Abstract
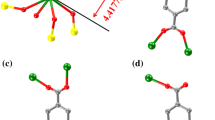
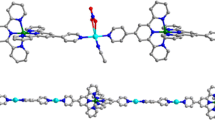
New coordination polymers with lanthanide ions [Ln(Fur)3(H2O)x]n⋅Solv (Ln = Gd (I), Sm (II); Fur = 2-furoic acid anion; x = 2 (I), 3 (II); Solv = MeCN (I)) are synthesized. The structures of the synthesized compounds are determined by X-ray diffraction (CIF files CCDC nos. 2130014 (I) and 2130015 (II)). The coordination environment of the complexing agent (LnO8) corresponds to a distorted square antiprism (I) or a distorted triangular dodecahedron (II). Complexes I and II represent polymeric chains in which the Fur– anions perform the bridging function. The crystal lattice is stabilized by intra- and intermolecular hydrogen bonds between the coordinated water molecules, acid anions, and solvate molecules. The study of the thermal behavior of compound I by simultaneous thermal analysis in an argon atmosphere shows a low stability of the complex: its decomposition starts at 69°С, and the organic moiety destructs gradually without pronounced thermal effects. According to the data of X-ray photoelectron spectroscopy, gadolinium(III) oxide is the final product of thermal decomposition.






Similar content being viewed by others
REFERENCES
Peters, J.A., Nieuwenhuizen, M.S., and Raber, D.J., J. Magn. Reson., 1985, vol. 65, p. 417.
Piguet, C. and Geraldes, C.F., Handbook on the Physics and Chemistry of Rare Earths, Amsterdam: Elsevier, 2003.
Chebotar’, I.V., Novikov, I.A., Subbot, A.M., and Mayanskii, N.A., Sovremennye Tekhnologii v Meditsine, 2017, vol. 9, p. 23.
Wang, Y., Li, X., Wang, T., et al., Inorg. Chem., 2010, vol. 49, p. 969.
Babailov, S., Sens. Actuators, B, 2017, vol. 251, p. 108.
Koehler, J. and Meiler, J., Prog. Nucl. Magn. Reson. Spectrosc., 2011, vol. 59, p. 360.
Peters, J.A., Sinnema, A., Kieboom, P.G., and Bekkum, H., J. Am. Chem. Soc., 1985, vol. 107, p. 12.
Mashkovskii, M.D. Lekarstvennye sredstva (Pharmaceuticals), Moscow: Novaya volna, 2005.
Makota, H. and Gomi, T., Radiol. Contrast Ag. Radiopharm., 2015, vol. 37, p. 583.
Krylov, V.V., Drozdovskii, B.Ya., and Tsyb, A.F., Uspekhi Sovremennogo Estestvoznaniya, 2003, no. 10, p. 73.
Bartolomé. E., Bartolomé, J., Arauzo, A., et al., J. Mater. Chem. С, 2018, vol. 6, p. 5286.
Gusev, A., Kiskin, M., Lutsenko, I., et al., J. Lumin., 2021, vol. 238, p. 118305.
Li, X., **, L., Lu, S., et al., J. Mol. Struct., 2002, vol. 604, p. 65.
Yin, M. and Sun, J., J. Alloy. Comp., 2004, vol. 381, p. 50.
Lutsenko, I.A., Baravikov, D.E., Kiskin, M.A., et al., Russ. J. Coord. Chem., 2020, vol. 46, p. 411. https://doi.org/10.31857/S0132344X20060055
Lutsenko, I.A., Yambulatov, D.S., Kiskin, M.A., et al., Russ. J. Coord. Chem., 2020, vol. 46, p. 787. https://doi.org/10.1134/S1070328420120040
Lutsenko, I.A., Yambulatov, D.S., Kiskin, M.A., et al., Chem. Select., 2020, vol. 5, p. 11837.
Lutsenko, I.A., Kiskin, M.A., Koshenskova, K.A., et al., Russ. Chem. Bull., 2021, vol. 70, p. 463.
Uvarova, M.A., Lutsenko, I.A., Kiskin, M.A., et al., Polyhedron, 2021, vol. 203, p. 115241.
Lutsenko, I.A., Baravikov, D.E., Koshenskova, K.A., et al., RSC Adv., 2022, vol. 12, p. 5173.
Lutsenko, I.A., Nikiforova, M.E., Kosheskova, K.A., et al., Russ. J. Coord. Chem., 2021, vol. 47, p. 879. https://doi.org/10.31857/S0132344X22020049
Fomina, I.G., Kiskin, M.A., Martynov, A.G., et al., Russ. J. Inorg. Chem., 2004, vol. 49, p. 1369.
Sheldrick, G.M., Acta Crystallogr., Sect. C: Struct. Chem., 2015, vol. 71, p. 3.
Dolomanov, O.V., Bourhis, L.J., Gildea, R.J., et al., J. Appl. Crystallogr., 2009, vol. 42, p. 339.
Jordi, C., Eliseo, R., and Santiago, A., Organometallics, 2005, vol. 24, p. 1556.
ACKNOWLEDGMENTS
XRD studies were carried out using the equipment of the Center of Molecular Structure Investigation at the Nesmeyanov Institute of Organoelement Compounds (Russian Academy of Sciences). Elemental analysis, IR spectroscopy, and STA were carried out and the SEM images of the micromorphology and the energy dispersive spectra were obtained using the equipment of the Center for Collective Use of Physical Methods of Investigation at the Kurnakov Institute of General and Inorganic Chemistry (Russian Academy of Sciences).
Funding
This work was supported by the Ministry of Science and Higher Education of the Russian Federation in terms of the state assignment of the Kurnakov Institute of General and Inorganic Chemistry (Russian Academy of Sciences).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
The issue is dedicated to the 70th birthday of Academician V.I. Ovcharenko
Translated by E. Yablonskaya
Rights and permissions
About this article
Cite this article
Uvarova, M.A., Lutsenko, I.A., Nikiforova, M.E. et al. Gd(III) and Sm(III) 1D Coordination Polymers with 2-Furoic Acid: Synthesis, Structures, and Thermal Behavior. Russ J Coord Chem 48, 457–463 (2022). https://doi.org/10.1134/S1070328422080073
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328422080073