Abstract
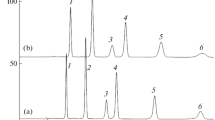
The chromatographic behavior of petanin, petunidin-3-O-[6-O-(4-O-(E)-para-coumaroyl-O-α-rhamnopyranosyl)-β-glucopyranoside]-5-O-β-glucopyranoside, is studied for the first time under conditions of reversed-phase high performance liquid chromatography in mobile phases with different pH values. The relationship between chromatographic behavior (retention time and peak efficiency) and transitions between different forms of anthocyanins is discussed. Analysis of the data obtained in the 2 to 6.5 range of mobile pH phases, the absorption spectra of petanin, and the results from studying the effect of adding tetrabutylammonium bromide to the mobile phase shows that increasing the pH results in the formation of uncharged and anionic forms, in addition to the pseudobase form.





Similar content being viewed by others
REFERENCES
T. Fossen, L. Cabrita, and Ø. M. Andersen, Food Chem. 63, 435 (1998).
C. R. Welch, Q. Wu, and J. E. Simon, Curr. Anal. Chem. 4, 75 (2008).
R. Boulton, Am. J. Enol. Vitic. 52, 67 (2001).
P. Trouillas, J. C. Sancho-Garcia, V. de Freitas, et al., Chem. Rev. 116, 4937 (2016).
Ya. Yu. Kul’chenko, V. I. Deineka, L. A. Deineka, and I. P. Blinova, Tonk. Khim. Tekhnol. 12 (6), 32 (2017).
V. I. Deineka, Ya. Yu. Kul’chenko, I. P. Blinova, et al., Khim. Rastit. Syr’ya, No. 1, 129 (2018).
R. Brouillard and J. Lang, Can. J. Chem. 68, 755 (1990).
A. L. Macanita, P. F. Jr. Moreira, J. C. Lima, et al., J. Phys. Chem. A 106, 1248 (2002).
V. I. Deineka, L. A. Deineka, and I. I. Saenko, J. Anal. Methods Chem. 2015, D732918 (2015).
A. Sidorov, V. Deineka, M. Kostenko, et al., Res. J. Pharm. Biol. Chem. Sci. 7, 2318 (2016).
V. I. Minkin, B. Ya. Simkin, and R. M. Minyaev, Theory of Molecular Structure (Feniks, Rostov-on-Don, 1997), p. 440 [in Russian].
N. W. Preston and C. F. Timberlake, J. Chromatogr. 214, 222 (1981).
R. A. Asenstofer, P. G. Iland, M. E. Tate, and G. P. Jones, Anal. Biochem. 318, 291 (2003).
V. I. Deineka, L. A. Deineka, I. I. Saenko, and A. N. Chulkov, Russ. J. Phys. Chem. A 89, 1300 (2015).
V. I. Deineka, L. A. Deineka, I. P. Blinova, et al., Sorbtsion. Khromatogr. Protsessy 16, 377 (2016).
O. Podlaha and B. Töregård, J. Sep. Sci. 5, 553 (1982).
I. I. Saenko, V. I. Deineka, and L. A. Deineka, J. Anal. Chem. 70, 892 (2015).
V. I. Deineka, A. N. Sidorov, L. A. Deineka, and I. I. Tynyanaya, Sorbtsion. Khromatogr. Protsessy 16, 384 (2016).
R. Brouillard, S. Chassaing, G. Isorez, et al., in Recent Advances in Polyphenol Research, Ed. by C. Santos-Buelga, M. T. Escribano-Bailon, and V. Lattancio (Willey-Blackwell, London, 2010), Vol. 2, p. 8.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by V. Avdeeva
Rights and permissions
About this article
Cite this article
Deineka, V.I., Kul’chenko, Y.Y. & Deineka, L.A. Chromatographic Behavior of Petanin in Reversed-Phase HPLC in Mobile Phases with a Wide Range of pH. Russ. J. Phys. Chem. 93, 572–576 (2019). https://doi.org/10.1134/S0036024419030038
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024419030038