Abstract
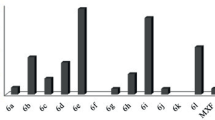
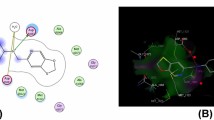
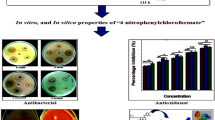
A novel series of 2-[(2-R-4-fluoro-3-(methylsulfanyl)phenyl]-4,5-dihydro-1,3-oxazoles were designed, synthesized, and evaluated for their antibacterial and antioxidant activities against different microorganisms. The synthesized compounds were characterized by different spectral techniques like 1H and 13C NMR, IR, and mass spectroscopy. Derivatives with R = vinyl, isopropyl, H, and 4-chlorophenyl exhibited excellent antibacterial properties against S. aureus, P. aeruginosa, and E. coli. 4-Methylphenyl and 4-methoxyphenyl analogs showed better activity against all the tested bacteria. Compounds with R = 4-methoxyphenyl and isopropyl displayed the highest DPPH radical scavenging activity with IC50 values of 24.39 and 25.29 µg/mL, respectively, and those with R = vinyl and isopropyl demonstrated excellent antioxidant activity according to the H2O2 radical scavenging assay with IC50 values of 20.24 and 18.76 µg/mL, respectively. Furthermore, molecular docking analysis revealed the highest binding energies, ΔG = –5.67, –5.33, and –5.13 kcal/mol for compounds with R = isopropyl, phenyl, and tert-butoxy, respectively, and hydrogen bonding interactions with the following amino acid residues of DNA gyrase subunit A of E. coli (PDB code: 4CKL): Arg15 (3.07 Å), Thr87 (2.28 Å), Gly16, Glu85, and Ile14.


Similar content being viewed by others
REFERENCES
White, D.G. and McDermott, P.F., J. Dairy Sci., 2001, vol. 84, p. E151. https://doi.org/10.3168/jds.S0022-0302(01)70209-3
Bax, R., Mullan, N., and Verhoef, J., Int. J. Antimicrob. Agents, 2000, vol. 16, p. 51. https://doi.org/10.1016/S0924-8579(00)00189-8
Heinemann, J.A., Ankenbauer, R.G., and Amabile Cuevas, C.F., Drug Discovery Today, 2000, vol. 5, p. 195. https://doi.org/10.1016/S1359-6446(00)01483-5
Walsh, C., Nature, 2000, vol. 406, p. 775. https://doi.org/10.1038/35021219
Ritter, T.K. and Wong, C.H., Angew. Chem., Int. Ed., 2001, vol. 40, p. 3508. https://doi.org/10.1002/1521-3773(20011001)40
Fun, H.-K., Hemamalini, M., Asiri, A.M., Khan, S.A., and Khan, K.A., Acta Crystallogr., Sect. E, 2010, vol. 66, p. o773. https://doi.org/10.1107/S1600536810008160
Kang, Y.Y., Shin, K.J., Yoo, K.H., Seo, K.J., Hong, C.Y., Lee, C.S., Park, S.Y., Kim, D.J., and Park, S.W., Bioorg. Med. Chem. Lett., 2000, vol. 10, p. 95. https://doi.org/10.1016/S0960-894X(99)00646-0
Mishra, A., Jain, S.K., and Asthana, J.G., Orient. J. Chem., 1998, vol. 14, p. 151. http://www.orientjchem.org/?p=21769
Conti, P., Dallanoce, C., Amici, M.D., Micheli, C.D., and Klotz, K.N., Bioorg. Med. Chem., 1998, vol. 6, p. 401. https://doi.org/10.1016/S0968-0896(97)10051-7
Ko, D.H., Maponya, M.F., Khalil, M.A., Oriaku, E.T., and You, Z., J. Med. Chem. Res., 1998, vol. 8, p. 313.
Srivastava, S., Bajpai, L.K., Batra, S., Bhaduri, A.P., Maikhuri, J.P., Gupta, G., and Dhar, J.D., Bioorg. Med. Chem., 1999, vol. 7, p. 2607. https://doi.org/10.1016/S0968-0896(99)00188-1
Wipf, P., Fletcher, J.M., and Scarone, L., Tetrahedron Lett., 2005, vol. 46, p. 5463. https://doi.org/10.1016/j.tetlet.2005.06.063
Razavi, H., Powers, E.T., Purkey, H.E., Adamski-Werner, S.L., Chiang, K.P., Dendle, M.T.A., and Kelly, J.W., Bioorg. Med. Chem. Lett., 2005, vol. 15, p. 1075. https://doi.org/10.1016/j.bmcl.2004.12.022
Lv, P.C., Li, H.Q., Xue, J.Y., Shi, L., and Zhu, H.L., Eur. J. Med. Chem., 2009, vol. 44, p. 908. https://doi.org/10.1016/j.ejmech.2008.01.013
Cao, P., Huang, X.-F., Ding, H., Ge, H.-M., Li, H.-Q., Ruan, B.-F., and Zhu, H.L., Chem. Biodiversity, 2007, vol. 4, p. 881. https://doi.org/10.1002/cbdv.200790075
Lebel, H., Parmentier, M., Leogane, O., Ross, K., and Spitz, C., Tetrahedron, 2012, vol. 68, p. 3396. https://doi.org/10.1016/j.tet.2012.02.044
**, Y. and Du, D.M., Tetrahedron, 2012, vol. 68, p. 3633. https://doi.org/10.1016/j.tet.2012.02.078
Pridgen, L.N., Killmer, L.B., and Webb, R.L., J. Org. Chem., 1982, vol. 47, p. 1985. https://doi.org/10.1021/jo00132a001
Deng, T. and Cai, C., J. Fluorine Chem., 2013, vol. 156, p. 183. https://doi.org/10.1016/j.jfluchem.2013.09.014
Guiu, E., Claver, C., and Castillon, S., J. Organomet. Chem., 2004, vol. 689, p. 1911. https://doi.org/10.1016/j.jorganchem.2004.03.015
Zurabyan, S.E., Antonenko, T.S., and Khorlin, A.Y., Carbohydr. Res., 1970, vol. 15, p. 21. https://doi.org/10.1016/S0008-6215(00)80289-X
Stephane, F., Valerie, T., Christian, B., and Thierry, B., Tetrahedron, 2003, vol. 59, p. 773. https://doi.org/10.1016/S0040-4020(02)01593-4
Clinical and Laboratory Standard Institute. CLSI document M07-A9: Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically, Approved Standard, 9th ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2012.
Mollica, A., Macedonio, G., Stefanucci, A., Costante, R., Carradori, S., Cataldi, V., Di Giulio, M., Cellini, L., Silvestri, R., Giordano, C., Scipioni, A., Morosetti, S., Punzi, P., and Mirzaie, S., Lett. Drug Des. Discovery, 2017, vol. 14, p. 1. https://doi.org/10.2174/1570180814666170213161341
Blois, M.S., Nature, 1958, vol. 181, p. 1199. https://doi.org/10.1038/1811199a0
Sroka, Z. and Cisowski, W., Food Chem. Toxicol., 2003, vol. 41, p. 753. https://doi.org/10.1016/s0278-6915(02)00329-0
Morris, G.M., Huey, R., Lindstrom, W., Sanner, M.F., Belew, R.K., Goodsell, D.S., and Olson, A.J., J. Comput. Chem., 2009, vol. 30, p. 2785. https://doi.org/10.1002/jcc.21256
Harder, E., Damm, W., Maple, J., Wu, C., Reboul, M., **ang, J.Y., Wang, L., Lupyan, D., Dahlgren, M.K., Knight, J.L., Kaus, J.W., Cerutti, D.S., Krilov, G., Jorgensen, W.L., Abel, R., and Friesner, R.A., J. Chem. Theory Comput., 2015, vol. 12, p. 281. https://doi.org/10.1021/acs.jctc.5b00864
ACKNOWLEDGMENTS
A.T. Rao is thankful to CCST, IST, JNTUH University, Hyderabad, for providing laboratory and other facilities, CBT, IST, JNTUH for biological studies, and Osmania University for providing molecular modeling techniques.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest.
Supplementary information
Rights and permissions
About this article
Cite this article
Rao, A.T., Naveen, K., Mutyalanaidu, G. et al. Design, Synthesis, and Characterization of Novel Substituted Dihydrooxazoles: Biological Evaluation and Molecular Docking. Russ J Org Chem 58, 125–135 (2022). https://doi.org/10.1134/S1070428022010183
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428022010183