Abstract
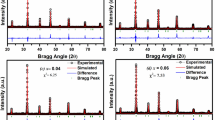
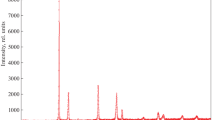
Ca1 – 2xBi2xMo1 – xGexO4 solid solutions with a scheelite-like structure (space group I41/a) and the homogeneity range х = 0.0–0.4 were prepared using standard ceramic technology. The unit cell parameter с and unit cell volume increase as the dopant concentration increases due to the changing size of Ca/BiO8 polyhedra. The predominant thermal expansion of Ca/BiO8 polyhedra was inferred from the temperature-dependent unit cell parameters and Raman spectral patterns. The Ca/Bi–O and Mo/Ge–O bond lengths were calculated. The increasing dopant concentration decreases the thermal expansion coefficient of the ceramics and increases the electrical conductivity and activation energy of the complex oxides compared to the matrix compound. The effective oxygen diffusion coefficient is determined.











Similar content being viewed by others
REFERENCES
H. Kato, N. Matsudo, and A. Kudo, Chem. Lett. 33, 1216 (2004). https://doi.org/10.1246/cl.2004.1216
S. D. Ramarao, S. Roopas Kiran, and V. R. K. Murthy, Mater. Res. Bull. 56, 71 (2014). https://doi.org/10.1016/j.materresbull.2014.04.064
G.-K. Choi, J. Kim, S. H. Yoon, et al., J. Eur. Ceram. Soc. 27, 3063 (2007). https://doi.org/10.1016/j.jeurceramsoc.2006.11.037
V. B. Mikhailik, H. Kraus, G. Miller, et al., J. Appl. Phys. 97, 083523 (2005). https://doi.org/10.1063/1.1872198
B. K. Maji, H. Jena, R. Asuvathraman, et al., J. Alloys Compd. 640, 475 (2015). https://doi.org/10.1016/j.jallcom.2015.04.054
A. Petrov and P. Kofstad, J. Solid State Chem. 30, 83 (1979). https://doi.org/10.1016/0022-4596(79)90133-6
H.-N. Im, M.-B. Choi, S.-Y. Jeon, et al., Ceram. Int. 37, 49 (2011). https://doi.org/10.1016/j.ceramint.2010.08.004
J. Cheng, Ch. Liu, W. Cao, et al., Mater. Res. Bull. 46, 185 (2011). https://doi.org/10.1016/j.materresbull.2010.11.019
S. K. Arora, R. S. Godbole, and D. Lakshminarayana, J. Mater. Sci. 18, 1359 (1983). https://doi.org/10.1007/BF01111955
J. Cheng, W. Bao, Ch. Han, et al., J. Power Sources 195, 1849 (2010). https://doi.org/10.1016/j.jpowsour.2009.10.017
J. Cheng and J. He, Mater. Lett. 209, 525 (2017). https://doi.org/10.1016/j.matlet.2017.08.094
T. Esaka, Solid State Ionics 136–137, 1 (2000). https://doi.org/10.1016/S0167-2738(00)00377-5
W. Bollmann, Cryst. Res. Technol. 18, 100 (1978). https://doi.org/10.1002/crat.19780130816
M. A. Rigdon and R. E. Grace, J. Am. Ceram. Soc. 56, 475 (1973). https://doi.org/10.1111/j.1151-2916.1973.tb12527.x
H.-H. Guo, D. Zhou, L.-X. Pang, et al., J. Eur. Ceram. Soc. 39, 2365 (2019). https://doi.org/10.1016/j.jeurceramsoc.2019.02.010
Z. A. Mikhaylovskaya, E. S. Buyanova, S. A. Petrova, et al., Chim. Techno Acta 8, 20218204 (2021). https://doi.org/10.15826/chimtech.2021.8.2.04
N. I. Matskevich, A. N. Semerikova, N. V. Gelfond, et al., Russ. J. Inorg. Chem. 65, 743 (2020). https://doi.org/10.1134/S0036023620050162
P. E. Dergacheva, I. V. Kul’bakin, A. A. Ashmarin, et al., Russ. J. Inorg. Chem. 66, 1229 (2021). https://doi.org/10.1134/S0036023621080040
O. S. Kaimieva, I. E. Sabirova, E. S. Buyanova, et al., Russ. J. Inorg. Chem. 67, 1348 (2022). https://doi.org/10.1134/S0036023622090054
Yu. V. Emel’yanova, M. V. Morozova, Z. A. Mikhailovskaya, et al., Russ. J. Electrochem. 45, 382 (2009). https://doi.org/10.1134/S1023193509040041
J. Laugier and B. Bochu, LMGP-Suite of Programs for the Interpretation of X-ray Experiments (ENSP, Lab. Materiaux Genie Phys, Grenoble, 2003).
R. S. Bubnova, V. A. Firsova, and S. K. Filatov, Glass Phys. Chem. 39, 347 (2013). https://doi.org/10.1134/S108765961303005X
P. S. Peercy and G. A. Samara, Phys. Rev. B 8, 2033 (1973). https://doi.org/10.1103/PHYSREVB.8.2033
A. V. Klimova, Z. A. Mikhailovskaya, E. S. Buyanova, et al., Russ. J. Electrochem. 57, 825 (2021). https://doi.org/10.1134/S102319352108005X
S. N. Achary, S. J. Patwe, M. D. Mathews, et al., J. Phys. Chem. Solids 67, 774 (2006). https://doi.org/10.1016/j.jpcs.2005.11.009
R. D. Shannon, Acta Crystallogr., Sect. A: Found. Crystallogr. 32, 751 (1976). https://doi.org/10.1107/S0567739476001551
P. G. Zverev, Phys. Status Solidi C 1, 3101 (2004). https://doi.org/10.1002/PSSC.200405413
Z. A. Mikhaylovskaya, E. S. Buyanova, S. A. Petrova, et al., Chim. Techno Acta 9, 20229410 (2022). https://doi.org/10.15826/chimtech.2022.9.4.10
H. M. Rietveld, J. Appl. Crystalogr. 2, 65 (1969). https://doi.org/10.1107/S0021889869006558
Z. A. Mikhaylovskaya, I. Abrahams, S. A. Petrova, et al., J. Solid State Chem. 291, 121627 (2020). https://doi.org/10.1016/j.jssc.2020.121627
J. T. S. Irvine, D. C. Sinclair, and A. R. West, Adv. Mater. 2, 132 (1990). https://doi.org/10.1002/adma.19900020304
Y. B. Abraham, N. A. W. Holzwarth, R. T. Williams, et al., Phys. Rev. B 64, 245109 (2001). https://doi.org/10.1103/PhysRevB.64.245109
H. Zhao, F. Zhang, X. Guo, et al., J. Phys. Chem. Solids 71, 1639 (2010). https://doi.org/10.1016/j.jpcs.2010.08.013
ACKNOWLEDGMENTS
Equipment of the Ural-M Shared Facilities Center of the Institute of Metallurgy, Ural Branch of the Russian Academy of Sciences was used in the X-ray structural experiments. The Raman spectra were measured in the Geoanalyst Shared Facilities Center of the Institute of Geology and Geochemistry, Ural Branch of the Russian Academy of Sciences.
Funding
This work was fulfilled as part of the Government task to the Institute of Geology and Geochemistry (theme No. AAAA-A19-119071090011-6). The retrofit and development of the Geoanalitik Shared Facilities Center of the Institute of Geology and Geochemistry is carried out under a grant from the Ministry of Higher Education and Science of the Russian Federation (agreement No. 075-15-2021-680).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by O. Fedorova
Rights and permissions
About this article
Cite this article
Kaimieva, O.S., Mikhailovskaya, Z.A., Buyanova, E.S. et al. Structure and Electrical Conductivity of Bismuth- and Germanium-Doped Calcium Molybdates. Russ. J. Inorg. Chem. 68, 386–395 (2023). https://doi.org/10.1134/S0036023623600235
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023623600235