Abstract
Metformin is a well-known anti-diabetic drug that has been repurposed for several emerging applications, including as an anti-cancer agent. It boasts the distinct advantages of an excellent safety and tolerability profile and high cost-effectiveness at less than one US dollar per daily dose. Epidemiological evidence reveals that metformin reduces the risk of cancer and decreases cancer-related mortality in patients with diabetes; however, the exact mechanisms are not well understood. Energy metabolism may be central to the mechanism of action. Based on altering whole-body energy metabolism or cellular state, metformin’s modes of action can be divided into two broad, non-mutually exclusive categories: “direct effects”, which induce a direct effect on cancer cells, independent of blood glucose and insulin levels, and “indirect effects” that arise from systemic metabolic changes depending on blood glucose and insulin levels. In this review, we summarize an updated account of the current knowledge on metformin antitumor action, elaborate on the underlying mechanisms in terms of the hallmarks of cancer, and propose potential applications for repurposing metformin for cancer therapeutics.
Similar content being viewed by others
Introduction
Metformin is one of the most commonly prescribed anti-diabetic drugs worldwide. Its history can be traced back to 1918 when guanidine, found in traditional herbal medicine in Europe known as Galega officinalis, was shown to lower glycemia [1]. A series of guanidine derivatives, including metformin, was subsequently synthesized [2]. Over time, the benefits associated with repurposing metformin for several challenging diseases, including obesity [3], cardiovascular diseases [4, 5], liver diseases [6], renal diseases [7], aging-related diseases [8], and cancers [9] have been shown. Epidemiological studies have revealed that metformin exerts protective effects on people with diabetes suffering from cancer [10,11,12]. Intriguingly, several clinical studies have also reported encouraging outcomes in non-diabetic cancer patients [13,14,15]. Given that metformin is safe, well-tolerated, and cost-effective, it is extremely appealing as a focus of antitumor research. Subsequent evidence has shown that metformin inhibits tumor growth, invasion, and metastasis both in vitro and in mouse tumor models for hepatocellular carcinoma [16], ocular melanoma [17], head and neck squamous cell carcinoma [18], and breast cancer [19], among others. Moreover, metformin has been used as a synergistic therapy for cancer, as it enhances sensitivity to radiotherapy [15, 20], chemotherapy [14, 21], and immunotherapy [22] and decreases side effects at lower therapeutic dosages of anticancer treatments.
Great interest has been attached to the basic and clinical study of metformin in cancer. The central mechanism by which metformin attenuates tumorigenesis and progression is through the regulation of energy metabolism. The master pathway of metformin anticancer activity is the activation of the adenosine monophosphate-activated protein kinase (AMPK)/mammalian target of rapamycin (mTOR) pathway triggered by inhibition of complex I in the mitochondrial respiratory chain [23,24,25]. However, the vague performance in a clinical study was in contrast with the excellent performance in a preclinical study. Metformin did not show any benefit in cancer treatment in some clinical trials. Therefore, there are great challenges in the clinical translation of metformin.
Abundant reviews have elaborated on the topic of metformin and cancer from different perspectives, such as specific cancer types [26, 27], diabetes [28, 29], pharmacology [30, 31], and molecular mechanisms [32, 33]. However, insight into the therapeutic repurposing of metformin is still insufficient [34]. Based on the literature review, we recognize that metformin exerts protective effects against multiple tumor types and an increasing number of subtypes [35, 36]. Hence, the mechanisms of action of metformin must be closely related to the hallmarks of cancer [37], which have been proposed as a common set of functional capabilities crucial to the transformation from normalcy to malignancy. This review focused on the effects of metformin on cancer cells in terms of the hallmarks of cancer and updated the clinical translation of metformin in cancer treatment. In this review, we aim to (1) update the readers on the molecular mechanisms through which metformin exhibits antitumor activities, (2) map the effects of metformin on cancer cells in terms of the hallmarks of cancer (Fig. 1), and (3) summarize seminal clinical trials and therapeutic prospects of metformin for cancer treatment.
Update on metformin’s molecular antitumor mechanisms of action
The classic modes of metformin’s antitumor effects are the inhibition of respiratory complex I in the mitochondria and the activation of AMPK in succession. Recently, metformin was defined to inhibit complex I by binding in the quinone channel and exert an independent localized chaotropic effect by combining cryo-electron microscopy and enzyme kinetics [38]. Although the direct interaction between metformin and complex I is essential, metformin-induced complex I inhibition is not a consequence of the direct interaction but instead occurs through an indirect mechanism [39]. Ma et al. [40] conducted another novel study that focused on direct molecular targets of metformin and identified PEN2, a subunit of γ-secretase, as a direct molecular target of metformin. PEN2 binds to ATP6AP1, inhibits the activity of v-ATPase without increasing AMP or ADP, and then activates the lysosomal AMP-independent AMPK pathway.
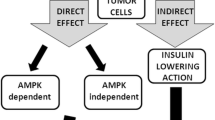
Epidemiologic studies indicate that metformin decreased the risk of cancer incidence compared to other anti-diabetic medications. Hence, the anticarcinogenic effects of metformin were traditionally divided into direct (blood glucose- and insulin-independent) and indirect (blood glucose- and insulin-dependent) effects, being mindful that none of the effects are mutually exclusive.
Direct effects of metformin
Metformin can exert direct effects on cancer cells independent of blood glucose and insulin levels, partly through AMPK activation. It is generally acknowledged that metformin inhibits complex 1 (NADH-coenzyme Q oxidoreductase) of the mitochondrial respiratory chain, which leads to membrane depolarization, reactive oxygen species (ROS) release, and a decrease in the ATP/ADP ratio [41, 42]. Metformin requires a robust inner mitochondrial membrane potential to accumulate within the mitochondrial matrix and reversibly inhibits complex 1 [23]. This inhibition of complex I limits the electron flow to complex III, where ROS are generated. Mitochondrial complex III ROS are hypoxic activators of HIF-1 [43]. Therefore, metformin reduces the hypoxic stabilization of HIF-1α protein and HIF-dependent target genes. Additionally, metformin reduces DNA damage and the production of oxidative stress through mitochondrial respiratory chain inhibition [44]. Metformin depleted the tricarboxylic acid (TCA) cycle and blocked the production of biosynthetic precursors. Nearly all TCA cycle metabolites decrease considerably with metformin treatment [45]. Metformin can also inhibit cancer cell growth by decreasing the cellular energy status, and the effects can be reversed by the expression of the metformin-resistant yeast-derived complex I NADH dehydrogenase NDI1 [46].
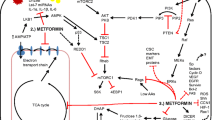
A series of complicated signal pathways are activated by metformin (Fig. 2). First, metformin is a well-known AMPK activator and a key enzyme in glucose homeostasis, gluconeogenesis, and lipid metabolism. AMPK is directly activated by an increase in either the AMP/ATP or ADP/ATP ratio [47] and is indirectly activated by upstream kinases, including LKB1 [48], Ca(2+)/calmodulin-dependent protein kinase kinase (CaMKK) beta [49] and TGFβ-activated kinase-1 (TAK1) [50]. Wu et al. [51] recently demonstrated that metformin protects AMPK-mediated phosphorylation of serine 99, thus increasing TET2 stability and 5-hydroxymethylcytosine (5hmC) levels. A pathway linking diabetes to cancer was revealed through the definition of a novel ‘phospho-switch’ that regulates TET2 stability and a regulatory pathway that links glucose and AMPK to TET2 and 5hmC.
AMPK-dependent mTOR complex 1 (mTORC1) inhibition occurs via multiple downstream effectors that switch on ATP-producing processes and switch off ATP-consuming pathways [52]. These effects can be mediated by the activation of TSC1/TSC2 tumor suppressor genes [53, 54]. TSC1/2 can inhibit mTORC1 and the phosphorylation of its downstream effectors 4EBP1 and S6K [55]. mTORC1 inhibition also occurs as a result of the direct phosphorylation of S722 and S792 on Raptor, a vital mTORC1-binding partner [56].
Metformin can inhibit mTOR through Rag GTPase inactivation or REDD1 activation independent of AMPK activation. Metformin can also inhibit mTORC1 signaling independent of AMPK or TSC1/2, although it is dependent on Rag GTPases. Metformin inhibits growth by inhibiting the mitochondrial respiratory capacity, which inhibits the transit of the RagA-RagC GTPase heterodimer through the nuclear pore complex (NPC). A key transcriptional target, acyl-CoA dehydrogenase family member-10 (ACAD10), is activated when metformin induces the nuclear exclusion of the GTPase RagC, thereby inhibiting mTORC1 [57]. REDD1 (REgulated in Development and DNA damage responses 1), also known as RTP801, Dig2, or DDIT4, has been deemed a hypoxia-inducible factor-1 (HIF-1) target gene and plays a significant role in inhibiting mTORC1 signaling during hypoxic stress. Several other pathways are involved in the anticancer action of metformin, including PI3K/AKT/mTOR [58,59,60], K-Ras [61], nemo-like kinase (NLK) [62], c-Jun-N-terminal kinase (JNK) [63], and Stat3-Bcl-2 [64].
Recently, some studies revealed prognostic and predictive biomarkers, as well as a promising therapeutic target of metformin. ** them evade senescence. Metformin also effectively blocks senescence induced by E6/E7 inhibition or chemotherapy in HPV-positive cancer cells.
Locking phenotypic plasticity
Cancer stem cells (CSCs), a cluster of tumor cells possessing clonogenicity and self-renewal abilities, may play a role in tumor recurrence and metastasis. Metformin has been shown to be preferentially cytotoxic to CSCs compared to non-CSCs [132]. Clonal cell growth and cancer sphere formation are hallmarks of CSCs that can be inhibited by metformin. Metformin has been reported to suppress the expression of CSC markers, including CD44, EpCAM, EZH2, Notch-1, Nanog, and Oct4 in pancreatic cells [133]; CD44 and Sox2 in gastric cancer [110]; Nanog, c-Myc, and TLF4 in NSCLC [62]; and upregulate the expression of differentiation markers, such as Kruppel-like factor 4 (KLF4) and MUC5AC in gastric cancer [110]. Metformin suppresses the self-renewal ability and tumorigenicity of osteosarcoma stem cells via ROS-mediated apoptosis and autophagy [104].
Inspiring inflammation and immunity in cancer (Fig. 4)
Metformin regulates diverse factors to modulate immune cells in the tumor microenvironment to inspire immunity in cancer. Metformin can modulate tumor infiltrating lymphocytes (TIL), tumor-associated macrophages (TAMs), Treg, myeloid-derived suppressor cells (MDSCs), and PDL1 to increase the number and function of T cells and decrease T cell surveillance escape. Metformin can also downregulate PDL1 to increase cytotoxic T cells
Breakthroughs in cancer immunotherapy have expanded the possibilities for cancer therapy over the last decade. Although cancer immunity has continued to be underappreciated, an increasing number of studies have focused on the relationship between cancer, immunity, and potential immunotherapy. Metformin has been found to interact with immune regulators, such as inhibitory immune checkpoints, M2-like tumor-associated macrophages (TAMs), regulatory T cells (T-regs), and myeloid-derived suppressor cells (MDSCs), to inhibit immune destruction.
CD8+ tumor-infiltrating lymphocytes (TILs) inevitably undergo immune exhaustion, which is characterized by decreased production of multiple cytokines, such as IL-2, TNFα, and IFNγ, followed by a reduction in apoptosis. Metformin increases CD8+ TILs and protects them from exhaustion and apoptosis in the TME. Furthermore, the adoptive transfer of metformin-treated antigen-specific CD8+ T cells efficiently migrates into tumors and maintains multifunctionality in a manner that is sensitive to the AMPK inhibitor compound C [134]. Metformin also inhibits TAM infiltration during prostate cancer initiation and progression by inhibiting the COX2/PGE2 axis [135].
A window of opportunity trial for HNSCC has demonstrated that metformin modulates metabolism in the HNSCC microenvironment [136]. Metformin decreases infiltration of FOXP3+ T regulatory cells in intratumor regions, increases CD8+ cytotoxic T cell infiltration in the peritumoral leading edge stroma, and increases the CD8/FOXP3 ratio both in the tumor and leading-edge stroma of primary HNSCC tumors [137]. Metformin may positively interact with the immune TME in HNSCC, regardless of HPV status. Metformin inhibits CCR1 surface expression in HNSCC cells and the expression of CCL15 in M2-type TAMs, which promote HNSCC cell resistance to gefitinib under hypoxic conditions through the CCL15-CCR1-NF-κB pathway [138].
In a zebrafish model of nonalcoholic fatty liver disease (NAFLD)-associated hepatocellular carcinoma, metformin was found to alter macrophage polarization and exacerbate the liver inflammatory microenvironment and cancer progression [139]. In addition, metformin rescued the effects of a high-fat diet (HFD) on liver tumorigenesis (angiogenesis, steatosis, lipotoxicity), inflammation, and T cell recruitment to the liver.
Combined with immune checkpoint blockade (ICB), metformin has been found to decrease T-reg and MDSC levels and increase CD8+ levels in murine models [140]. Notably, only long-term metformin treatment is sufficient to reduce cancer cell growth. Programmed cell death 1 (PD-1)/programmed death ligand 1 (PD-L1), a representative ICB, has initiated a new era in cancer treatment. Cha et al. [40] revealed that PEN2 is the direct molecular target of metformin using a photoactive metformin probe. Bridges et al. [38] recently defined the inhibitory drug-target interactions of metformin with mammalian respiratory complex I by combining cryo-electron microscopy and enzyme kinetics. The identification of the direct target of metformin’s anticancer effects may help to further investigation for drug development. Second, in in vitro research, the direct effects are emphasized and well-studied, while the indirect effects cannot be mimicked. Some in vivo and clinical studies suggest that indirect insulin-dependent effects may be of great significance in at least some cancers, such as breast cancer and lung cancer. The research directions varied for the two modes of anticancer effects (Table 2). In terms of “direct effects,” more effort should be put into markers such as LKB1, Rag GTPases and REDD1. In terms of “indirect effects,” more effort should be put into markers such as blood glucose and insulin levels, insulin resistance, expression of insulin receptors and insulin-like growth factor receptor 1, and targets in the liver. Understanding the markers helps to predict the therapeutic response of patients. For example, the synergistic effects between metformin and gefitinib were reported to rely on the presence of wild-type LKB1 in NSCLC cells [178]. Third, since indirect effects could not be simulated in vitro, more in vivo models are needed to reexamine the direct and indirect effects of metformin and the possible interactions. Some attempts have been made to develop related models. For instance, in research on the anticancer activity of metformin, hyperglycemic mice were reported to lose sensitivity to metformin compared with normoglycemic mice, probably through increased c-Myc expression, glycolytic enzymes hexokinase 2 and pyruvate dehydrogenase kinase 1 [179]. We expect more in vivo models with more complex designs that are currently used in most in vitro research.
There is a discrepancy in the antitumor effect of metformin between clinical research and preclinical studies, although metformin has shown notable benefits for cancer prevention and treatment in preclinical research, and the related molecular mechanisms have been extensively studied. The challenges mainly include (1) simulating pharmacokinetics consistent with clinical settings, including appropriate metformin concentrations and dosing time; (2) exploring suitable synergetic therapies and patients who are more sensitive to metformin; and (3) utilizing other forms of biguanides, such as phenformin or modified biguanides, which have better performances. Therefore, further research is required regarding the critical aspects mentioned above. First, many preclinical studies have employed metformin at concentrations that are considerably higher than what would be deemed safe in clinical settings [180]. The plasma concentrations of metformin were reported to be 5–30 μmol/L in patients taking clinical doses of 1.5–2 g per day, which were the most common doses for diabetes and were used in most clinical trials for cancer treatment [181]. However, the concentrations in most preclinical studies in vitro (300 μmol/L–10 mmol/L) were dozens or even a thousand times the clinical concentrations. The dosages of in vivo studies (200–1000 mg/kg per day) were 6–30 times the clinical dosages (approximately 30 mg/kg per day) with metformin diluted in the drinking water or intraperitoneally injected. Recent studies have noted this issue. Metformin can suppress cancer at a clinically safe concentration in vitro [40] and in vivo [16, 35, 183]. In the clinical setting, improved outcomes were observed only among patients with early-stage NSCLC or those who took metformin before the NSCLC diagnosis [184]. Therefore, it enlightens us to apply metformin once cancer is diagnosed or even in people with a high risk of cancer if possible. Second, based on our understanding of metformin’s effects on cancer hallmarks, metformin could be a useful adjuvant agent in combination therapy to combat cancer synergistically in certain patients with certain cancers [36]. It can be administered along with chemotherapy, radiotherapy, immunotherapy or targeted therapy. Taking immunotherapy as an example, a high level of lactate can lead to tumor immune tolerance, while metformin was reported to increase the level of lactate in the intra- and extracellular environment [31, 185]. However, from the current evidence, acidification of the TME made tumors more susceptible to metformin due to the loss of NAD+ regeneration capacity [78, 79, 186]. Therefore, whether metformin can cause tumor immune tolerance by increasing the acidification of the TME remains an interesting issue to explore. If indeed, the combination of metformin and immunotherapy might be a possible direction in further research. Besides, metformin was reported to show no more benefits in some cancers with certain mutations [158] or advanced stages [164, 175, 176] or in patients without diabetes [157, 158]. We call for further high-quality clinical trials on metformin combined with other therapies in different types of physiological conditions and cancers. Third, it was reported that phenformin may outperform metformin owing to its unique pharmacokinetic characteristics, which include better absorption and inhibition of the mitochondria [187]. Although the incidence of lactic acidosis associated with phenformin is higher than that associated with metformin, phenformin is in any case safer than other cancer treatments. Moreover, given the pharmacokinetic differences between metformin and phenformin, we can obtain more insights regarding drug modification. Once there is more evidence, we expect metformin or other forms of biguanides to exert a greater influence on anticancer therapy, at the appropriate dosage, on patients of appropriate metabolic state, and in combination with other therapies.
Conclusion
The review details the possible molecular mechanisms of metformin in cancer prevention and treatment, elucidates its role in terms of cancer hallmarks, and more importantly, analyses current challenges and future directions in clinical translation.
Availability of data and materials
All data generated are included in this published article or from public sources.
References
Watanabe CK. Studies in the metabolism changes induced by administration of guanidine bases: I. Influence of injected guanidine hydrochloride upon blood sugar content. J Biol Chem. 1918;33(2):253–65.
Werner EA, Bell J. CCXIV.—The preparation of methylguanidine, and of ββ-dimethylguanidine by the interaction of dicyanodiamide, and methylammonium and dimethylammonium chlorides respectively. J Chem Soc Trans. 1922;121:1790–4.
Apolzan JW, Venditti EM, Edelstein SL, et al. Long-term weight loss with metformin or lifestyle intervention in the diabetes prevention program outcomes study. Ann Intern Med. 2019;170(10):682–90.
UK Prospective Diabetes Study (UKPDS) Group. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). Lancet. 1998;352(9131):854–65.
Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HA. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med. 2008;359(15):1577–89.
Marchesini G, Brizi M, Bianchi G, Tomassetti S, Zoli M, Melchionda N. Metformin in non-alcoholic steatohepatitis. Lancet. 2001;358(9285):893–4.
De Broe ME, Kajbaf F, Lalau JD. Renoprotective effects of metformin. Nephron. 2018;138(4):261–74.
Campbell JM, Bellman SM, Stephenson MD, Lisy K. Metformin reduces all-cause mortality and diseases of ageing independent of its effect on diabetes control: a systematic review and meta-analysis. Ageing Res Rev. 2017;40:31–44.
Evans JM, Donnelly LA, Emslie-Smith AM, Alessi DR, Morris AD. Metformin and reduced risk of cancer in diabetic patients. BMJ. 2005;330(7503):1304–5.
Decensi A, Puntoni M, Goodwin P, et al. Metformin and cancer risk in diabetic patients: a systematic review and meta-analysis. Cancer Prev Res (Phila). 2010;3(11):1451–61.
Stevens RJ, Ali R, Bankhead CR, et al. Cancer outcomes and all-cause mortality in adults allocated to metformin: systematic review and collaborative meta-analysis of randomised clinical trials. Diabetologia. 2012;55(10):2593–603.
Heckman-Stoddard BM, DeCensi A, Sahasrabuddhe VV, Ford LG. Repurposing metformin for the prevention of cancer and cancer recurrence. Diabetologia. 2017;60(9):1639–47.
Rothermundt C, Hayoz S, Templeton AJ, et al. Metformin in chemotherapy-naive castration-resistant prostate cancer: a multicenter phase 2 trial (SAKK 08/09). Eur Urol. 2014;66(3):468–74.
Brown JR, Chan DK, Shank JJ, et al. Phase II clinical trial of metformin as a cancer stem cell-targeting agent in ovarian cancer. JCI Insight. 2020;5(11): e133247.
Gulati S, Desai J, Palackdharry SM, et al. Phase 1 dose-finding study of metformin in combination with concurrent cisplatin and radiotherapy in patients with locally advanced head and neck squamous cell cancer. Cancer. 2020;126(2):354–62.
Chen HP, Shieh JJ, Chang CC, et al. Metformin decreases hepatocellular carcinoma risk in a dose-dependent manner: population-based and in vitro studies. Gut. 2013;62(4):606–15.
Zhuang A, Chai P, Wang S, et al. Metformin promotes histone deacetylation of optineurin and suppresses tumour growth through autophagy inhibition in ocular melanoma. Clin Transl Med. 2022;12(1): e660.
Sikka A, Kaur M, Agarwal C, Deep G, Agarwal R. Metformin suppresses growth of human head and neck squamous cell carcinoma via global inhibition of protein translation. Cell Cycle. 2012;11(7):1374–82.
Zakikhani M, Dowling R, Fantus IG, Sonenberg N, Pollak M. Metformin is an AMP kinase-dependent growth inhibitor for breast cancer cells. Cancer Res. 2006;66(21):10269–73.
Wink KC, Belderbos JS, Dieleman EM, et al. Improved progression free survival for patients with diabetes and locally advanced non-small cell lung cancer (NSCLC) using metformin during concurrent chemoradiotherapy. Radiother Oncol. 2016;118(3):453–9.
Saif MW, Rajagopal S, Caplain J, et al. A phase I delayed-start, randomized and pharmacodynamic study of metformin and chemotherapy in patients with solid tumors. Cancer Chemother Pharmacol. 2019;84(6):1323–31.
Crist M, Yaniv B, Palackdharry S, et al. Metformin increases natural killer cell functions in head and neck squamous cell carcinoma through CXCL1 inhibition. J Immunother Cancer. 2022;10(11): e005632.
Owen MR, Doran E, Halestrap AP. Evidence that metformin exerts its anti-diabetic effects through inhibition of complex 1 of the mitochondrial respiratory chain. Biochem J. 2000;348(Pt 3):607–14.
Zhou G, Myers R, Li Y, et al. Role of AMP-activated protein kinase in mechanism of metformin action. J Clin Invest. 2001;108(8):1167–74.
Spiering MJ. The mystery of metformin. J Biol Chem. 2019;294(17):6689–91.
Clements A, Gao B, Yeap SHO, Wong MKY, Ali SS, Gurney H. Metformin in prostate cancer: two for the price of one. Ann Oncol. 2011;22(12):2556–60.
Eibl G, Rozengurt E. Metformin: review of epidemiology and mechanisms of action in pancreatic cancer. Cancer Metastasis Rev. 2021;40(3):865–78.
Pernicova I, Korbonits M. Metformin—mode of action and clinical implications for diabetes and cancer. Nat Rev Endocrinol. 2014;10(3):143–56.
Farmer RE, Ford D, Forbes HJ, et al. Metformin and cancer in type 2 diabetes: a systematic review and comprehensive bias evaluation. Int J Epidemiol. 2017;46(2):728–44.
EmamiRiedmaier A, Fisel P, Nies AT, Schaeffeler E, Schwab M. Metformin and cancer: from the old medicine cabinet to pharmacological pitfalls and prospects. Trends Pharmacol Sci. 2013;34(2):126–35.
Jara JA, López-Muñoz R. Metformin and cancer: between the bioenergetic disturbances and the antifolate activity. Pharmacol Res. 2015;101:102–8.
Vancura A, Bu P, Bhagwat M, Zeng J, Vancurova I. Metformin as an anticancer agent. Trends Pharmacol Sci. 2018;39(10):867–78.
Wang G, Lin F, Wan Q, Wu J, Luo M. Mechanisms of action of metformin and its regulatory effect on microRNAs related to angiogenesis. Pharmacol Res. 2021;164: 105390.
Mallik R, Chowdhury TA. Metformin in cancer. Diabetes Res Clin Pract. 2018;143:409–19.
Iliopoulos D, Hirsch HA, Struhl K. Metformin decreases the dose of chemotherapy for prolonging tumor remission in mouse xenografts involving multiple cancer cell types. Cancer Res. 2011;71(9):3196–201.
Coyle C, Cafferty FH, Vale C, Langley RE. Metformin as an adjuvant treatment for cancer: a systematic review and meta-analysis. Ann Oncol. 2016;27(12):2184–95.
Hanahan D. Hallmarks of cancer: new dimensions. Cancer Discov. 2022;12(1):31–46.
Bridges HR, Blaza JN, Yin Z, Chung I, Pollak MN, Hirst J. Structural basis of mammalian respiratory complex I inhibition by medicinal biguanides. Science. 2023;379(6630):351–7.
El-Mir MY, Nogueira V, Fontaine E, Avéret N, Rigoulet M, Leverve X. Dimethylbiguanide inhibits cell respiration via an indirect effect targeted on the respiratory chain complex I. J Biol Chem. 2000;275(1):223–8.
Ma T, Tian X, Zhang B, et al. Low-dose metformin targets the lysosomal AMPK pathway through PEN2. Nature. 2022;603(7899):159–65.
Whitaker-Menezes D, Martinez-Outschoorn UE, Flomenberg N, et al. Hyperactivation of oxidative mitochondrial metabolism in epithelial cancer cells in situ: visualizing the therapeutic effects of metformin in tumor tissue. Cell Cycle. 2011;10(23):4047–64.
Pierotti MA, Berrino F, Gariboldi M, et al. Targeting metabolism for cancer treatment and prevention: metformin, an old drug with multi-faceted effects. Oncogene. 2013;32(12):1475–87.
Bell EL, Klimova TA, Eisenbart J, et al. The Qo site of the mitochondrial complex III is required for the transduction of hypoxic signaling via reactive oxygen species production. J Cell Biol. 2007;177(6):1029–36.
Algire C, Moiseeva O, Deschênes-Simard X, et al. Metformin reduces endogenous reactive oxygen species and associated DNA damage. Cancer Prev Res (Phila). 2012;5(4):536–43.
Janzer A, German NJ, Gonzalez-Herrera KN, Asara JM, Haigis MC, Struhl K. Metformin and phenformin deplete tricarboxylic acid cycle and glycolytic intermediates during cell transformation and NTPs in cancer stem cells. Proc Natl Acad Sci USA. 2014;111(29):10574–9.
Wheaton WW, Weinberg SE, Hamanaka RB, et al. Metformin inhibits mitochondrial complex I of cancer cells to reduce tumorigenesis. Elife. 2014;3: e02242.
Hardie DG, Ross FA, Hawley SA. AMPK: a nutrient and energy sensor that maintains energy homeostasis. Nat Rev Mol Cell Biol. 2012;13(4):251–62.
Woods A, Johnstone SR, Dickerson K, et al. LKB1 is the upstream kinase in the AMP-activated protein kinase cascade. Curr Biol. 2003;13(22):2004–8.
Woods A, Dickerson K, Heath R, et al. Ca2+/calmodulin-dependent protein kinase kinase-beta acts upstream of AMP-activated protein kinase in mammalian cells. Cell Metab. 2005;2(1):21–33.
Momcilovic M, Hong SP, Carlson M. Mammalian TAK1 activates Snf1 protein kinase in yeast and phosphorylates AMP-activated protein kinase in vitro. J Biol Chem. 2006;281(35):25336–43.
Wu D, Hu D, Chen H, et al. Glucose-regulated phosphorylation of TET2 by AMPK reveals a pathway linking diabetes to cancer. Nature. 2018;559(7715):637–41.
Hardie DG, Ashford ML. AMPK: regulating energy balance at the cellular and whole body levels. Physiology (Bethesda). 2014;29(2):99–107.
Gao X, Zhang Y, Arrazola P, et al. Tsc tumour suppressor proteins antagonize amino-acid-TOR signalling. Nat Cell Biol. 2002;4(9):699–704.
Inoki K, Zhu T, Guan KL. TSC2 mediates cellular energy response to control cell growth and survival. Cell. 2003;115(5):577–90.
Fingar DC, Salama S, Tsou C, Harlow E, Blenis J. Mammalian cell size is controlled by mTOR and its downstream targets S6K1 and 4EBP1/eIF4E. Genes Dev. 2002;16(12):1472–87.
Gwinn DM, Shackelford DB, Egan DF, et al. AMPK phosphorylation of raptor mediates a metabolic checkpoint. Mol Cell. 2008;30(2):214–26.
Wu L, Zhou B, Oshiro-Rapley N, et al. An ancient, unified mechanism for metformin growth inhibition in C. elegans and cancer. Cell. 2016;167(7):1705-1718.e1713.
Fu YL, Zhang QH, Wang XW, He H. Antidiabetic drug metformin mitigates ovarian cancer SKOV3 cell growth by triggering G2/M cell cycle arrest and inhibition of m-TOR/PI3K/Akt signaling pathway. Eur Rev Med Pharmacol Sci. 2017;21(5):1169–75.
Sun R, Zhai R, Ma C, Miao W. Combination of aloin and metformin enhances the antitumor effect by inhibiting the growth and invasion and inducing apoptosis and autophagy in hepatocellular carcinoma through PI3K/AKT/mTOR pathway. Cancer Med. 2020;9(3):1141–51.
Zi FM, He JS, Li Y, et al. Metformin displays anti-myeloma activity and synergistic effect with dexamethasone in in vitro and in vivo xenograft models. Cancer Lett. 2015;356(2 Pt B):443–53.
Iglesias DA, Yates MS, van der Hoeven D, et al. Another surprise from metformin: novel mechanism of action via K-Ras influences endometrial cancer response to therapy. Mol Cancer Ther. 2013;12(12):2847–56.
Suwei D, Liang Z, Zhimin L, et al. NLK functions to maintain proliferation and stemness of NSCLC and is a target of metformin. J Hematol Oncol. 2015;8:120.
Li B, Zhou P, Xu K, et al. Metformin induces cell cycle arrest, apoptosis and autophagy through ROS/JNK signaling pathway in human osteosarcoma. Int J Biol Sci. 2020;16(1):74–84.
Feng Y, Ke C, Tang Q, et al. Metformin promotes autophagy and apoptosis in esophageal squamous cell carcinoma by downregulating Stat3 signaling. Cell Death Dis. 2014;5(2): e1088.
**e J, Ye J, Cai Z, et al. GPD1 enhances the anticancer effects of metformin by synergistically increasing total cellular glycerol-3-phosphate. Cancer Res. 2020;80(11):2150–62.
Barbieri F, Bosio AG, Pattarozzi A, et al. Chloride intracellular channel 1 activity is not required for glioblastoma development but its inhibition dictates glioma stem cell responsivity to novel biguanide derivatives. J Exp Clin Cancer Res. 2022;41(1):53.
Barbieri F, Würth R, Pattarozzi A, et al. Inhibition of chloride intracellular channel 1 (CLIC1) as biguanide class-effect to impair human glioblastoma stem cell viability. Front Pharmacol. 2018;9:899.
Liu Y, Wang Z, Li M, et al. Chloride intracellular channel 1 regulates the antineoplastic effects of metformin in gallbladder cancer cells. Cancer Sci. 2017;108(6):1240–52.
Belfiore A, Frasca F. IGF and insulin receptor signaling in breast cancer. J Mammary Gland Biol Neoplasia. 2008;13:381–406.
Pollak M. Insulin and insulin-like growth factor signalling in neoplasia. Nat Rev Cancer. 2008;8:915–28.
Nestler JE, Powers LP, Matt DW, et al. A direct effect of hyperinsulinemia on serum sex hormone-binding globulin levels in obese women with the polycystic ovary syndrome. J Clin Endocrinol Metab. 1991;72(1):83–9.
Key T, Appleby P, Barnes I, Reeves G. Endogenous sex hormones and breast cancer in postmenopausal women: reanalysis of nine prospective studies. J Natl Cancer Inst. 2002;94(8):606–16.
Gonda TA, Tu S, Wang TC. Chronic inflammation, the tumor microenvironment and carcinogenesis. Cell Cycle. 2009;8(13):2005–13.
Warburg O. On the origin of cancer cells. Science. 1956;123(3191):309–14.
Fendt SM, Bell EL, Keibler MA, et al. Metformin decreases glucose oxidation and increases the dependency of prostate cancer cells on reductive glutamine metabolism. Cancer Res. 2013;73(14):4429–38.
Lord SR, Collins JM, Cheng WC, et al. Transcriptomic analysis of human primary breast cancer identifies fatty acid oxidation as a target for metformin. Br J Cancer. 2020;122(2):258–65.
Lord SR, Cheng WC, Liu D, et al. Integrated pharmacodynamic analysis identifies two metabolic adaption pathways to metformin in breast cancer. Cell Metab. 2018;28(5):679-688.e674.
Yoshida J, Ohishi T, Abe H, et al. Mitochondrial complex I inhibitors suppress tumor growth through concomitant acidification of the intra- and extracellular environment. iScience. 2021;24(12): 103497.
Benjamin D, Robay D, Hindupur SK, et al. Dual inhibition of the lactate transporters MCT1 and MCT4 is synthetic lethal with metformin due to NAD+ depletion in cancer cells. Cell Rep. 2018;25(11):3047-3058.e3044.
Gui DY, Sullivan LB, Luengo A, et al. Environment dictates dependence on mitochondrial complex I for NAD+ and aspartate production and determines cancer cell sensitivity to metformin. Cell Metab. 2016;24(5):716–27.
Elgendy M, Cirò M, Hosseini A, et al. Combination of hypoglycemia and metformin impairs tumor metabolic plasticity and growth by modulating the PP2A-GSK3β-MCL-1 axis. Cancer Cell. 2019;35(5):798-815.e795.
Giovannucci E, Harlan DM, Archer MC, et al. Diabetes and cancer: a consensus report. CA Cancer J Clin. 2010;60(4):207–21.
Shlomai G, Neel B, LeRoith D, Gallagher EJ. Type 2 diabetes mellitus and cancer: the role of pharmacotherapy. J Clin Oncol. 2016;34(35):4261–9.
Chen H, Zheng X, Zong X, et al. Metabolic syndrome, metabolic comorbid conditions and risk of early-onset colorectal cancer. Gut. 2021;70(6):1147–54.
Kinnaird A, Zhao S, Wellen KE, Michelakis ED. Metabolic control of epigenetics in cancer. Nat Rev Cancer. 2016;16(11):694–707.
Sharma U, Rando OJ. Metabolic inputs into the epigenome. Cell Metab. 2017;25(3):544–58.
Zhong T, Men Y, Lu L, et al. Metformin alters DNA methylation genome-wide via the H19/SAHH axis. Oncogene. 2017;36(17):2345–54.
Cuyàs E, Fernández-Arroyo S, Verdura S, et al. Metformin regulates global DNA methylation via mitochondrial one-carbon metabolism. Oncogene. 2018;37(7):963–70.
Kim KH, Roberts CW. Targeting EZH2 in cancer. Nat Med. 2016;22(2):128–34.
Wan L, Xu K, Wei Y, et al. Phosphorylation of EZH2 by AMPK suppresses PRC2 methyltransferase activity and oncogenic function. Mol Cell. 2018;69(2):279-291.e275.
Yuan H, Han Y, Wang X, et al. SETD2 restricts prostate cancer metastasis by integrating EZH2 and AMPK signaling pathways. Cancer Cell. 2020;38(3):350-365.e357.
Cuyàs E, Verdura S, Llorach-Pares L, et al. Metformin directly targets the H3K27me3 demethylase KDM6A/UTX. Aging Cell. 2018;17(4): e12772.
Panwalkar P, Tamrazi B, Dang D, et al. Targeting integrated epigenetic and metabolic pathways in lethal childhood PFA ependymomas. Sci Transl Med. 2021;13(614): eabc0497.
Li K, Zhang TT, Hua F, Hu ZW. Metformin reduces TRIB3 expression and restores autophagy flux: an alternative antitumor action. Autophagy. 2018;14(7):1278–9.
Pulito C, Mori F, Sacconi A, et al. Metformin-induced ablation of microRNA 21–5p releases Sestrin-1 and CAB39L antitumoral activities. Cell Discov. 2017;3:17022.
** D, Guo J, Wu Y, et al. Metformin-repressed miR-381-YAP-snail axis activity disrupts NSCLC growth and metastasis. J Exp Clin Cancer Res. 2020;39(1):6.
Cifarelli V, Lashinger LM, Devlin KL, et al. Metformin and rapamycin reduce pancreatic cancer growth in obese prediabetic mice by distinct microRNA-regulated mechanisms. Diabetes. 2015;64(5):1632–42.
**e W, Wang L, Sheng H, et al. Metformin induces growth inhibition and cell cycle arrest by upregulating microRNA34a in renal cancer cells. Med Sci Monit. 2017;23:29–37.
Wang L, Li K, Lin X, et al. Metformin induces human esophageal carcinoma cell pyroptosis by targeting the miR-497/PELP1 axis. Cancer Lett. 2019;450:22–31.
Bernstein E, Caudy AA, Hammond SM, Hannon GJ. Role for a bidentate ribonuclease in the initiation step of RNA interference. Nature. 2001;409(6818):363–6.
Ashinuma H, Takiguchi Y, Kitazono S, et al. Antiproliferative action of metformin in human lung cancer cell lines. Oncol Rep. 2012;28(1):8–14.
Kato K, Gong J, Iwama H, et al. The antidiabetic drug metformin inhibits gastric cancer cell proliferation in vitro and in vivo. Mol Cancer Ther. 2012;11(3):549–60.
Miyoshi H, Kato K, Iwama H, et al. Effect of the anti-diabetic drug metformin in hepatocellular carcinoma in vitro and in vivo. Int J Oncol. 2014;45(1):322–32.
Zhao B, Luo J, Wang Y, et al. Metformin suppresses self-renewal ability and tumorigenicity of osteosarcoma stem cells via reactive oxygen species-mediated apoptosis and autophagy. Oxid Med Cell Longev. 2019;2019:9290728.
Wang Y, Xu W, Yan Z, et al. Metformin induces autophagy and G0/G1 phase cell cycle arrest in myeloma by targeting the AMPK/mTORC1 and mTORC2 pathways. J Exp Clin Cancer Res. 2018;37(1):63.
Zimmermann M, Arachchige-Don AP, Donaldson MS, Patriarchi T, Horne MC. Cyclin G2 promotes cell cycle arrest in breast cancer cells responding to fulvestrant and metformin and correlates with patient survival. Cell Cycle. 2016;15(23):3278–95.
Chen G, Xu S, Renko K, Derwahl M. Metformin inhibits growth of thyroid carcinoma cells, suppresses self-renewal of derived cancer stem cells, and potentiates the effect of chemotherapeutic agents. J Clin Endocrinol Metab. 2012;97(4):E510–20.
Wu Y, Zheng Q, Li Y, et al. Metformin targets a YAP1-TEAD4 complex via AMPKα to regulate CCNE1/2 in bladder cancer cells. J Exp Clin Cancer Res. 2019;38(1):376.
Babcook MA, Sramkoski RM, Fujioka H, et al. Combination simvastatin and metformin induces G1-phase cell cycle arrest and Ripk1- and Ripk3-dependent necrosis in C4-2B osseous metastatic castration-resistant prostate cancer cells. Cell Death Dis. 2014;5(11): e1536.
Courtois S, Durán RV, Giraud J, et al. Metformin targets gastric cancer stem cells. Eur J Cancer. 2017;84:193–201.
Janjetovic K, Harhaji-Trajkovic L, Misirkic-Marjanovic M, et al. In vitro and in vivo anti-melanoma action of metformin. Eur J Pharmacol. 2011;668(3):373–82.
Mogavero A, Maiorana MV, Zanutto S, et al. Metformin transiently inhibits colorectal cancer cell proliferation as a result of either AMPK activation or increased ROS production. Sci Rep. 2017;7(1):15992.
Varghese S, Samuel SM, Varghese E, Kubatka P, Büsselberg D. High glucose represses the anti-proliferative and pro-apoptotic effect of metformin in triple negative breast cancer cells. Biomolecules. 2019;9(1):16.
Ferretti AC, Hidalgo F, Tonucci FM, et al. Metformin and glucose starvation decrease the migratory ability of hepatocellular carcinoma cells: targeting AMPK activation to control migration. Sci Rep. 2019;9(1):2815.
Park JH, Kim YH, Park EH, et al. Effects of metformin and phenformin on apoptosis and epithelial–mesenchymal transition in chemoresistant rectal cancer. Cancer Sci. 2019;110(9):2834–45.
Wang JC, Li GY, Wang B, et al. Metformin inhibits metastatic breast cancer progression and improves chemosensitivity by inducing vessel normalization via PDGF-B downregulation. J Exp Clin Cancer Res. 2019;38(1):235.
Ye J, Qi L, Chen K, et al. Metformin induces TPC-1 cell apoptosis through endoplasmic reticulum stress-associated pathways in vitro and in vivo. Int J Oncol. 2019;55(1):331–9.
He Y, Tai S, Deng M, et al. Metformin and 4SC-202 synergistically promote intrinsic cell apoptosis by accelerating ΔNp63 ubiquitination and degradation in oral squamous cell carcinoma. Cancer Med. 2019;8(7):3479–90.
Lindsay C, Kostiuk M, Conrad D, et al. Antitumour effects of metformin and curcumin in human papillomavirus positive and negative head and neck cancer cells. Mol Carcinog. 2019;58(11):1946–59.
Zheng Z, Bian Y, Zhang Y, Ren G, Li G. Metformin activates AMPK/SIRT1/NF-κB pathway and induces mitochondrial dysfunction to drive caspase3/GSDME-mediated cancer cell pyroptosis. Cell Cycle. 2020;19(10):1089–104.
Yang J, Zhou Y, **e S, et al. Metformin induces ferroptosis by inhibiting UFMylation of SLC7A11 in breast cancer. J Exp Clin Cancer Res. 2021;40(1):206.
Alimova IN, Liu B, Fan Z, et al. Metformin inhibits breast cancer cell growth, colony formation and induces cell cycle arrest in vitro. Cell Cycle. 2009;8(6):909–15.
Muñoz-Espín D, Serrano M. Cellular senescence: from physiology to pathology. Nat Rev Mol Cell Biol. 2014;15(7):482–96.
Campisi J. Cellular senescence as a tumor-suppressor mechanism. Trends Cell Biol. 2001;11(11):S27–31.
Yi G, He Z, Zhou X, et al. Low concentration of metformin induces a p53-dependent senescence in hepatoma cells via activation of the AMPK pathway. Int J Oncol. 2013;43(5):1503–10.
Zhou X, Chen J, Chen L, et al. Negative regulation of Sirtuin 1 by AMP-activated protein kinase promotes metformin-induced senescence in hepatocellular carcinoma xenografts. Cancer Lett. 2017;411:1–11.
Li P, Zhao M, Parris AB, Feng X, Yang X. p53 is required for metformin-induced growth inhibition, senescence and apoptosis in breast cancer cells. Biochem Biophys Res Commun. 2015;464(4):1267–74.
Cufí S, Vazquez-Martin A, Oliveras-Ferraros C, et al. Metformin lowers the threshold for stress-induced senescence: a role for the microRNA-200 family and miR-205. Cell Cycle. 2012;11(6):1235–46.
Hu Q, Peng J, Jiang L, et al. Metformin as a senostatic drug enhances the anticancer efficacy of CDK4/6 inhibitor in head and neck squamous cell carcinoma. Cell Death Dis. 2020;11(10):925.
Yang B, Damodaran S, Khemees TA, et al. Synthetic lethal metabolic targeting of androgen-deprived prostate cancer cells with metformin. Mol Cancer Ther. 2020;19(11):2278–87.
Hoppe-Seyler K, Herrmann AL, Däschle A, et al. Effects of metformin on the virus/host cell crosstalk in human papillomavirus-positive cancer cells. Int J Cancer. 2021;149(5):1137–49.
Song CW, Lee H, Dings RP, et al. Metformin kills and radiosensitizes cancer cells and preferentially kills cancer stem cells. Sci Rep. 2012;2:362.
Bao B, Wang Z, Ali S, et al. Metformin inhibits cell proliferation, migration and invasion by attenuating CSC function mediated by deregulating miRNAs in pancreatic cancer cells. Cancer Prev Res (Phila). 2012;5(3):355–64.
Eikawa S, Nishida M, Mizukami S, Yamazaki C, Nakayama E, Udono H. Immune-mediated antitumor effect by type 2 diabetes drug, metformin. Proc Natl Acad Sci USA. 2015;112(6):1809–14.
Liu Q, Tong D, Liu G, et al. Metformin inhibits prostate cancer progression by targeting tumor-associated inflammatory infiltration. Clin Cancer Res. 2018;24(22):5622–34.
Curry J, Johnson J, Tassone P, et al. Metformin effects on head and neck squamous carcinoma microenvironment: window of opportunity trial. Laryngoscope. 2017;127(8):1808–15.
Amin D, Richa T, Mollaee M, et al. Metformin effects on FOXP3(+) and CD8(+) T cell infiltrates of head and neck squamous cell carcinoma. Laryngoscope. 2020;130(9):E490–8.
Yin X, Han S, Song C, et al. Metformin enhances gefitinib efficacy by interfering with interactions between tumor-associated macrophages and head and neck squamous cell carcinoma cells. Cell Oncol (Dordr). 2019;42(4):459–75.
de Oliveira S, Houseright RA, Graves AL, et al. Metformin modulates innate immune-mediated inflammation and early progression of NAFLD-associated hepatocellular carcinoma in zebrafish. J Hepatol. 2019;70(4):710–21.
Veeramachaneni R, Yu W, Newton JM, et al. Metformin generates profound alterations in systemic and tumor immunity with associated antitumor effects. J Immunother Cancer. 2021;9(7): e002773.
Cha JH, Yang WH, **a W, et al. Metformin promotes antitumor immunity via endoplasmic-reticulum-associated degradation of PD-L1. Mol Cell. 2018;71(4):606-620.e607.
Wen M, Cao Y, Wu B, et al. PD-L1 degradation is regulated by electrostatic membrane association of its cytoplasmic domain. Nat Commun. 2021;12(1):5106.
Sun L, **e C, Wang G, et al. Gut microbiota and intestinal FXR mediate the clinical benefits of metformin. Nat Med. 2018;24(12):1919–29.
Pryor R, Norvaisas P, Marinos G, et al. Host-microbe-drug-nutrient screen identifies bacterial effectors of metformin therapy. Cell. 2019;178(6):1299-1312.e1229.
Bauer PV, Duca FA, Waise TMZ, et al. Metformin alters upper small intestinal microbiota that impact a glucose-SGLT1-sensing glucoregulatory pathway. Cell Metab. 2018;27(1):101-117.e105.
Zhang E, ** L, Wang Y, et al. Intestinal AMPK modulation of microbiota mediates crosstalk with brown fat to control thermogenesis. Nat Commun. 2022;13(1):1135.
Dong TS, Chang HH, Hauer M, et al. Metformin alters the duodenal microbiome and decreases the incidence of pancreatic ductal adenocarcinoma promoted by diet-induced obesity. Am J Physiol Gastrointest Liver Physiol. 2019;317(6):G763–72.
Huang X, Hong X, Wang J, et al. Metformin elicits antitumour effect by modulation of the gut microbiota and rescues Fusobacterium nucleatum-induced colorectal tumourigenesis. EBioMedicine. 2020;61: 103037.
Ayoub R, Ruddy RM, Cox E, et al. Assessment of cognitive and neural recovery in survivors of pediatric brain tumors in a pilot clinical trial using metformin. Nat Med. 2020;26(8):1285–94.
Ramos-Peñafiel C, Olarte-Carrillo I, Cerón-Maldonado R, et al. Effect of metformin on the survival of patients with ALL who express high levels of the ABCB1 drug resistance gene. J Transl Med. 2018;16(1):245.
Tapia E, Villa-Guillen DE, Chalasani P, et al. A randomized controlled trial of metformin in women with components of metabolic syndrome: intervention feasibility and effects on adiposity and breast density. Breast Cancer Res Treat. 2021;190:69–78.
Nwanaji-Enwerem JC, Chung FF, Van der Laan L, et al. An epigenetic aging analysis of randomized metformin and weight loss interventions in overweight postmenopausal breast cancer survivors. Clin Epigenet. 2021;13(1):224.
Pimentel I, Lohmann AE, Ennis M, et al. A phase II randomized clinical trial of the effect of metformin versus placebo on progression-free survival in women with metastatic breast cancer receiving standard chemotherapy. Breast. 2019;48:17–23.
Rocca A, Cortesi P, Cortesi L, et al. Phase II study of liposomal doxorubicin, docetaxel and trastuzumab in combination with metformin as neoadjuvant therapy for HER2-positive breast cancer. Ther Adv Med Oncol. 2021;13:1758835920985632.
Gennari A, Foca F, Zamarchi R, et al. Insulin-like growth factor-1 receptor (IGF-1R) expression on circulating tumor cells (CTCs) and metastatic breast cancer outcome: results from the TransMYME trial. Breast Cancer Res Treat. 2020;181(1):61–8.
Goodwin PJ, Parulekar WR, Gelmon KA, et al. Effect of metformin vs placebo on and metabolic factors in NCIC CTG MA.32. J Natl Cancer Inst. 2015;107(3): djv006.
Goodwin PJ, Chen BE, Gelmon KA, et al. Effect of metformin vs placebo on invasive disease-free survival in patients with breast cancer: the MA.32 randomized clinical trial. JAMA. 2022;327(20):1963–73.
Li L, Jiang L, Wang Y, et al. Combination of metformin and gefitinib as first-line therapy for nondiabetic advanced NSCLC patients with egfr mutations: a randomized, double-blind phase II trial. Clin Cancer Res. 2019;25(23):6967–75.
Chak A, Buttar NS, Foster NR, et al. Metformin does not reduce markers of cell proliferation in esophageal tissues of patients with Barrett’s esophagus. Clin Gastroenterol Hepatol. 2015;13(4):665–72.
Park JJ, Kim BC, Hong SP, et al. The effect of metformin in treatment of adenomas in patients with familial adenomatous polyposis. Cancer Prev Res (Phila). 2021;14(5):563–72.
Zell JA, McLaren CE, Morgan TR, et al. A phase IIa trial of metformin for colorectal cancer risk reduction among individuals with history of colorectal adenomas and elevated body mass index. Cancer Prev Res (Phila). 2020;13(2):203–12.
Bever KM, Borazanci EH, Thompson EA, et al. An exploratory study of metformin with or without rapamycin as maintenance therapy after induction chemotherapy in patients with metastatic pancreatic adenocarcinoma. Oncotarget. 2020;11(21):1929–41.
Braghiroli MI, de Celis Ferrari AC, Pfiffer TE, et al. Phase II trial of metformin and paclitaxel for patients with gemcitabine-refractory advanced adenocarcinoma of the pancreas. Ecancermedicalscience. 2015;9:563.
Kordes S, Pollak MN, Zwinderman AH, et al. Metformin in patients with advanced pancreatic cancer: a double-blind, randomised, placebo-controlled phase 2 trial. Lancet Oncol. 2015;16(7):839–47.
Soliman PT, Zhang Q, Broaddus RR, et al. Prospective evaluation of the molecular effects of metformin on the endometrium in women with newly diagnosed endometrial cancer: a window of opportunity study. Gynecol Oncol. 2016;143(3):466–71.
Bateman NW, Teng PN, Hope E, et al. Jupiter microtubule-associated homolog 1 (JPT1): a predictive and pharmacodynamic biomarker of metformin response in endometrial cancers. Cancer Med. 2020;9(3):1092–103.
Petchsila K, Prueksaritanond N, Insin P, Yanaranop M, Chotikawichean N. Effect of metformin for decreasing proliferative marker in women with endometrial cancer: a randomized double-blind placebo-controlled trial. Asian Pac J Cancer Prev. 2020;21(3):733–41.
Yang BY, Gulinazi Y, Du Y, et al. Metformin plus megestrol acetate compared with megestrol acetate alone as fertility-sparing treatment in patients with atypical endometrial hyperplasia and well-differentiated endometrial cancer: a randomised controlled trial. BJOG. 2020;127(7):848–57.
Janda M, Robledo KP, Gebski V, et al. Complete pathological response following levonorgestrel intrauterine device in clinically stage 1 endometrial adenocarcinoma: results of a randomized clinical trial. Gynecol Oncol. 2021;161(1):143–51.
Broekman KE, Hof MAJ, Touw DJ, et al. Phase I study of metformin in combination with carboplatin/paclitaxel chemotherapy in patients with advanced epithelial ovarian cancer. Invest New Drugs. 2020;38(5):1454–62.
Nguyen MM, Martinez JA, Hsu CH, et al. Bioactivity and prostate tissue distribution of metformin in a preprostatectomy prostate cancer cohort. Eur J Cancer Prev. 2018;27(6):557–62.
Roy S, Malone S, Grimes S, Morgan SC. Impact of concomitant medications on biochemical outcome in localised prostate cancer treated with radiotherapy and androgen deprivation therapy. Clin Oncol (R Coll Radiol). 2021;33(3):181–90.
Mark M, Klingbiel D, Mey U, et al. Impact of addition of metformin to abiraterone in metastatic castration-resistant prostate cancer patients with disease progressing while receiving abiraterone treatment (MetAb-Pro): phase 2 pilot study. Clin Genitourin Cancer. 2019;17(2):e323–8.
Bilusic M, Toney NJ, Donahue RN, et al. A randomized phase 2 study of bicalutamide with or without metformin for biochemical recurrence in overweight or obese prostate cancer patients (BIMET-1). Prostate Cancer Prostatic Dis. 2022;25(4):735–40.
Pujalte Martin M, Borchiellini D, Thamphya B, et al. TAXOMET: a French prospective multicentric randomized phase II study of docetaxel plus metformin versus docetaxel plus placebo in metastatic castration-resistant prostate cancer. Clin Genitourin Cancer. 2021;19(6):501–9.
Skinner H, Hu C, Tsakiridis T, et al. Addition of metformin to concurrent chemoradiation in patients with locally advanced non-small cell lung cancer: the NRG-LU001 phase 2 randomized clinical trial. JAMA Oncol. 2021;7(9):1324–32.
López-Otín C, Galluzzi L, Freije JMP, Madeo F, Kroemer G. Metabolic control of longevity. Cell. 2016;166(4):802–21.
Morgillo F, Sasso FC, Della Corte CM, et al. Synergistic effects of metformin treatment in combination with gefitinib, a selective EGFR tyrosine kinase inhibitor, in LKB1 wild-type NSCLC cell lines. Clin Cancer Res. 2013;19(13):3508–19.
Litchfield LM, Mukherjee A, Eckert MA, et al. Hyperglycemia-induced metabolic compensation inhibits metformin sensitivity in ovarian cancer. Oncotarget. 2015;6(27):23548–60.
Dowling RJO, Niraula S, Stambolic V, Goodwin PJ. Metformin in cancer: translational challenges. J Mol Endocrinol. 2012;48(3):R31–43.
Graham GG, Punt J, Arora M, et al. Clinical pharmacokinetics of metformin. Clin Pharmacokinet. 2011;50(2):81–98.
Hawley SA, Gadalla AE, Olsen GS, Hardie DG. The antidiabetic drug metformin activates the AMP-activated protein kinase cascade via an adenine nucleotide-independent mechanism. Diabetes. 2002;51(8):2420–5.
Schar** NE, Menk AV, Whetstone RD, Zeng X, Delgoffe GM. Efficacy of PD-1 blockade is potentiated by metformin-induced reduction of tumor hypoxia. Cancer Immunol Res. 2017;5(1):9–16.
Lin J, Gill A, Zahm SH, et al. Metformin use and survival after non-small cell lung cancer: a cohort study in the US Military health system. Int J Cancer. 2017;141(2):254–63.
Liu S, Washio J, Sato S, et al. Rewired cellular metabolic profiles in response to metformin under different oxygen and nutrient conditions. Int J Mol Sci. 2022;23(2):989.
Peppicelli S, Toti A, Giannoni E, et al. Metformin is also effective on lactic acidosis-exposed melanoma cells switched to oxidative phosphorylation. Cell Cycle. 2016;15(14):1908–18.
Daugan M, Dufaÿ Wojcicki A, d’Hayer B, Boudy V. Metformin: an anti-diabetic drug to fight cancer. Pharmacol Res. 2016;113(Pt A):675–85.
Dowling RJ, Niraula S, Chang MC, et al. Changes in insulin receptor signaling underlie neoadjuvant metformin administration in breast cancer: a prospective window of opportunity neoadjuvant study. Breast Cancer Res. 2015;17(1):32.
Roy S, Malone S, Grimes S, Morgan SC. Impact of concomitant medications on biochemical outcome in localised prostate cancer treated with radiotherapy and androgen deprivation therapy. Eur Urol. 2021;33(3):181–90.
Rothermundt C, Hayoz S, Templeton AJ, et al. Metformin in chemotherapy-naive castration-resistant prostate cancer: a multicenter phase 2 trial (SAKK 08/09). Clin Genitourin Cancer. 2014;66(3):468–74.
Hu JR, Yeh HC, Mueller NT, et al. Effects of a behavioral weight loss intervention and metformin treatment on serum urate: results from a randomized clinical trial. Nutrients. 2021;13(8):2673.
Mueller NT, Differding MK, Zhang M, et al. Metformin affects gut microbiome composition and function and circulating short-chain fatty acids: a randomized trial. Diabetes Care. 2021;44(7):1462–71.
Tilves C, Yeh HC, Maruthur N, et al. A behavioral weight-loss intervention, but not metformin, decreases a marker of gut barrier permeability: results from the SPIRIT randomized trial. Int J Obes (Lond). 2022;46(3):655–60.
Funding
This work was supported by the National Natural Science Foundation of China (82103240, 12275178, and 82073889), the Science and Technology Commission of Shanghai Municipality (20DZ2270800, 23ZR1438400), Shanghai Key Clinical Specialty, Shanghai Eye Disease Research Center (2022ZZ01003), Cross-disciplinary Research Fund of Shanghai Ninth People's Hospital, Shanghai Jiao Tong University School of Medicine (JYJC202210), and the Innovative research team of high-level local universities in Shanghai (SHSMU-ZDCX20210902, SHSMU-ZDCX20210900).
Author information
Authors and Affiliations
Contributions
AZ, SG and RJ provided direction and guidance throughout the preparation of this manuscript. YH and YZ collected and interpreted the studies and were major contributors to the writing and editing of the manuscript. AZ and YY reviewed and made significant revisions to the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
No potential conflicts of interest were reported by the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Hua, Y., Zheng, Y., Yao, Y. et al. Metformin and cancer hallmarks: shedding new lights on therapeutic repurposing. J Transl Med 21, 403 (2023). https://doi.org/10.1186/s12967-023-04263-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12967-023-04263-8