Abstract
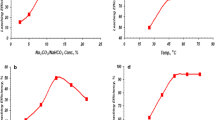
The efficiency of using ammonium carbonate lixiviant for extracting uranium (U), copper (Cu), and zinc (Zn) from Abu Thor gibbsite bearing shale ore material, which assays 0.11% U, 0.56% Cu, and 2.8% Zn, was evaluated. 99.6% of U, 98.1% of Cu, and 87.5% of Zn were leached under the optimum conditions: ammonium carbonate concentration 12%, solid/liquid ratio 1/3, leaching time 3.5 h, leaching temperature 85°C, and addition of 0.8 mL of hydrogen peroxide as oxidant. Amberlite IRA400 anion-exchange resin was used for U separation at pH 8.5, while direct precipitation using Na2S solution was then applied for Cu and Zn recovery. Finally, a technical flowsheet was suggested for the preparation of U, Cu, and Zn compounds.










Similar content being viewed by others
REFERENCES
El Aassy, I.E., Ahmed, F.Y., El Shamy, A.S., Shata, A.E., Ammar, F.A., Fouad, A.A., and Abd Rabboh, A., Abstracts of Papers, 1st Conf. of Applied Geology, Helwan Univ., 2006.
Amer, T.E., Abd El Wahab, G.M., Abdellah, W.M., Al Shami, A.S., and El Shahat, M.F., Bull. Fac. Sci. Zagazig Univ., 2017, vol. 2017, no. 1, pp. 82–97.
Amer, T.E., PhD Thesis, Fac. Sci., Cairo Univ. (Egypt), 1997.
Abdel Monem, A.A., El-Assay, I.E., Hegab, O.A., and El-Fayoumy, I.F., J. Sediments, J. Egypt, 1997, vol. 5, pp. 117–132.
Dabbour, G.M. and Mahdy, M.A., Abstracts of Papers, 4th Conf. on Nuclear Science and Applications, Cairo (Egypt), 1988, pp. 230–237.
Ghonaim, A.E., El-Hazek, N.T., Ahmed, F.Y., and Abdel Fattah, N.A., Al-Azhar Bull. Sci., 2004, vol. 51, pp. 29–42.
Lottering, M.J., Lorenzen, L., Phala, N.S., Smit, J.T., and Schalkwyk, G.A.C., Miner. Eng., 2008, vol. 21, pp. 16–22.
Guettaf, H., Becis, A., Ferhat, K., Hanou, K., Bouchiha, D., Yakoubi, K., and Ferrad, F., Phys. Procedia, 2009, vol. 2, pp. 765–771.
Kim, J.S., Chung, K.W., Lee, H.I., Yoon, H.S., and Kumar, J.R., J. Radioanal. Nucl. Chem., 2014, vol. 299, pp. 81–87.
Gajda, D., Kiegiel, K., Zakrzewska-Koltuniewicz, G., Chajduk, E., Bartosiewicz, I., and Wolkowicz, S., J. Radioanal. Nucl. Chem., 2015, vol. 303, no. 1, pp. 521–529. https://doi.org/10.1007/s10967-014-3362-0.
Abu-Khoziem, H.A., Abdel Wahab, S., Rezik, A., Khalid, E., and Abdellah, W.M., Int. J. Environ. Anal. Chem., 2020, vol. 10, pp. 1–17.
Ramazanova, R.À., Seraya, N.V., Samoilov, V.I., Daumova, G.K., and Azbanbayev, E.M., Metallurgist, 2020, vol. 64, no. 2, pp. 73–78.
Abdel Wahab, S., Rezik, A., Abu Khoziem, H., Khalid, E., and Abdellah, W.M., Euro-Mediterr. J. Environ. Integr., 2019, vol. 4, no. 19, pp. 1–11. https://doi.org/10.1007/s41207-019-0106-0
Orabi, A.H., El-Sheikh, E.M., Saleh, W.H., El-Saied, F.A., El-Gendy, H.S., and Ismaiel, D., J. Radiat. Res. Appl. Sci., 2019, vol. 12, no. 1, pp. 10–23.
Suri, A.K., Ghosh, S.K., and Padmanabhan, N.P.H., Int. Symp. on Uranium Raw Material for the Nuclear Fuel Cycle, Vienna, 2009, pp. 231–246.
Khan, Y., Shah, S. and Siddiq, M., J. Chem. Soc. Pak., 2012, vol. 34, pp. 826–840.
Li, H., Zhao, W., Duan, Z., Yong, W., and Qiang, D., Uranium Mining Metall., 2006, vol. 25, no. 1, pp. 9–14.
Abhilash, B.D. and Pandey, B.D., Miner. Process. Extractive Metall. Rev., 2013, vol. 34, no. 2, pp. 81–113.
Afifi, S.Y., Abo-Aly, M.M., and Elashry, S.M., Arab J. Nucl. Sci. Appl., 2017, vol. 50, no. 4, pp. 213–28.
Ma, A., Zheng, X., Li, S., Wang, Y., and Zhu, S., Roy. Soc. Open Sci., 2018, vol. 5, ID 180660.
Hunter, E., PhD Thesis, Faculty and the Board of Trustees of the Colorado School of Mines (Mining and Earth Systems Engineering), 2013.
Carmen, C. and Kunin, R., React. Polym., Ion Exch., Sorbents, 1986, vol. 4, no. 2, pp. 77–89.
Lunt, D., Boshof, P., Boylett, M., and El-Ansary, Z., J. South Afr. Inst. Min. Metall., 2007, vol. 107, pp. 420–426.
Santos, E.A. and Ladeira, A.C., Environ. Sci. Technol., 2011, vol. 45, no. 8, pp. 3591–3597.
Leavitt, J.J., Howe, K.J., and Cabaniss, S.E., Appl. Geochem., 2011, vol. 26, pp. 2019–2026.
Rosenberg, E., Pinson, G. and Tsosie, R., Johnson Matthey Technol. Rev., 2016, vol. 60, no. 1, p. 59.
Quinn, J.E., Sedger, D.S., Brennan, A.T., Ring, R.J., and Soldenhoff, K., Hydrometallurgy, 2020, vol. 194, ID 105360.
Bhattacharyya, D., Jumawan, A.B., Jr., and Grieves, R.B., Sep. Sci. Technol., 1979, vol. 14, no. 5, pp. 441–452.
Alvarez, M.T., Crespo, C., and Mattiasson, B., Chemosphere, 2007, vol. 66, no. 9, pp. 1677–1683.
Choi, Y., Peter, K., and Jacques, M., Patent EP2086885A1, 2007.
Abdel Wahab, G.M., PhD Thesis, Ain Shams Univ., Faculty of Science, Chemical Department, 2008.
Amer, T.E., El-Hazek, M.N., Abdel Fattah, W.M., and El-Shahat, M.F., J. Isotope Radiat. Res., 2009, vol. 41, no. 4, pp. 1163–1177.
Prokkola, H., Tuulia, N. E. and Lassi, U., J. Chem. Eng., 2020, vol. 4, no. 3, p. 51.
Pohi, A., J. Water, Air Soil Pollut., 2020, vol. 231, no. 10, ID 503. https://doi.org/10.1007/s11270-020-04863-w
Yatim, S.M., Zainuddin, N.A., Mokhtar, N.S., Syahjidan, H.N., and Kamusur, S.H., Abstract of Papers, Int. Conf. on Chemical and Material Engineering (ICCME), 2020.
Shapiro, L. and Brannock, W.W., U.S. Geol. Survey Bull., 1962, p. 114.
Marczenko, Z., Spectrophotometric Determination of Elements, New York: Wiley, 2000.
Lazo, P., Cullaj, A., Dogjani, S., and Lubonja, F., Asian J. Chem., 2000, vol. 12, no. 3, pp. 732–738.
Mathew, K.J., Burger, S., Ogt, S.V., Mason, P.M., and Narayanan, U.I., Abstracts of Papers, The Eighth Int. Conf. on Methods and Applications of Radioanalytical Chemistry MARC VIII, Kaailua-Kona, Hawaii, 2009.
Ali, H.H., Ph.D. Thesis, Cairo (Egypt), Faculty of Science, Al-Azhar Univ., 2016.
Kweto, B., Groot, D.R., Stassen, E., Suthiram, J., and Zeevaart, J.R., J. Radioanal. Nucl. Chem., 2014, vol. 302, pp. 131–137.
Abdellah, W.M., Radiochemistry, 2020, vol. 62, no. 3, pp. 347–358. https://doi.org/10.1134/S106636222003007
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Zaki, D.I. Recovery of Uranium, Copper, and Zinc from Abu Thor Gibbsite Bearing Shale Ore Material, Abu Zeneima Area, Southwestern Sinai, Egypt. Radiochemistry 63, 724–733 (2021). https://doi.org/10.1134/S1066362221060035
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1066362221060035