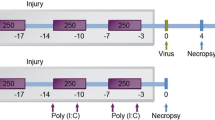
Abstract
Background
IL-33, which is known to induce type 2 immune responses via group 2 innate lymphoid cells, has been reported to contribute to neutrophilic airway inflammation in chronic obstructive pulmonary disease. However, its role in the pathogenesis of emphysema remains unclear.
Methods
We determined the role of interleukin (IL)-33 in the development of emphysema using porcine pancreas elastase (PPE) and cigarette smoke extract (CSE) in mice. First, IL-33−/− mice and wild-type (WT) mice were given PPE intratracheally. The numbers of inflammatory cells, and the levels of cytokines and chemokines in the bronchoalveolar lavage (BAL) fluid and lung homogenates, were analyzed; quantitative morphometry of lung sections was also performed. Second, mice received CSE by intratracheal instillation. Quantitative morphometry of lung sections was then performed again.
Results
Intratracheal instillation of PPE induced emphysematous changes and increased IL-33 levels in the lungs. Compared to WT mice, IL-33−/− mice showed significantly greater PPE-induced emphysematous changes. No differences were observed between IL-33−/− and WT mice in the numbers of macrophages or neutrophils in BAL fluid. The levels of hepatocyte growth factor were lower in the BAL fluid of PPE-treated IL-33−/− mice than WT mice. IL-33−/− mice also showed significantly greater emphysematous changes in the lungs, compared to WT mice, following intratracheal instillation of CSE.
Conclusion
These observations suggest that loss of IL-33 promotes the development of emphysema and may be potentially harmful to patients with COPD.
Similar content being viewed by others
Background
Chronic obstructive pulmonary disease (COPD) is characterized by airflow obstruction that is not fully reversible, leading to respiratory insufficiency and functional disability [1, 36, 37]. In the present study, however, VEGF levels in the lungs were not different between IL-33−/− and WT mice following instillation of PPE or CS; therefore, VEGF may not have been affected in our emphysema models.
This study had some limitations. First, although IL-33-/- mice displayed enhanced emphysematous changes, this only occurred when IL-33 was completely deficient. Treatment with anti-ST2 receptor antibody did not enhance emphysema development, suggesting that partial IL-33 blockade is not sufficient to induce emphysema. Whether blockade of the IL-33-ST2 axis is harmful or beneficial for COPD in the clinical setting remains unclear. Second, treatment with IL-33 did not significantly suppress emphysema development. However, modifications to the dose and timing of IL-33 administration may produce different outcomes. Thus, the effects of IL-33 supplementation for COPD remain unclear. Third, we have not assessed the CS-induced emphysema model, which more closely mimics human emphysema. We have shown that a congenital IL-33 defect might enhance elastase- and CSE-induced emphysema. Collectively, our results indicate that clinical manipulations of the IL-33-ST2 axis may have varying effects on lung structure.
Conclusions
We demonstrated that the loss of IL-33 may enhance the development of emphysema. Our data suggested that although IL-33 may affect CS-induced airway inflammation, complete loss of IL-33 may enhance emphysema and potentially be harmful in patients with COPD.
Abbreviations
- ANOVA:
-
One-way analysis of variance
- BAL:
-
Bronchoalveolar lavage
- COPD:
-
Chronic obstructive pulmonary disease
- CS:
-
Cigarette smoke
- CSE:
-
Cigarette smoke extract
- Cst:
-
Static lung compliance
- CXCL5:
-
C-X-C motif ligand 5
- ELISA:
-
Enzyme-linked immunosorbent assay
- H&E:
-
Hematoxylin and eosin
- HGF:
-
Hepatocyte growth factor
- IL:
-
Interleukin
- ILC2:
-
Group 2 innate lymphoid cell
- KC:
-
Keratinocyte chemoattractant
- Lm:
-
Mean linear intercept
- MCP-1:
-
Chemoattractant protein-1
- MIP-2:
-
Macrophage inflammatory protein-2
- NK:
-
Natural killer
- MMP-9:
-
Matrix metalloproteinase-9
- PBS:
-
Phosphate-buffered saline
- PPE:
-
Porcine pancreas elastase
- TIMP-1:
-
Tissue inhibitor of metalloproteinase-1
- TNF-α:
-
Tumor necrosis factor-alpha
- VEGF:
-
Vascular endothelial growth factor
- WT:
-
Wild-type
References
Vestbo J, Hurd SS, Agusti AG, Jones PW, Vogelmeier C, Anzueto A, Barnes PJ, Fabbri LM, Martinez FJ, Nishimura M, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am J Respir Crit Care Med. 2013;187:347–65.
**ong H, Huang Q, Shuai T, Zhu L, Zhang C, Zhang M, Wang Y, Liu J. Assessment of comorbidities and prognosis in patients with COPD diagnosed with the fixed ratio and the lower limit of normal: a systematic review and meta-analysis. Respir Res. 2020;21:189.
Calvi C, Podowski M, Lopez-Mercado A, Metzger S, Misono K, Malinina A, Dikeman D, Poonyagariyon H, Ynalvez L, Derakhshandeh R, et al. Hepatocyte growth factor, a determinant of airspace homeostasis in the murine lung. PLoS Genet. 2013;9:e1003228.
Hegab AE, Kubo H, Yamaya M, Asada M, He M, Fu**o N, Mizuno S, Nakamura T. Intranasal HGF administration ameliorates the physiologic and morphologic changes in lung emphysema. Mol Ther. 2008;16:1417–26.
Kennelly H, Mahon BP, English K. Human mesenchymal stromal cells exert HGF dependent cytoprotective effects in a human relevant pre-clinical model of COPD. Sci Rep. 2016;6:38207.
Kasahara Y, Tuder RM, Taraseviciene-Stewart L, Le Cras TD, Abman S, Hirth PK, Waltenberger J, Voelkel NF. Inhibition of VEGF receptors causes lung cell apoptosis and emphysema. J Clin Invest. 2000;106:1311–9.
Yoshimoto T, Matsushita K. Innate-type and acquired-type allergy regulated by IL-33. Allergol Int. 2014;63(Suppl 1):3–11.
Kondo Y, Yoshimoto T, Yasuda K, Futatsugi-Yumikura S, Morimoto M, Hayashi N, Hoshino T, Fujimoto J, Nakanishi K. Administration of IL-33 induces airway hyperresponsiveness and goblet cell hyperplasia in the lungs in the absence of adaptive immune system. Int Immunol. 2008;20:791–800.
Wu H, Yang S, Wu X, Zhao J, Zhao J, Ning Q, Xu Y, **e J. Interleukin-33/ST2 signaling promotes production of interleukin-6 and interleukin-8 in systemic inflammation in cigarette smoke-induced chronic obstructive pulmonary disease mice. Biochem Biophys Res Commun. 2014;450:110–6.
Kearley J, Silver JS, Sanden C, Liu Z, Berlin AA, White N, Mori M, Pham TH, Ward CK, Criner GJ, et al. Cigarette smoke silences innate lymphoid cell function and facilitates an exacerbated type I interleukin-33-dependent response to infection. Immunity. 2015;42:566–79.
Qiu C, Li Y, Li M, Li M, Liu X, McSharry C, Xu D. Anti-interleukin-33 inhibits cigarette smoke-induced lung inflammation in mice. Immunology. 2013;138:76–82.
Yasuda K, Muto T, Kawagoe T, Matsumoto M, Sasaki Y, Matsushita K, Taki Y, Futatsugi-Yumikura S, Tsutsui H, Ishii KJ, et al. Contribution of IL-33-activated type II innate lymphoid cells to pulmonary eosinophilia in intestinal nematode-infected mice. Proc Natl Acad Sci USA. 2012;109:3451–6.
Fujii U, Miyahara N, Taniguchi A, Waseda K, Morichika D, Kurimoto E, Koga H, Kataoka M, Gelfand EW, Cua DJ, et al. IL-23 is essential for the development of elastase-induced pulmonary inflammation and emphysema. Am J Respir Cell Mol Biol. 2016;55:697–707.
Morichika D, Miyahara N, Fujii U, Taniguchi A, Oda N, Senoo S, Kataoka M, Tanimoto M, Kakuta H, Kiura K, et al. A retinoid X receptor partial agonist attenuates pulmonary emphysema and airway inflammation. Respir Res. 2019;20:2.
Oda N, Miyahara N, Taniguchi A, Morichika D, Senoo S, Fujii U, Itano J, Gion Y, Kiura K, Kanehiro A, Maeda Y. Requirement for neuropeptide Y in the development of type 2 responses and allergen-induced airway hyperresponsiveness and inflammation. Am J Physiol Lung Cell Mol Physiol. 2019;316:L407-l417.
Dunnill MS. Evaluation of a simple method of sampling the lung for quantitative histological analysis. Thorax. 1964;19:443–8.
Hanaoka M, Droma Y, Chen Y, Agatsuma T, Kitaguchi Y, Voelkel NF, Kubo K. Carbocisteine protects against emphysema induced by cigarette smoke extract in rats. Chest. 2011;139:1101–8.
Robbesom AA, Versteeg EM, Veerkamp JH, van Krieken JH, Bulten HJ, Smits HT, Willems LN, van Herwaarden CL, Dekhuijzen PN, van Kuppevelt TH. Morphological quantification of emphysema in small human lung specimens: comparison of methods and relation with clinical data. Mod Pathol. 2003;16:1–7.
Fujii U, Miyahara N, Taniguchi A, Oda N, Morichika D, Murakami E, Nakayama H, Waseda K, Kataoka M, Kakuta H, et al. Effect of a retinoid X receptor partial agonist on airway inflammation and hyperresponsiveness in a murine model of asthma. Respir Res. 2017;18:23.
Kurimoto E, Miyahara N, Kanehiro A, Waseda K, Taniguchi A, Ikeda G, Koga H, Nishimori H, Tanimoto Y, Kataoka M, et al. IL-17A is essential to the development of elastase-induced pulmonary inflammation and emphysema in mice. Respir Res. 2013;14:5.
Kabata H, Moro K, Fukunaga K, Suzuki Y, Miyata J, Masaki K, Betsuyaku T, Koyasu S, Asano K. Thymic stromal lymphopoietin induces corticosteroid resistance in natural helper cells during airway inflammation. Nat Commun. 2013;4:1–10.
Taniguchi A, Miyahara N, Waseda K, Kurimoto E, Fujii U, Tanimoto Y, Kataoka M, Yamamoto Y, Gelfand EW, Yamamoto H, et al. Contrasting roles for the receptor for advanced glycation end-products on structural cells in allergic airway inflammation vs. airway hyperresponsiveness. Am J Physiol Lung Cell Mol Physiol. 2015;309:L789-800.
Churg A, Zhou S, Wright JL. Series “matrix metalloproteinases in lung health and disease”: Matrix metalloproteinases in COPD. Eur Respir J. 2012;39:197–209.
Drube S, Heink S, Walter S, Lohn T, Grusser M, Gerbaulet A, Berod L, Schons J, Dudeck A, Freitag J, et al. The receptor tyrosine kinase c-Kit controls IL-33 receptor signaling in mast cells. Blood. 2010;115:3899–906.
Pushparaj PN, Tay HK, H’Ng SC, Pitman N, Xu D, McKenzie A, Liew FY. Retraction. The cytokine interleukin-33 mediates anaphylactic shock. Proc Natl Acad Sci USA. 2012;109:13877.
Xu D, Jiang HR, Li Y, Pushparaj PN, Kurowska-Stolarska M, Leung BP, Mu R, Tay HK, McKenzie AN, McInnes IB, et al. IL-33 exacerbates autoantibody-induced arthritis. J Immunol. 2010;184:2620–6.
Mahmutovic Persson I, Menzel M, Ramu S, Cerps S, Akbarshahi H, Uller L. IL-1beta mediates lung neutrophilia and IL-33 expression in a mouse model of viral-induced asthma exacerbation. Respir Res. 2018;19:16.
Smithgall MD, Comeau MR, Yoon BR, Kaufman D, Armitage R, Smith DE. IL-33 amplifies both Th1- and Th2-type responses through its activity on human basophils, allergen-reactive Th2 cells, iNKT and NK cells. Int Immunol. 2008;20:1019–30.
Bourgeois E, Van LP, Samson M, Diem S, Barra A, Roga S, Gombert JM, Schneider E, Dy M, Gourdy P, et al. The pro-Th2 cytokine IL-33 directly interacts with invariant NKT and NK cells to induce IFN-gamma production. Eur J Immunol. 2009;39:1046–55.
MacNee W. Pathogenesis of chronic obstructive pulmonary disease. Proc Am Thorac Soc. 2005;2:258–66.
Carter RI, Ungurs MJ, Mumford RA, Stockley RA. Aalpha-Val360: a marker of neutrophil elastase and COPD disease activity. Eur Respir J. 2013;41:31–8.
Foronjy R, Nkyimbeng T, Wallace A, Thankachen J, Okada Y, Lemaitre V, D’Armiento J. Transgenic expression of matrix metalloproteinase-9 causes adult-onset emphysema in mice associated with the loss of alveolar elastin. Am J Physiol Lung Cell Mol Physiol. 2008;294:L1149-1157.
Mercer PF, Shute JK, Bhowmik A, Donaldson GC, Wedzicha JA, Warner JA. MMP-9, TIMP-1 and inflammatory cells in sputum from COPD patients during exacerbation. Respir Res. 2005;6:151.
Kollet O, Shivtiel S, Chen YQ, Suriawinata J, Thung SN, Dabeva MD, Kahn J, Spiegel A, Dar A, Samira S, et al. HGF, SDF-1, and MMP-9 are involved in stress-induced human CD34+ stem cell recruitment to the liver. J Clin Invest. 2003;112:160–9.
Lee JH, Hailey KL, Vitorino SA, Jennings PA, Bigby TD, Breen EC. Cigarette smoke triggers IL-33-associated inflammation in a model of late-stage chronic obstructive pulmonary disease. Am J Respir Cell Mol Biol. 2019;61:567–74.
Jakkula M, Le Cras TD, Gebb S, Hirth KP, Tuder RM, Voelkel NF, Abman SH. Inhibition of angiogenesis decreases alveolarization in the develo** rat lung. Am J Physiol Lung Cell Mol Physiol. 2000;279:L600-607.
Le Cras TD, Markham NE, Tuder RM, Voelkel NF, Abman SH. Treatment of newborn rats with a VEGF receptor inhibitor causes pulmonary hypertension and abnormal lung structure. Am J Physiol Lung Cell Mol Physiol. 2002;283:L555-562.
Acknowledgements
The authors gratefully thank Eri Ando, Suzuka Yukimaru, and Mao Yabuuchi for their technical assistance.
Funding
The work reported here was supported in part by Novartis Pharma Research Grants 2019 (to N. Miyahara).
Author information
Authors and Affiliations
Contributions
Conceived and designed the research: NM, DM. Performed the experiments: DM, NM, AT, UF, NO, SS. Analyzed the data: DM, NM, AT, UF, NO, KK, NM. Interpreted the results of the experiments: DM, AK, YK, MY, MH, TS, SA, KK, YM, NM. Prepared the figures: DM, NO, AT, NM. Drafted the manuscript: DM, AT, NM. Approved the final version of the manuscript: DM, AT, NO, UF, SS, JI, AK, YK, MY, MH, TS, SA, KK, YM, NM. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was approved by the Institutional Animal Care and Use Committee of Okayama University Medical School. Approved number is OKU-2014526.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1.
Treatment with anti-ST2 antibody in PPE-instilled mice. The mice received intraperitoneal injection of anti-ST2 antibody or IgG antibody on days -1, 1, 2, 4, 7, and 10 following instillation of PBS or PPE. BAL and lung morphometric measurements were performed on day 21 as described in the Materials and Methods. (A) Cell composition in BAL fluid. (B) Lm values. Data are shown as the mean ± SEM (n = 8 in each group). PBS/IgG: PBS-instilled mice treated with IgG. PPE/IgG: PPE-instilled mice treated with IgG. PPE/anti-ST2: PPE-instilled mice treated with anti-ST2 antibody. *P < 0.05 compared to PBS/PBS; There were no significant differences between PPE/IgG and PPE/anti-ST2.
Additional file 2.
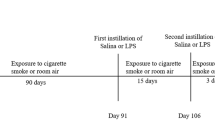
Morphometric assessment of emphysema induced by intraperitoneal administration of cigarette smoke extract (CSE). The experimental protocol.
Additional file 3.
(B) Representative images of H&E-stained lung tissue (magnification: ×200).
Additional file 4.
(C) Lm values. WT PBS: WT mice treated with PBS. IL-33-/- PBS: IL-33-/- mice treated with PBS. WT CSE: WT mice treated with CSE. IL-33-/- CSE: IL-33-/- mice treated with CSE. The results for each group are shown as the mean ± SEM; n = 6–9 in each group. *P < 0.05 compared to WT PBS; **P < 0.05 compared to IL-33−/− PBS; #P < 0.05 compared to WT PPE.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Morichika, D., Taniguchi, A., Oda, N. et al. Loss of IL-33 enhances elastase-induced and cigarette smoke extract-induced emphysema in mice. Respir Res 22, 150 (2021). https://doi.org/10.1186/s12931-021-01705-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12931-021-01705-z