Abstract
Background
Growing concern about the emergence of antibiotic resistance is compelling the pharmaceutical industry to search for new antimicrobial agents. The availability of genome sequences has enabled the development of computational mining as an important tool in the discovery of natural products with antibiotic effect.
Results
NRPPUR (Non-Ribosomal Peptide and Polyketide Urmite) is a new bioinformatic tool that was created to detect polyketides and non-ribosomal peptide gene clusters (PKS and NRPS) in bacterial genomes using the rpsBlast program. The NRPPUR database was constructed locally by assembling all 3505 available sequences of NRPS-PKS that have been identified by in silico approaches to date, with 164 Biosynthetic Gene Clusters (BGCs) derived from the published literature that have demonstrated antimicrobial activity in vitro. The in silico analysis of 49 intestinal human bacterial genomes using the NRPPUR made it possible to identify 91 BGCs including 89 clusters that had never previously been described. On average, intestinal human bacterial genomes devote nearly 0.8% (±1.4% s.d.) of their genome to NRPS/PKS biosynthesis, with Bacillus vallismortis, Streptomyces massiliensis and Bacillus subtilis genomes apportioning 8.4, 3.6 and 3.15% of their genomes, respectively. When using the cross-streak method, S. massiliensis displayed antibacterial activity against many Gram-positive and negative bacteria including methicillin-resistant Staphylococcus aureus (MRSA).
Conclusions
NRPPUR has proven to be a very useful tool for the primary in silico selection of species with potential antimicrobial activity and human microbiota could be the future source of new antimicrobial discoveries. Further exploration of this and other ecological niches, coupled with high-throughput antibacterial activity screening should be envisaged.
Similar content being viewed by others
Background
Antibiotics have achieved major advances in medicine and surgery, saving patients’ lives and extending the expected human lifespan [1]. Following the golden era when natural antibiotics were discovered and prescribed in 1925–1950, the chemistry era followed in the 1970s, with synthetic tweaking to improve activity. However, in 2000, the resistance era, largely due to the overuse and misuse of these medications, began [2, 3]. This coincided with the development of new technologies such as the manipulation of recombinant DNA and the high-throughput synthesis of chemicals that has given rise to hopes of drug discoveries other than antibiotics [4]. Although the high-throughput biochemical screening of large collections of syntheses has provided some interesting leads, the complexity and diversity of these molecules has been insufficient to provide the same level of bioactivity as found in naturally occurring antibiotics. It has been suggested that the coexistence of microbes with other microbes and fungi in the environment leads to selection of the most potent targets so that the best source of new antibiotics are compounds naturally produced by microorganisms [5]. It has therefore been recommended that natural products are revisited as an alternative to synthetic collections following the methods in the “golden age of the discovery of antibiotics” that screened microbial cell extracts from soil to find new antibiotic scaffolds. It has been also recommended that new technologies are embraced to overcome problems of compound discovery. Thus, the exploration of genome sequences of microorganisms and data from metagenomics of the microbial dark matter- microorganisms that have resisted to easy cultivation in the laboratory [6] has revealed a very large spectrum of potential for secondary metabolites with potential antibiotic functions [7].
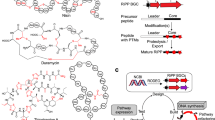
Microbial secondary metabolites are organic compounds that are not directly involved in primary growth and development, but rather have auxiliary functions including defense and communication [8]. Natural antimicrobial products consist mainly of two groups i) bacteriocins [9] where biosynthesis is carried out conventionally via ribosome, and ii) polyketides (PKS) and non-ribosomal peptides (NRPS) where biosynthesis is ribosome independent. The atypical biosynthesis of NRPS and PKS known as “thiotempling” is supported by a multi-enzymatic, multi-domain synthases NRPSs and PKSs, respectively that add amino-acid monomers for NRPS and acyl Coenzyme A for PKS products. The primary sequence of the peptide product is determined by the sequential arrangement of active sites called modules within NRPSs and modular PKSs. These modules contain multiple functional domains that are necessary for catalyzing each condensation and chain elongation or modification reaction [10,11,12]. Genes encoding biosynthetic enzymes for the synthesis of these secondary metabolites are typically co-localized on the chromosome and are referred to as “biosynthetic gene clusters” (BGCs). Since the first elucidation of the PKS gene cluster for erythromycin in the early 1990s [13, 14], many gene clusters responsible for the biosynthesis of NRPS and PKS have been reported and deposited in International Nucleotide Sequence Database Collection (INSDC) entries (DDBJ/GenBank/EMBL) [15]. In addition, the community-driven website developed many specialized pieces of software such as Antismash [16,17,18] and Streptome DB [19] that enabled the detection of NRPS and PKS [20,21,22,23,24] in a wide range of microorganisms such as Bacteria, Fungi, Archaea and Eukarya. The general principle behind in silico mining consists of using a library of enzymes/protein domains commonly observed in secondary metabolite biosynthetic pathways to identify homologues in the genome sequences of the organisms of interest. For this task, sequence based comparison software, such as BLAST [25] or DIAMOND [42] where NRP-PK are produced at a high rate and which may constitute one of the defensive mechanisms used by microorganisms to survive [43, 44]. Indeed, while most natural products were isolated from environmental microbial strains recently, Staphylococcus lugdunensis, a human commensal, was described to have a protective role against Staphylococcus aureus colonization in the nasal human microbiota. This may be mediated by the antibacterial non-ribosomal peptide, Lugdunin [45]. Thus, human microbiota could be the future source of new antimicrobial discoveries, and further exploration of this ecological niche, coupled with newer technologies such as cell-free assays and high-throughput screening, should be envisaged. Further transcriptomic and gene silencing approaches can confirm the implication of NRPS/PKS clusters in the observed antimicrobial potency. Moreover, our results showed that Bacillus, Paenibacillus and Streptomyces genomes were outlier in the number of NRPS/PKS clusters. While Streptomyces species are known to be prolific producers of antibiotics and other natural products, the high rate of NRPS/PKS in Bacillus spp. is likely to reflect their abundance in the microbiota and their particular ecological role involving multiple interactions with cohabiting microbes. Given that, these species should be vigorously pursued for new antimicrobial product discoveries.
Conclusions
This work is a pioneering study to search for new NRPS-PKS naturally produced by the human digestive microbiota and showing potent antibiotic activity in vitro. The NRPPUR database integrates the latest experimentally verified information and provides standardized domain descriptions related to the gene clusters. Our database serves as a useful reference to facilitate research and development related to secondary metabolite types NRPS and PKS with potential antibiotic activity. A web interface (http://www.mediterranee-infection.com/article.php?laref=955&titre=nrppur-database-) has been developed allowing rpsBlast analyses to be performed to search for NRPS-PKS.
Abbreviations
- ACP:
-
Acyl Carrier Protein
- A-domain:
-
Adenylation domain
- AT:
-
Acyltransferase
- BGCs:
-
Biosynthetic Gene Clusters
- C-domain:
-
Condensation domain
- KS:
-
Ketosynthase
- MRSA:
-
Methicillin-Resistant Staphylococcus aureus
- NRPPUR:
-
Non-Ribosomal Peptide and Polyketide Urmite
- NRPS:
-
Non-Ribosomal Peptide gene clusters
- NRPs:
-
Non-Ribosomal Peptides
- PCP-domain:
-
Peptidyl Carrier domain
- PKs:
-
Polyketides
- PKS:
-
Polyketides gene clusters
- RAST:
-
Rapid Annotation using Subsystems Technology
- RPS-BLAST:
-
Reverse Position-Specific Blast
References
Gould IM, Bal AM. New antibiotic agents in the pipeline and how they can overcome microbial resistance. Virulence. 2013;4(2):185–91.
Boucher HW, Talbot GH, Bradley JS, Edwards JE, Gilbert D, Rice LB, et al. Bad bugs, no drugs: no ESKAPE! An update from the Infectious Diseases Society of America. Clin Infect Dis Off Publ Infect Dis Soc Am. 2009;48(1):1):1–12.
Alekshun MN, Levy SB. Molecular mechanisms of antibacterial multidrug resistance. Cell. 2007;128(6):1037–50.
Brown ED, Wright GD. Antibacterial drug discovery in the resistance era. Nature. 2016;529(7586):336–43.
Lewis K. Antibiotics: Recover the lost art of drug discovery. Nature. 2012;485(7399):439–40.
Lock C. Mining the microbial dark matter. Nature. 2015;522:270–3.
Drissi F, Buffet S, Raoult D, Merhej V. Common occurrence of antibacterial agents in human intestinal microbiota. Front Microbiol. 2015;6:441.
Schwarzer D, Finking R, Marahiel MA. Nonribosomal peptides: from genes to products. Nat Prod Rep. 2003;20(3):275–87.
Ahmad V, Khan MS, Jamal QMS, Alzohairy MA, Al Karaawi MA, Siddiqui MU. Antimicrobial potential of bacteriocins: in therapy, agriculture and food preservation. Int J Antimicrob Agents. 2016.
Weissman KJ. The structural biology of biosynthetic megaenzymes. Nat Chem biol. Sep. 2014;11(9):660–70.
Jenke-Kodama H, Dittmann E. Bioinformatic perspectives on NRPS/PKS megasynthases: advances and challenges. Nat Prod Rep. 2009;26(7):874–83.
Khosla C, Kapur S, Cane DE. Revisiting the modularity of modular polyketide synthases. Curr Opin Chem Biol. 2009;13:135–43.
Cortes J, Haydock SF, Roberts GA, Bevitt DJ, Leadlay PF. An unusually large multifunctional polypeptide in the erythromycin-producing polyketide synthase of Saccharopolyspora erythraea. Nature. 1990;348:176–8.
Donadio S, Staver MJ, McAlpine JB, Swanson SJ, Katz L. Modular organization of genes required for complex polyketide biosynthesis. Science. 1991;252:675–9.
Benson DA, Karsch-Mizrachi I, Clark K, Lipman DJ, Ostell J, Sayers EW. GenBank. Nucleic Acids Res. 2012;40:D48–53.
Medema MH, Blin K, Cimermancic P, de Jager V, Zakrzewski P, Fischbach MA, Weber T, Takano E, Breitling R. AntiSMASH: rapid identification, annotation and analysis of secondary metabolite biosynthesis gene clusters in bacterial and fungal genome sequences. Nucleic Acids Res. 2011;39:W339–46.
Blin K, Medema MH, Kazempour D, Fischbach MA, Breitling R, Takano E, Weber T. Nucleic Acids Res. 2013;41:W204–12.
Weber T, Blin K, Duddela S, Krug D, Kim HU, Bruccoleri R, Lee SY, Fischbach M, Müller R, Wohlleben W, Breitling R, Takano E, Medema MH. Nucleic Acids Res. 2015;43:W237–43.
Lucas X, Senger C, Erxleben A, Grüning BA, Döring K, Mosch J, et al. StreptomeDB: a resource for natural compounds isolated from Streptomyces species. Nucleic Acids Res. 2013;41(Database issue):D1130–6.
Starcevic A, Zucko J, Simunkovic J, Long PF, Cullum J, Hranueli D. ClustScan: an integrated program package for the semi-automatic annotation of modular biosynthetic gene clusters and in silico prediction of novel chemical structures. Nucleic Acids Res. 2008;36:6882–92.
Weber T, Rausch C, Lopez P, Hoof I, Gaykova V, Huson DH, Wohlleben W. CLUSEAN: a computer-based framework for the automated analysis of bacterial secondary metabolite biosynthetic gene clusters. J Biotechnol. 2009;140:13–7.
Conway KR, Boddy CN. ClusterMine360: a database of microbial PKS/NRPS biosynthesis. Nucleic Acids Res. 2013;41(Database issue):D402–7.
Ichikawa N, Sasagawa M, Yamamoto M, Komaki H, Yoshida Y, Yamazaki S, Fujita N. DoBISCUIT: a database of secondary metabolite biosynthetic gene clusters. Nucleic Acids Res. 2013;41(Database issue):D408–14.
Skinnider MA, Dejong CA, Rees PN, Johnston CW, Li H, Webster AL, Wyatt MA, Magarvey NA. Genomes to natural products PRediction informatics for secondary Metabolomes (PRISM). Nucleic Acids Res. 2015;43(20):9645–62.
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ. Basic local alignment search tool. J Mol Biol. 1990;215(3):403–10.
Buchfink B, **e C, Huson DH. Fast and sensitive protein alignment using DIAMOND. Nat Methods. 2015;12(1):59–60.
Finn RD, Clements J, Eddy SR. HMMER web server: interactive sequence similarity searching. Nucleic Acids Res. 2011;39(Web Server issue):W29–37.
Buysse JM. The role of genomics in antibacterial target discovery. Curr Med Chem. 2001;8(14):1713–26.
Selzer PM, Brutsche S, Wiesner P, Schmid P, Müllner H. Target-based drug discovery for the development of novel antiinfectives. Int J Med Microbiol. 2000;290(2):191–201.
Lagier JC, Khelaifia S, Alou MT, Ndongo S, Dione N, Hugon P, et al. Culture of previously uncultured members of the human gut microbiota by culturomics. Nat Microbiol. 2016;1:16203.
Aziz RK, Bartels D, Best AA, DeJongh M, Disz T, Edwards RA, Formsma K, et al. The RAST server: rapid annotations using subsystems technology. BMC Genomics. 2008;9:75.
Wang H, Fewer DP, Holm L, Rouhiainen L, Sivonen K. Atlas of nonribosomal peptide and polyketide biosynthetic pathways reveals common occurrence of nonmodular enzymes. Proc Natl Acad Sci U S A. 2014;111(25):9259–64.
Marchler-Bauer A, Bo Y, Han L, He J, Lanczycki CJ, Lu S, et al. CDD/SPARCLE: functional classification of proteins via subfamily domain architectures. Nucleic Acids Res. 2017;45(D1):D200–3.
Katoh K, Standley DM. MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol Biol Evol. 2013;30:772–80.
Williston EH, Zia-Walrath P, Youmans GP. Plate methods for testing antibiotic activity of actinomycetes against virulent human type tubercle bacilli. J Bacteriol. 1947;54:563–8.
Lin TP, Chen CL, Chang LK, Tschen JS, Liu ST. Functional and transcriptional analyses of a fengycin synthetase gene, fenC, from Bacillus subtilis. J Bacteriol. 1999;181(16):5060–7.
Nakano MM, Magnuson R, Myers A, Curry J, Grossman AD, Zuber P. srfA is an operon required for surfactin production, competence development, and efficient sporulation in Bacillus subtilis. J Bacteriol. 1991;173(5):1770–8.
Pfleiderer A, Lagier JC, Armougom F, Robert C, Vialettes B, Raoult D. Culturomics identified 11 new bacterial species from a single anorexia nervosa stool sample. Eur J Clin Microbiol Infect Dis. 2013;32(11):1471–81.
Wright GD. The antibiotic resistome: the nexus of chemical and genetic diversity. Nat Rev Microbiol. 2007;5(3):175–86 Review.
Ansari MZ, Yadav G, Gokhale RS, Mohanty D. NRPS-PKS: a knowledge-based resource for analysis of NRPS/PKS megasynthases. Nucleic Acids Res. 2004;32(Web Server):W405–13.
Lertcanawanichakul M, Sawangnop S. A comparison of Two Methods used for measuring the antagonistic activity of Bacillus Species. Walailak J Sci Tech. 2008;5:161–71.
Ley RE, Peterson DA, Gordon JI. Ecological and evolutionary forces sha** microbial diversity in the human intestine. Cell. 2006;124(4):837–48.
Hibbing ME, Fuqua C, Parsek MR, Peterson SB. Bacterial competition: surviving and thriving in the microbial jungle. Nat Rev Microbiol. 2010;8(1):15–25.
Prasad S, Manasa P, Buddhi S, Singh SM, Shivaji S. Antagonistic interaction networks among bacteria from a cold soil environment. FEMS Microbiol Ecol. 2011;78(2):376–85.
Zipperer A, Konnerth MC, Laux C, Berscheid A, Janek D, Weidenmaier C, et al. Human commensals producing a novel antibiotic impair pathogen colonization. Nature. 2016;535(7613):511–6.
Acknowledgements
We thank Pierre Pontarotti who provided expertise that greatly assisted the research and Sylvain Buffet for setting up the NRPPUR server.
Funding
This study was financially supported by IHU Méditerranée Infection Foundation. VM was supported by a Chairs of Excellence program from the Centre National de la Recherche Scientifique (CNRS). The funders had no role in study design, data collection and interpretation or the decision to submit the work.
Availability of data and materials
The NRPPUR can be accessed at http://www.mediterranee-infection.com/article.php?laref=955&titre=nrppur-database-.
Author information
Authors and Affiliations
Contributions
VM, JMR and DR conceived of the project. SF and VM designed the study, processed the experimental data, performed the analysis and carried out the interpretation of data. AR implemented and benchmarked the code with contribution from OC.SF wrote the paper with help of VM. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Additional file
Additional file 1:
Figure S1. A Venn diagram of PKS, NRPS, and hybrid gene-cluster numbers in NRPUR database. The gene-cluster numbers of the total, bacteria, eukarya and archaea are shown in black, red, green, and grey, respectively. Table S1. The 164 NRPS-PKS BGC sequences identified from the literature search resulting from experimental data with validated antibacterial activity. Table S2. Summary of NRPS and PKS gene clusters found in the genomes of bacteria from the gut. Figure S2. Distribution of NRPS, PKS, and hybrid gene clusters in bacteria from the gut a) in all studied bacteria, b) in the studied phyla (PDF 313 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Fritz, S., Rajaonison, A., Chabrol, O. et al. Full-length title: NRPPUR database search and in vitro analysis identify an NRPS-PKS biosynthetic gene cluster with a potential antibiotic effect. BMC Bioinformatics 19, 463 (2018). https://doi.org/10.1186/s12859-018-2479-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12859-018-2479-5