Abstract
Background
Neck circumference, a proxy for upper-body subcutaneous fat, is a unique and pathogenic fat depot that confers additional metabolic risk. The purpose of present study was to determine whether neck circumference associates with nonalcoholic fatty liver disease (NAFLD) in postmenopausal women with normal body mass index.
Methods
A cross-sectional survey (n = 2492) and a 3.1-year follow-up investigation (n = 1354) were conducted among Chinese postmenopausal women with normal BMI (18.5 to < 25 kg/m2). Neck circumference was measured horizontally at the lower margin of the laryngeal prominence.
Results
In the cross-sectional analysis, large neck circumference was associated with the presence of NAFLD (odds ratio 2.28; 95% CI 1.74–2.98; highest tertile versus lowest tertile) after adjustment for confounding factors. Among 1354 subjects without the NAFLD at baseline, 429 (31.7%) incident NAFLD cases occurred at 3.1 years. Neck circumference was positively associated with triglycerides, homeostasis model assessment of insulin resistance, C-reactive protein, and negatively associated with high-density lipoprotein cholesterol and adiponectin. Individuals with large baseline neck circumference had a significantly higher risk of NAFLD than those with small neck circumference. The multivariable adjusted hazard ratio was 1.42 (95% CI 1.15–1.97; p for trend = 0.004) for the highest versus the lowest tertile of neck circumference, and was 1.22 (95% CI 1.10–1.41; p = 0.006) per 1-standard deviation increment in neck circumference.
Conclusions
Among postmenopausal women with normal BMI, relatively large neck circumference levels are associated with an increased risk of NAFLD.
Similar content being viewed by others
Introduction
Nonalcoholic fatty liver disease (NAFLD) is characterized by excessive fat accumulation in the liver with the absence of alcohol and other liver diseases. It is well established that adiposity is a major contributor to NAFLD. Data from epidemiological studies revealed that NAFLD occurrence in up to 70% of overweight adults worldwide [1] and in 90% of morbidly obese adults globally [2]. Nonetheless, a remarkable proportion of individuals having NAFLD with a relatively normal body mass index (BMI), a condition termed as ‘‘non-obese” NAFLD. The prevalence of NAFLD was around 10% in nonobese adults in Western countries, and about 8–19% of Asians with BMI less than 25 kg/ m2 are also found to have NAFLD [3]. Given this trend, a reliable predictor of NAFLD in nonobese people is essential for its prevention and management.
As we best known, the larger the BMI, the higher the metabolic risk. However, individuals within the same BMI status could have considerable differences in the amount and distribution of regional fat depots, leading to variable metabolic risks [4, 5]. Numerous researches highlight the metabolic risk conferred by specific patterns of fat distribution, particularly upper body adiposity [6]. Notably, several studies have recently shown that upper-body subcutaneous adipose tissue, estimated by neck circumference, is a unique fat depot that confer additional metabolic risks beyond generalized and abdominal adiposity [7, 8]. It is reported that neck circumference levels are significantly associated with hyperlipidemia, impaired glucose homeostasis, and insulin resistance, especially in women [7]. All of these supported the investigation of a possible association of neck circumference with NAFLD.
Due, in part, to a transfer from subcutaneous to abdominal visceral fat, women after menopause are prone to metabolic changes [9]. Such metabolic alterations are associated with increased NAFLD risk among normal BMI populations. However, studies that assess neck fat accumulation, as a proxy for upper-body subcutaneous fat, and its relationship with NAFLD among postmenopausal women with normal BMI are still lacking. In the current study, we evaluated the relationship between neck circumference and NAFLD among postmenopausal women with normal BMI.
Participants and methods
Study subjects
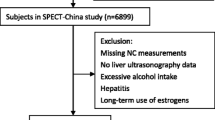
Participants were recruited from the China Cardiometabolic Disease and Cancer Cohort (4C) Study, a nationwide prospective cohort study investigating the associations of metabolic factors with specific clinical outcomes, including diabetes, cardiovascular disease, cancer, and all-cause mortality [10, 11]. The data presented in this study are based on the subsamples from the Chongming District in Shanghai, China. From May to November 2011, a total of 2765 postmenopausal women with normal BMI of Chinese origins were enrolled in the study. From June to December 2014, the subjects were invited for follow-up assessments. The cross-sectional survey included 2492 subjects, and the follow-up investigation included 1354 individuals without NAFLD at baseline (Fig. 1). Subjects with the following conditions were excluded from this study: virus hepatitis, autoimmune hepatitis, drug-induced liver disease, current drinkers, ex-drinkers, presence of tumor, biliary obstructive diseases, thyroid dysfunction, total parenteral nutrition, Wilson’s disease, severe renal insufficiency, significant hematologic disorders, and current treatment with systemic corticosteroids.
The study protocol was approved by the Ethics Committee of ** countries. Third, given the diagnosis of NAFLD was based on ultrasound imaging, NAFLD patients in our study were in at least a moderate stage of the disease. Therefore, in the present study, we were unable to determine the relationship between neck circumference and mild-stage NAFLD. Fourth, we did not assess the impact of excluded data on the relationship between neck circumference and incident NAFLD.
Conclusions
Large neck circumference was significantly associated with an increased risk of NAFLD among postmenopausal women with normal BMI. Measurement of neck circumference may provide a more complete understanding of NAFLD risk associated with variation in fat distribution among postmenopausal women with normal BMI.
Availability of data and materials
The datasets generated during and/or analyzed during the current study are not publicly available but are available from the corresponding author on reasonable request.
Abbreviations
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate transaminase
- BMI:
-
Body mass index
- CRP:
-
C-reactive protein
- DBP:
-
Diastolic blood pressure
- FPG:
-
Fasting plasma glucose
- GGT:
-
γ-Glutamyltransferase
- HbA1c :
-
Glycated hemoglobin
- HDL-C:
-
High-density lipoprotein cholesterol
- HOMA-IR:
-
Homeostasis model assessment-insulin resistance
- LDL-C:
-
Low-density lipoprotein cholesterol
- NAFLD:
-
Nonalcoholic fatty liver disease
- PPG:
-
Postprandial plasma glucose
- SBP:
-
Systolic blood pressure
- WC:
-
Waist circumference
References
Bellentani S, Saccoccio G, Masutti F, Crocè LS, Brandi G, Sasso F, Cristanini G, Tiribelli C. Prevalence of and risk factors for hepatic steatosis in Northern Italy. Ann Intern Med. 2000;132:112–7.
Silverman JF, O’Brien KF, Long S, Leggett N, Khazanie PG, Pories WJ, Norris HT, Caro JF. Liver pathology in morbidly obese patients with and without diabetes. Am J Gastroenterol. 1990;85:1349–55.
Fan J-G, Kim S-U. Wong VW-S: New trends on obesity and NAFLD in Asia. J Hepatol. 2017;67:862–73.
Tchkonia T, Thomou T, Zhu Y, Karagiannides I, Pothoulakis C, Jensen MD, Kirkland JL. Mechanisms and metabolic implications of regional differences among fat depots. Cell Metab. 2013;17:644–56.
Romero-Corral A, Somers VK, Sierra-Johnson J, Korenfeld Y, Boarin S, Korinek J, Jensen MD, Parati G, Lopez-Jimenez F. Normal weight obesity: a risk factor for cardiometabolic dysregulation and cardiovascular mortality. Eur Heart J. 2010;31:737–46.
Nielsen S, Guo Z, Johnson CM, Hensrud DD, Jensen MD. Splanchnic lipolysis in human obesity. J Clin Investig. 2004;113:1582–8.
Preis SR, Massaro JM, Hoffmann U, D’Agostino RB, Levy D, Robins SJ, Meigs JB, Vasan RS, O’Donnell CJ, Fox CS. Neck circumference as a novel measure of cardiometabolic risk: the Framingham Heart study. J Clin Endocrinol Metab. 2010;95:3701–10.
Zhou JY, Ge H, Zhu MF, Wang LJ, Chen L, Tan YZ, Chen YM, Zhu HL. Neck circumference as an independent predictive contributor to cardio-metabolic syndrome. Cardiovasc Diabetol. 2013;12:76.
JafariNasabian P, Inglis JE, Reilly W, Kelly OJ, Ilich JZ. Aging human body: changes in bone, muscle and body fat with consequent changes in nutrient intake. J Endocrinol. 2017;234:R37–51.
Lu J, He J, Li M, Tang X, Hu R, Shi L, Su Q, Peng K, Xu M, Xu Y, et al. Predictive value of fasting glucose, postload glucose, and hemoglobin a on risk of diabetes and complications in Chinese adults. Diabetes Care. 2019;42:1539–48.
Wang T, Lu J, Su Q, Chen Y, Bi Y, Mu Y, Chen L, Hu R, Tang X, Yu X, et al. Ideal cardiovascular health metrics and major cardiovascular events in patients with prediabetes and diabetes. JAMA Cardiology. 2019;4:874–83.
Chitturi S, Farrell GC, Hashimoto E, Saibara T, Lau GKK, Sollano JD. Non-alcoholic fatty liver disease in the Asia-Pacific region: definitions and overview of proposed guidelines. J Gastroenterol Hepatol. 2007;22:778–87.
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28:412–9.
Suzuki A, Abdelmalek MF. Nonalcoholic fatty liver disease in women. Womens Health (Lond Engl). 2009;5:191–203.
Fox CS, Massaro JM, Hoffmann U, Pou KM, Maurovich-Horvat P, Liu C-Y, Vasan RS, Murabito JM, Meigs JB, Cupples LA, et al. Abdominal visceral and subcutaneous adipose tissue compartments: association with metabolic risk factors in the Framingham Heart Study. Circulation. 2007;116:39–48.
Lovejoy JC, Champagne CM, de Jonge L, **e H, Smith SR. Increased visceral fat and decreased energy expenditure during the menopausal transition. Int J Obes. 2005;2008(32):949–58.
Gupte AA, Pownall HJ, Hamilton DJ. Estrogen: an emerging regulator of insulin action and mitochondrial function. Journal of Diabetes Research. 2015;2015:916585.
Browning JD, Szczepaniak LS, Dobbins R, Nuremberg P, Horton JD, Cohen JC, Grundy SM, Hobbs HH. Prevalence of hepatic steatosis in an urban population in the United States: impact of ethnicity. Hepatology. 2004;40:1387–95.
Barros RP, Gabbi C, Morani A, Warner M, Gustafsson JA. Participation of ERalpha and ERbeta in glucose homeostasis in skeletal muscle and white adipose tissue. Am J Physiol Endocrinol Metab. 2009;297:E124-133.
Riant E, Waget A, Cogo H, Arnal JF, Burcelin R, Gourdy P. Estrogens protect against high-fat diet-induced insulin resistance and glucose intolerance in mice. Endocrinology. 2009;150:2109–17.
Gupte AA, Pownall HJ, Hamilton DJ. Estrogen: an emerging regulator of insulin action and mitochondrial function. J Diabetes Res. 2015;2015:916585.
Wei JL, Leung JC-F, Loong TC-W, Wong GL-H, Yeung DK-W, Chan RS-M, Chan HL-Y, Chim AM-L, Woo J, Chu WC-W, Wong VW-S. Prevalence and severity of nonalcoholic fatty liver disease in non-obese patients: a population study using proton-magnetic resonance spectroscopy. Am J Gastroenterol. 2015;110(9):1306–14.
Liang J, Teng F, Li Y, Liu X, Zou C, Wang Y, Li H, Qi L. neck circumference and insulin resistance in Chinese adults: the Cardiometabolic Risk in Chinese (CRC) Study. Diabetes Care. 2013;36:e145-146.
Huang B-x, Zhu M-f, Wu T, Zhou J-y, Liu Y, Chen X-l, Zhou R-f, Wang L-J, Chen Y-m, Zhu H-l. Neck circumference, along with other anthropometric indices, has an independent and additional contribution in predicting fatty liver disease. PLoS ONE. 2015;10:e0118071.
Li Q, Wang N, Han B, Chen Y, Zhu C, Chen Y, **a F, Cang Z, Zhu C, Chen C, et al. Neck circumference as an independent indicator to non-alcoholic fatty liver disease in non-obese men. Nutr Metab. 2015;12:63.
Jensen MD, Haymond MW, Rizza RA, Cryer PE, Miles JM. Influence of body fat distribution on free fatty acid metabolism in obesity. J Clin Investig. 1989;83:1168–73.
Jensen MD. Role of body fat distribution and the metabolic complications of obesity. J Clin Endocrinol Metab. 2008;93:S57–63.
Hotamisligil GS. Inflammation, metaflammation and immunometabolic disorders. Nature. 2017;542:177–85.
Bergman RN, Ader M. Free fatty acids and pathogenesis of type 2 diabetes mellitus. Trends Endocrinol Metabol TEM. 2000;11:351–6.
Byrne CD, Targher G. NAFLD: a multisystem disease. J Hepatol. 2015;62:S47–64.
Hotamisligil GS. Endoplasmic reticulum stress and the inflammatory basis of metabolic disease. Cell. 2010;140:900–17.
Weisberg SP, McCann D, Desai M, Rosenbaum M, Leibel RL, Ferrante AW. Obesity is associated with macrophage accumulation in adipose tissue. J Clin Investig. 2003;112:1796–808.
Kershaw EE, Flier JS. Adipose tissue as an endocrine organ. J Clin Endocrinol Metab. 2004;89:2548–56.
Furukawa S, Fujita T, Shimabukuro M, Iwaki M, Yamada Y, Nakajima Y, Nakayama O, Makishima M, Matsuda M, Shimomura I. Increased oxidative stress in obesity and its impact on metabolic syndrome. J Clin Investig. 2004;114:1752–61.
Guilherme A, Virbasius JV, Puri V, Czech MP. Adipocyte dysfunctions linking obesity to insulin resistance and type 2 diabetes. Nat Rev Mol Cell Biol. 2008;9:367–77.
Cancello R, Henegar C, Viguerie N, Taleb S, Poitou C, Rouault C, Coupaye M, Pelloux V, Hugol D, Bouillot J-L, et al. Reduction of macrophage infiltration and chemoattractant gene expression changes in white adipose tissue of morbidly obese subjects after surgery-induced weight loss. Diabetes. 2005;54:2277–86.
Acknowledgements
The authors gratefully acknowledge the support of the study participants, study staff, and partner organizations participating in the baseline survey and follow-up investigation.
Funding
This work was supported by the National Natural Science Foundation of China (81970669, 81873565, 81670743, 81370953), the National Key Research and Development Plan ‘Precision Medicine Research’ (2017YFSF090203), the Shanghai Health System Outstanding Young Talents Training Program (XYQ2013098), the Shanghai Sailing Program (18YF1415800), the Shanghai Health and Family Planning Commission (21740173).
Author information
Authors and Affiliations
Contributions
JS, ZW, and WZ drafted the manuscript and performed the experiments. ZY, JF, QS, LQ, and GN conceived and designed the study. WZ, YN, XL, HZ, and NL recruited the subjects, processed samples, and contributed to the acquisition of data. JS and ZY analyzed the data. ZY revised the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The study protocol was approved by the Ethics Committee of **nhua Hospital Affiliated to Shanghai Jiao Tong University School of Medicine. Written informed consent was obtained from all participants before data collection.
Consent for publication
Not applicable.
Competing interests
No potential conflicts of interest relevant to this article were reported.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Supplementary Table S1
. Baseline characteristics of study participants.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Shi, J., Wang, Z., Zhang, W. et al. Neck circumference as an independent predictor for NAFLD among postmenopausal women with normal body mass index. Nutr Metab (Lond) 18, 30 (2021). https://doi.org/10.1186/s12986-021-00562-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12986-021-00562-3




