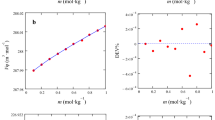
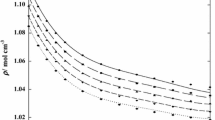
Abstract
The densities of acetonitrile, cyclohexene, and cyclohexanone are measured at 298.15, 303.15, and 313.15 K. Their dynamic viscosities in the 293.15–333.15 K range of temperatures in increments of 5 K under atmospheric pressure are determined. The densities and viscosities of the binary systems formed by acetonitrile, cyclohexene, and cyclohexanone are studied at three temperatures. Using experimental data, the excess molar volumes and viscosity deviations are calculated for the binary systems. All of the systems exhibit deviations from ideal behavior.




Similar content being viewed by others
REFERENCES
R. K. Shukla, A. Kumar, U. Srivastava, et al., Int. J. Thermophys. 37 (9), 1 (2016).
M. Gowrisankar, A. Venkatesulu, T. Srinivasa Krishna, et al., J. Chem. Thermodyn. 107, 104 (2017).
P. Venkateswara Rao, M. Gowrisankar, L. Venkatramana, et al., J. Chem. Thermodyn. 101, 92 (2016).
F. Ouaar, A. Negadi, I. Bahadur, et al., J. Chem. Thermodyn. 113, 291 (2017).
M. Raghavendra, M. Gowrisankar, T. Srinivasa Krishna, et al., J. Solution Chem. 47, 684 (2018).
B. Mukesh, M. Gowrisankar, T. Srinivasa Krishna, et al., J. Therm. Anal. Calorim. 132, 1167 (2018).
N. Nagamani, M. Gowrisankar, T. Srinivasa Krishna, et al., J. Therm. Anal. Calorim. 132, 1 (2018).
A. A. Touazi, E. Khellili, S. Didaoui, et al., J. Therm. Anal. Calorim. 134, 1223 (2018).
K. Yadav and S. S. Yadava, J. Solution Chem. 47, 1172 (2018).
M. Moosavi, K. Taghizadeh, M. Gholami, et al., J. Chem. Thermodyn. 113, 236 (2017).
O. N. Temkin, L. G. Bruk, D. S. Zakharova, et al., Kinet. Catal. 51, 691 (2010).
I. Rekkab-Hammoumraoui, A. Choukchou-Braham, L. Pirault-Roy, et al., Bull. Mater. Sci. 34, 1127 (2011).
I. Rekkab-Hammoumraoui and A. Choukchou-Braham, Bull. Chem. React. Eng. Catal. 13, 22 (2018).
F. C. Chen, Z. Y. Yang, Z. Y. Chen, et al., J. Mol. Liq. 209, 683 (2015).
O. Iulian and O. Ciocirlan, J. Chem. Eng. Data 57, 2640 (2012).
S. Singh, I. Bahadur, G. G. Redhi, et al., J. Mol. Liq. 200, 160 (2014).
P. Droliya and A. K. Nain, J. Chem. Thermodyn. 123, 146 (2018).
D. F. Grant-Taylor and D. D. Macdonald, Can. J. Chem. 54, 2813 (1976).
J. H. Dymond, M. A. Awan, N. F. Glen, et al., Int. J. Thermophys. 12, 433 (1991).
S. Miyanaga, K. Tamura, and S. Murakami, J. Chem. Thermodyn. 24, 1077 (1992).
R. Sadeghi, H. Shekaari, and R. Hosseini, Int. J. Thermophys. 30, 1491 (2009).
A. P. Jogdand and Dr. P. L. Kadam, J. Appl. Phys. 6, 14 (2014).
http://thermalinfo.ru/svojstva-zhidkostej/organicheskie-zhidkosti/dinamicheskaya-vyazkost-zhidkostej. Accessed March 3, 2019.
I. N. V’yunnik, A. M. Zholnovach, S. A. Belous, et al., Oniitekhim, 1 (1983).
G. Ritzoulis, N. Papadopoulos, and D. Jannakoudakis, J. Chem. Eng. Data 31, 146 (1986).
G. Moumouzias, D. K. Panopoulos, and G. Ritzoulis, J. Chem. Eng. Data 36, 20 (1991).
K. Purna ChandraRao, K. S. Reddy, and M. Ramakrishna, Fluid Phase Equilib. 41, 303 (1988).
V. K. Sharma, J. Kataria, and S. Bhagour, J. Mol. Liq. 195, 132 (2014).
S. Dernini, A. M. Polcaro, P. F. Ricci, et al., J. Chem. Eng. Data 32, 194 (1987).
F. Comelli and R. Francesconi, J. Chem. Eng. Data 40 (2), 25 (1995).
D. Rahul, M. Gowri Sankar, M. Chandra Sekhar, et al., J. Therm. Anal. Calorim. 125, 935 (2016).
N. G. Tsierkezos, E. A. Molinou, and A. C. Filippou, J. Solution Chem. 34, 1371 (2005).
M. V. Rathnam, S. Mohite, and M. S. Kumar, J. Chem. Eng. Data 55, 5946 (2010).
G. Srinivasa Rao, P. Anila, C. Rambabu, et al., Thermochim. Acta 590, 116 (2014).
P. Raja Sekar, R. Venkateswarlu, and K. S. Reddy, Canad. J. Chem. 68, 363 (1990).
ACKNOWLEDGMENTS
This work was supported by the Russian Science Foundation, project no. 16-19-10632.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by O. Kadkin
Rights and permissions
About this article
Cite this article
Zhuchkov, V.I., Malyugin, A.A., Raeva, V.M. et al. Transport and Thermodynamic Properties of Binary Mixtures Formed by Acetonitrile, Cyclohexene, and Cyclohexanone. Russ. J. Phys. Chem. 94, 713–719 (2020). https://doi.org/10.1134/S0036024420040287
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024420040287