47.
Zhang Z, Huang C, Jiang Q, Zheng Y, Liu Y, Liu S, et al. Guidelines for the diagnosis and treatment of osteoarthritis in China (2019 edition). Ann Transl Med. 2020;8:1213.
Zhang S, Wen L, Bian X, Lei Z, Li SZ. Single-Shot Refinement Neural Network for Object Detection. 2018 IEEE/CVF Conference on Computer Vision and Pattern Recognition. 2018. p. 4203–12.
ImageNet Large Scale Visual Recognition Challenge | SpringerLink [Internet]. [cited 2022 Nov 6]. Available from: https://springer.longhoe.net/article/https://doi.org/10.1007/s11263-015-0816-y
Liu W, Anguelov D, Erhan D, Szegedy C, Reed S, Fu C-Y, et al. SSD: single shot multibox detector. In: Leibe B, Matas J, Sebe N, Welling M, editors., et al., Computer Vision—ECCV 2016. Cham: Springer International Publishing; 2016. p. 21–37.
Kingma DP, Ba J. Adam: A Method for Stochastic Optimization [Internet]. ar**v; 2017 [cited 2022 Nov 6]. Available from: http://arxiv.org/abs/1412.6980
De Raadt A, Warrens MJ, Bosker RJ, Kiers HAL. Kappa coefficients for missing data. Educ Psychol Meas. 2019;79:558–76.
Bayramoglu N, Tiulpin A, Hirvasniemi J, Nieminen MT, Saarakkala S. Adaptive segmentation of knee radiographs for selecting the optimal ROI in texture analysis. Osteoarthr Cartil. 2020;28:941–52.
Guan B, Liu F, Haj-Mirzaian A, Demehri S, Samsonov A, Neogi T, et al. Deep learning risk assessment models for predicting progression of radiographic medial joint space loss over a 48-MONTH follow-up period. Osteoarthr Cartil. 2020;28:428–37.
Katz JN, Arant KR, Loeser RF. Diagnosis and treatment of hip and knee osteoarthritis: a review. JAMA. 2021;325:568–78.
Funding
This work was supported by a grant from The National Natural Science Foundation of China (No. 81902250, 82072464) and General Hospital of the People’s Liberation Army (No. 2019MBD-042).
Author information
Authors and Affiliations
Contributions
YW, GZ, and JY designed the study. JY and MN followed up with the patients and collected the relevant data and images. YW and JY analyzed and interpreted the data. JY and QJ wrote the manuscript. JY, QJ and MN prepared figures. The authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This study was approved by the ethics committee of General Hospital of PLA. All patients signed the informed consent to participate in the study.
Consent for publication
The authors affirm that patients provided informed consent regarding publishing their data and images.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Yang, J., Ji, Q., Ni, M. et al. Automatic assessment of knee osteoarthritis severity in portable devices based on deep learning. J Orthop Surg Res 17, 540 (2022). https://doi.org/10.1186/s13018-022-03429-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13018-022-03429-2