Abstract
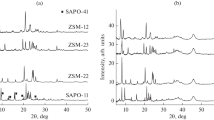
The authors study the effect of the content of zeolite ZSM-22 (15–70 wt %) in a support on the physicochemical properties of Pt/ZSM-22–Al2O3 catalysts. The dependence of the yield and composition of sunflower oil hydrodeoxygenation products on these catalysts on the temperature (310–340°C), pressure (3‒5 MPa), and weight hourly space velocity (WHSV) (0.8–3 h−1) is determined. The possibility is shown of the full hydrodeoxygenation of sunflower oil with the formation of C5+ hydrocarbons containing up to 72% of iso-paraffins with yields of 75–79 wt %.








Similar content being viewed by others
REFERENCES
Huber, G.W., Iborra, S., and Corma, A., Chem. Rev., 2006, vol. 106, no. 9, pp. 4044–4098. https://doi.org/10.1021/cr068360d
Sivasamy, A., Cheah, K.Y., Fornasiero, P., Kemausuor, F., Zinoviev, S., and Miertus, S., ChemSusChem, 2009, vol. 2, no. 4, pp. 278–300. https://doi.org/10.1002/cssc.200800253
Choudhary, T.V. and Phillips, C.B., Appl. Catal., A, 2011, vol. 397, nos. 1–2, pp. 1–12. https://doi.org/10.1016/j.apcata.2011.02.025
Khan, S., Kay Lup, A.N., Queshi, K.M., Abnisa, F., Wan Daud, W.M.A., Patah, M.F.A., Anal. Appl. Pyrolysis, 2019, vol. 140, pp. 1–24. https://doi.org/10.1016/j.jaap.2019.03.005
Douvartzides, S.L., Charisiou, N.D., Papageridis, K.N., and Goula, M.A., Energies, 2019, vol. 12, no. 5, article no. 809. https://doi.org/10.3390/en12050809
Smirnova, M.Yu., Kikhtyanin, O.V., Rubanov, A.E., Trusov, L.I., and Echevskii, G.V., Catal. Ind., 2013, vol. 5, no. 3, pp. 253–259. https://doi.org/10.1134/S2070050413030112
Pérez-Cisneros, E.S., Sales-Cruz, M., Lobo-Oehmichen, R., and Viveros-García, T., Comput. Chem. Eng., 2017, vol. 105, pp. 105–122. https://doi.org/10.1016/j.compchemeng.2017.01.018
Herskowitz, M., Landau, M.V., Reizner, Y., and Ber-ger, D., Fuel, 2013, vol. 111, pp. 157–164. https://doi.org/10.1016/j.fuel.2013.04.044
Wang, C., Liu, Q., Liu, X., Yan, L., Luo, C., Wang, L., Wang, B., and Tian, Z., Chin. J. Catal., 2013, vol. 34, no. 6, pp. 1128–1138. https://doi.org/10.1016/S1872-2067(11)60524-X
Wang, C., Tian, Z., Wang, L., Xu, R., Liu, Q., Qu, W., Ma, H., and Wang, B., ChemSusChem, 2012, vol. 5, no. 10, pp. 1974–1983. https://doi.org/10.1002/cssc.201200219
Wang, C., Liu, Q., Song, J., Li, W., Li, P., Xu, R., Ma, H., and Tian, Z., Catal. Today, 2014, vol. 234, pp. 153–160. https://doi.org/10.1016/j.cattod.2014.02.011
Zhang, M., Chen, Y., Wang, L., Zhang, Q., Tsang, C.-W., and Liang, C., Ind. Eng. Chem. Res., 2016, vol. 55, no. 21, pp. 6069–6078. https://doi.org/10.1021/acs.iecr.6b01163
Martens, J.A., Verboekend, D., Thomas, K., Vanbutsele, G., Pérez-Ramírez, J., and Gilson, J.-P., Catal. Today, 2013, vols. 218–219, pp. 135–142. https://doi.org/10.1016/j.cattod.2013.03.041
Hancsók, J., Krár, M., Magyar, S., Boda, L., Holló, A., and Kalló, D., Microporous Mesoporous Mater., 2007, vol. 101, nos. 1–2, pp. 148–152. https://doi.org/10.1016/j.micromeso.2006.12.012
Nepomnyashchiy, A.A., Buluchevskiy, E.A., Lavrenov, A.V., Yurpalov, V.L., Gulyaeva, T.I., Leont’eva, N.N., and Talzi, V.P., Russ. J. Appl. Chem., 2017, vol. 90, no. 12, pp. 1944–1952. https://doi.org/10.1134/S1070427217120084
Parmar, S., Pant, K.K., John, M., Kumar, K., Pai, S.M., and Newalkar, B.L., Energy Fuels, 2015, vol. 29, no. 2, pp. 1066–1075. https://doi.org/10.1021/ef502591q
Chi, K., Zhao, Z., Tian, Z., Hu, S., Yan, L., Li, T., Wang, B., Meng, X., Gao, S., Tan, M., and Liu, Y., Pet. Sci., 2013, vol. 10, no. 2, pp. 242–250. https://doi.org/10.1007/s12182-013-0273-6
Liu, S., Ren, J., Zhu, S., Zhang, H., Lv, E., Xu, J., and Li, Y.-W., J. Catal., 2015, vol. 330, pp. 485–496. https://doi.org/10.1016/j.jcat.2015.07.027
Liu, S., Ren, J., Zhang, H., Lv, E., Yang, Y., and Li, Y.-W., J. Catal., 2016, vol. 335, pp. 11–23. https://doi.org/10.1016/j.jcat.2015.12.009
Kim, S.K., Han, J.Y., Lee, H.S., Yum, T., Kim, Y., and Kim, J., Appl. Energy, 2014, vol. 116, pp. 199–205. https://doi.org/10.1016/j.apenergy.2013.11.062
Alcalá, R., Mavrikakis, M., and Dumesic, J.A., J. Catal., 2003, vol. 218, no. 1, pp. 178–190. https://doi.org/10.1016/S0021-9517(03)00090-3
Chen, N., Gong, S., Shirai, H., Watanabe, T., and Qian, E.W., Appl. Catal., A, 2013, vol. 466, pp. 105–115. https://doi.org/10.1016/j.apcata.2013.06.034
Sankaranarayanan, T.M., Banu, M., Pandurangan, A., and Sivasanker, S., Bioresour. Technol., 2011, vol. 102, no. 22, pp. 10717–10723. https://doi.org/10.1016/j.biortech.2011.08.127
Anand, M. and Sinha, A.K., Bioresour. Technol., 2012, vol. 126, pp. 148–155. https://doi.org/10.1016/j.biortech.2012.08.105
Guzman, A., Torres, J.E., Prada, L.P., and Nuñez, M.L., Catal. Today, 2010, vol. 156, nos. 1–2, pp. 38–43. https://doi.org/10.1016/j.cattod.2009.11.015
ACKNOWLEDGMENTS
The authors are grateful to engineers L.A. Buluchevskaya and E.N. Kudrya, junior researcher I.V. Muromtsev, researchers R.R. Izmailov and A.V. Babenko, and first-category electrical engineer S.N. Evdokimov for their work in studying the catalysts and the composition of liquid products in the sunflower oil hydrodeoxygenation process.
Our physicochemical studies were performed on equipment at the Boreskov Institute’s National Center for the Investigation of Catalysts.
Funding
This work was supported by the Russian Federation Ministry of Science and Higher Education within the governmental assignment for Boreskov Institute of Catalysis (project AAAA-A21-121011890075-1).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interests.
Additional information
Translated by E. Glushachenkova
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nepomnyashchii, A.A., Saibulina, E.R., Buluchevskiy, E.A. et al. Physicochemical and Catalytic Properties of Bifunctional Catalysts with Different Contents of Zeolite ZSM-22 in the Hydrodeoxygenation of Sunflower Oil. Catal. Ind. 16, 161–169 (2024). https://doi.org/10.1134/S2070050424700065
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2070050424700065