Abstract
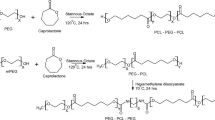
A series of biodegradable poly(ε-caprolactone)–poly(ethylene glycol)–poly(ε-caprolactone) (PCL–PEG–PCL) (PCEC) triblock copolymers had been successfully synthesized by ring-opening polymerization of ε-caprolactone initiated by PEG, which were characterized by proton nuclear magnetic resonance analysis (1H NMR), gel permeation chromatography (GPC), differential scanning calorimetry (DSC) and Fourier transform infrared spectroscopy (FTIR). The gel–sol phase transition diagram of synthesized copolymers was recorded using test tube inverting method. PCECs could form injectable solutions via self-assembly and automatically turned into non-flowing gels at physiological temperatures. Their aqueous solution displayed special gel–sol transition behavior with temperature increasing from 0 to 60°C, when the polymer concentration was above corresponding critical gel concentration (CGC). In vitro release experiments at pH 7.4 showed that after 24 h at least approximately 75–80% of doxorubicin (DOX) can be protected from enzyme or hydrolytic degradation. Therefore, thermosensitive PCECs have many advantages, such as simple drug formulation, no organic solvent, a sustained drug release behavior.








Similar content being viewed by others
REFERENCES
M. K. Nguyen and D. S. Lee, Macromol. Biosci. 10, 563 (2010).
S. W. Choi, S. Y. Choi, B. Jeong, S. W. Kim, and D. S. Lee, J. Polym. Sci., Part A: Polym. Chem. 37, 2207 (1999).
E. Ruel-Gariépy and J. C. Leroux, Eur. J. Pharm. Biopharm. 58, 409 (2004).
B. Jeong, Y. H. Bae, and S. W. Kim, Macromolecules 32, 7064 (1999).
M. L. Gou, C. Y. Gong, J. Zhang, X. H. Wang, X. H. Wang, Y. C. Gu, G. Guo, L. J. Chen, F. Luo, X. Zhao, Y. Q. Wei, and Z. Y. Qian, J. Biomed. Mater. Res., Part A 93, 219 (2010).
G. Bonacucina, M. Cespi, G. Mencarelli, G. Giorgioni, and G. F. Palmieri, Polymers 3, 779 (2011).
G. Ma, B. Miao, and C. Song, J. Appl. Polym. Sci. 116, 1985 (2010).
L. Piao, Z. Dai, M. Deng, X. Chen, and X. **g, Polymer 44, 2025 (2003).
A. Eatemadi, H. Daraee, H. T. Aiyelabegan, B. Negahdari, B. Rajeian, and N. Zarghami, Biomed. Pharmacother. 84, 1915 (2016).
W. J. Jia, Y. C. Gu, M. Gou, M. Dai, X. Li, B. Kan, J. L. Yang, Q. F. Song, Y. Q. Wei, and Z. Y. Qian, Drug Delivery 15, 409 (2008).
L. Yu and J. Ding, Chem. Soc. Rev. 37, 1473 (2008).
X. Wei, X. Lv, Q. Zhao, and L. Qiu, Acta Biomater. 9, 6953 (2013).
M. Boffito, P. Sirianni, A. M. D. Rienzo, and V. Chiono, J. Biomed. Mater. Res., Part A 103, 1276 (2015).
Ö. G. Kocabay and O. Ismail, Rev. Roum. Chim. 63, 1157 (2018).
J. Fu, X. Lv, and L. Qiu, RSC Adv. 5, 37451 (2015).
C. Y. Gong, S. Shi, L. Wu, M. L. Gou, Q. Q. Yin, Q. F. Guo, P. W. Dong, F. Zhang, F. Luo, X. Zhao, Y. Q. Wei, and Z. Y. Qian, Acta Biomater. 5, 3358 (2009).
N. V. Cuong, Y. T. Chen, and M. F. Hsieh, Biomed. Eng.: Appl., Basis, Commun. 25, 1340009 (2013).
W. **ao, J. Hou, J. Ma, B. Yu, J. Ren, W. **, J. Wu, D. Zheng, and K. Fan, Arch. Pharm. Res. (2014). https://doi.org/10.1007/s12272-014-0485-3
N. M. Dinan, F. Atyabi, M. R. Rouini, M. Amini, A. A. Golabchifar, and R. Dinarvand, Mater. Sci. Eng., C 39, 47 (2014).
C. Y. Gong, P. W. Dong, S. Shi, S. Z. Fu, J. L. Yang, G. Guo, X. Zhao, Y. Q. Wei, and Z. Y. Qian, J. Pharm. Sci. 98, 3707 (2009).
A. Eatemadi, M. Darabi, L. Afraidooni, N. Zarghami, H. Daraee, L. Eskandari, H. Mellatyar, and A. Akbarzadeh, Artif. Cells, Nanomed., Biotechnol., 44, 1008 (2016).
L. Barghi, D. Asgari, J. Barar, and H. Valizadeh, Adv. Pharm. Bull. 5, 51 (2015).
Y. Hu, L. Zhang, Y. Cao, H. Ge, X. Jiang, and C. Yang, Biomacromolecules 5, 1756 (2004).
S. Zamani and S. Khoee, Polymer 53, 5723 (2012).
A. M. Lapi, A. Altomare, M. Alderighi, A. Corti, A. Dessy, F. Chiellini, A. Solimando, and R. Solaro, J. Polym. Sci., Part A: Polym. Chem. 51, 3800 (2013).
C. B. Liu, C. Y. Gong, M. J. Huang, J. W. Wang, Y. F. Pan, Y. D. Zhang, G. Z. Li, M. L. Gou, K. Wang, M. J. Tu, Y. Q. Wei, and Z. Y. Qian, J. Biomed. Mater. Res., Part B 84, 165 (2008).
C. Y. Gong, Z. Y. Qian, C. B. Liu, M. J. Huang, Y. C. Gu, Y. J. Wen, B. Kan, K. Wang, M. Dai, X. Y. Li, M. L. Gou, M. J. Tu, and Y. Q. Wei, Smart Mater. Struct. 16, 927 (2007).
T. Q. M. Tran, M. F. Hsieh, K. L. Chang, Q. H. Pho, V. C. Nguyen, C. Y. Cheng, and C. M. Huang, Polymers 8, 1 (2016).
F. Guo, D. Guo, W. Zhang, Q. Yan, Y. Yang, W. Hong, and G. Yang, Eur. J. Pharm. Sci. 99, 328 (2017).
E. Khodaverdi, M. Gharechahi, M. Alibolandi, F. S. M. Tekie, B. Z. Khashyarmanesh, and F. Hadizadeh, Int. J. Pharm. Investig. 6, 78 (2016).
T. H. A. Nguyen and V. C. Nguyen, Adv. Nat. Sci. Nanosci. Nanotechnol. 1, 25012 (2010).
L. Barghi, D. Asgari, J. Barar, A. Nakhlband, and H. Valizadeh, Asian Pac. J. Cancer Prev. 15, 10281 (2014).
B. Bogdanov, A. Vidts, A. Van Den Bulcke, R. Verbeeck, and E. Schacht, Polymer 39, 1631 (1998).
W. Zhu, W. **e, X. Tong, and Z. Shen, Eur. Polym. J. 43, 3522 (2007).
L. Yu, G. Chang, H. Zhang, and J. Ding, J. Polym. Sci., Part A: Polym. Chem. 45, 1122 (2007).
E. Khodaverdi, A. Golmohammadian, S. A. Mohajeri, G. Zohuri, F. S. M. Tekie, and F. Hadizadeh, ISRN Pharm. 2012, 1 (2012).
M. J. Hwang, J. M. Suh, Y. H. Bae, S. W. Kim, and B. Jeong, Biomacromolecules 6, 885 (2005).
S. J. Bae, J. M. Suh, Y. S. Sohn, Y. H. Bae, S. W. Kim, and B. Jeong, Macromolecules 38, 5260 (2005).
W. Zhang, Q. Wu, L. Li, T. Cui, L. Sun, N. Wang, L. Liu, X. Li, and C. Gong, Colloids Surf., B 122, 309 (2014).
J. W. Lee, F. Hua, and D. S. Lee, J. Controlled Release 73, 315 (2001).
C. B. Liu, C. Y. Gong, Y. F. Pan, Y. D. Zhang, J. W. Wang, M. J. Huang, Y. S. Wang, K. Wang, M. L. Gou, M. J. Tu, Y. Q. Wei, and Z. Y. Qian, Colloids Surf., A 302, 430 (2007).
B. Jeong, S. W. Kim, and Y. H. Bae, Adv. Drug Delivery Rev. 64, 154 (2012).
E. Khodaverdi, A. Akbari, F. S. M. Tekie, S. A. Mohajeri, G. Zohuri, and F. Hadizadeh, PDA J. Pharm. Sci. Technol. 67, 135 (2013).
J. C. Gilbert, J. L. Richardson, M. C. Davies, K. J. Palin, and J. Hadgraft, J. Controlled Release 5, 113 (1987).
Y. Tamer and H. Yildirim, Polym. Adv. Technol. 26, 399 (2015).
A. Zhao, S. Zhou, Q. Zhou, and T. Chen, Pharm. Res. 27, 1627 (2010).
W. S. Shim, S. W. Kim, and D. S. Lee, Biomacromolecules 7, 1935 (2006).
X. Wei, C. Gong, M. Gou, S. Z. Fu, Q. Guo, S. Shi, F. Luo, G. Guo, L. Qiu, and Z. Qian, Int. J. Pharm. 381, 1 (2009).
Funding
This work was supported by Research Fund of the Yildiz Technical University. Project no. 2014-07-01-DOP04.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Gökçe Kocabay, Ö., İsmail, O. Biodegradable Thermosensitive Injectable Poly(ε-caprolactone)–Poly(ethylene glycol)–Poly(ε-caprolactone) Based Hydrogels for Biomedical Applications. Polym. Sci. Ser. A 63, 493–504 (2021). https://doi.org/10.1134/S0965545X21050072
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0965545X21050072