Abstract
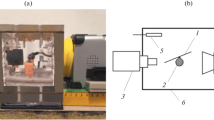
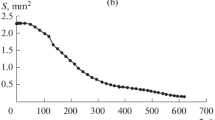
Experimental data have been presented on the evaporation of water–alcohol solution droplets with different concentrations. The droplets have been suspended by a thread and applied onto a planar surface. The dynamics of variations in the geometric parameters of the evaporating water–alcohol solution droplets has been studied with the use of high-speed microphotography. Infrared thermography has been employed to confirm the three-stage variation in the surface temperature of the evaporating droplets, namely, an initial dramatic decrease in the temperature, the stage of a constant temperature, and its smooth increase up to the ambient air temperature. The experimental data have shown an essential influence of the solution concentration on the droplet evaporation process. The higher the ethanol concentration in a droplet, the closer the character of variations in the surface temperature and geometric parameters of the droplet to the variations in the same parameters of an alcohol droplet.









Similar content being viewed by others
REFERENCES
Chandra, S., Di Marzo, M., Qiao, Y.M., and Tartarini, P., Fire Saf. J., 1996, vol. 27, p. 141.
Sefiane, K., Tadrist, L., and Douglas, M., Int. J. Heat Mass Transfer, 2003, vol. 46, p. 4527.
Cheng, A.K.H., Soolaman, D.M., and Yu, H.Z., J. Phys. Chem. B, 2006, vol. 110, p. 11267.
Sefiane, K., David, S., and Shanahan, M.E.R., J. Phys. Chem. B, 2008, vol. 112, p. 11317.
Shi, L., Shen, P., Zhang, D., Lin, Q., and Jiang, Q., Surf. Interface Anal., 2009, vol. 41, p. 951.
Saverchenko, V.I., Fisenko, S.P., and Khodyko, Yu.A., Colloid J., 2015, vol. 77, p. 71.
Kuznetsov, G.V., Feoktistov, D.V., and Orlova, E.G., Teplofiz. Aeromekh., 2016, vol. 23, p. 17.
Kuchma, A.E., Esipova, N.E., Mikheev, A.A., Shchekin, A.K., and Itskov, S.V., Colloid J., 2017, vol. 79, p. 779.
Liu, C., Bonaccurso, E., and Butt, H.J., Phys. Chem. Chem. Phys., 2008, vol. 10, p. 7150.
Oztürk, T. and Erbil, H.Y., Colloids Surf. A, 2018, vol. 553, p. 327.
David, S., Sefiane, K., and Tadrist, L., Colloids Surf. A, 2007, vol. 298, p. 108.
Dunn, G.J., Wilson, S.K., Duffy, B.R., David, S., and Sefiane, K., J. Fluid Mech., 2009, vol. 623, p. 329.
Bazargan, V. and Stoeber, B., Phys. Rev. E: Stat. Phys., Plasmas, Fluids, Relat. Interdiscip. Top., 2016, vol. 94, p. 033103.
Borodulin, V.Y., Letushko, V.N., Nizovtsev, M.I., and Sterlyagov, A.N., MATEC Web of Conferences, 2017, vol. 115, p. 08005.
Han, K., Song, G., Ma, X., and Yang, B., Appl. Therm. Eng., 2016, vol. 101, p. 568.
Terekhov, V.I. and Shishkin, N.E., Polzunovsk. Vestn., 2010, no. 1, p. 55.
Sefiane, K., Moffat, J.R., Matar, O.K., and Craster, R.V., Appl. Phys. Lett., 2008, vol. 93, p. 074103.
Hamamoto, Y., Christy, J.R.E., and Sefiane, K., J. Therm. Sci. Technol., 2012, vol. 7, p. 425.
Nakoryakov, V.E., Misyura, S.Y., and Elistratov, L., J. Eng. Thermophys., 2013, vol. 22, p. 1.
Bochkareva, E.M., Terekhov, V.V., Nazarov, A.D., and Miskiv, N.B., J. Phys.: Conf. Ser., 2017, vol. 891, p. 012010.
Brutin, D., Sobac, B., Rigollet, F., and Le Niliot, C., Exp. Therm. Fluid Sci., 2011, vol. 35, p. 521.
Fedorets, A.A., Dombrovsky, L.A., and Medvedev, D.N., JETP Lett., 2015, vol. 102, p. 452.
Borodulin, V.Y., Letushko, V.N., Nizovtsev, M.I., and Sterlyagov, A.N., Int. J. Heat Mass Transfer, 2017, vol. 109, p. 609.
Kuchma, A.E., Shchekin, A.K., Esipova, N.E., Tat’yanenko, D.V., Itskov, S.V., and Savin, A.V., Colloid J., 2017, vol. 79, p. 353.
Sterlyagov, A.N., Letushko, V.N., Nizovtsev, M.I., and Borodulin, V.Yu., J. Phys.: Conf. Ser., 2018, vol. 1105, p. 012068.
Zolotarev, V.I. and Demin, V.A., Opt. Spektrosk., 1977, vol. 43, p. 271.
Brutin, D., Zhu, Z.Q., Rahli, O., **e, J.C., Liu, Q.S., and Tadrist, L., Microgravity Sci. Technol., 2010, vol. 22, p. 387.
Lebedev-Stepanov, P.V., Vvedenie v samosborku ansamblei nanochastits (An Introduction to Self-Assembly of Nanoparticle Ensembles), Moscow: NIYaU MIFI, 2012.
Fuks, N.A., Rost i isparenie kapel’ v gazoobraznoi srede (Droplet Growth and Evaporation in Gaseous Medium), Moscow: Akad. Nauk SSSR, 1958.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by L. Tkachenko
Rights and permissions
About this article
Cite this article
Borodulin, V.Y., Letushko, V.N., Nizovtsev, M.I. et al. The Experimental Study of Evaporation of Water–Alcohol Solution Droplets. Colloid J 81, 219–225 (2019). https://doi.org/10.1134/S1061933X19030049
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061933X19030049