Abstract
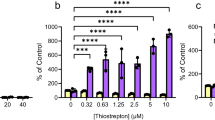
Emergence of ciprofloxacin stress-induced mutants in the cultures of a collection strain Pseudomonas aeruginosa ATCC 27853 and of two strains with a small colony phenotype, which were isolated from a swimming pool biofilm, was studied. In biofilm cultures of the small colony phenotype strains, which were already resistant to hypochlorite, prolonged incubation (up to 16 days) with sublethal ciprofloxacin concentrations was shown to result in emergence of the cells, which are resistant to the antibiotic and form colonies on media with rifampicin (100 μg/mL) and streptomycin (50 μg/mL). Under the same conditions, the mechanisms of temporary adaptation are switched on in the cells of strain ATCC 27853, which enabled its shortterm survival at an average level in liquid media and provided for colony formation on solid medium with ciprofloxacin (0.2 μg/mL). Only 20% of these colonies remained viable when transferred to a higher antibiotic concentration (2 μg/mL).
Similar content being viewed by others
References
Abella, M., Campoy, S., Erill, I., Rojo, F., and Barbe, J., Cohabitation of two different lexA regulons in Pseudomonas putida, J. Bacteriol., 2007, vol. 189, no. 24, pp. 8855–8862.
Chellappa, S.T., Maredia, R., Phipps, K., Haskins, W.E., and Weitao, T., Motility of Pseudomonas aeruginosa contributes to SOS-inducible biofilm formation, Res. Microbiol., 2013, vol. 164, pp. 1019–1027.
Cirz, R.T., O’Neill, B.M., Hammond, J.A., Head, S.R., and Romesberg, F.E., Defining the Pseudomonas aeruginosa SOS response and its role in the global response to the antibiotic ciprofloxacin, J. Bacteriol., 2006, vol. 188, pp. 7101–7110.
Davies, J.A., Harrison, J.J., Marques, L.L., Foglia, G.R., Stremick, C.A., Storey, D.G., Turner, R.J., Olson, M.E., and Ceri, H., The GacS sensor kinase controls phenotypic reversion of small colony variants isolated from biofilms of Pseudomonas aeruginosa PA14, FEMS Microbiol. Ecol., 2007, vol. 59, pp. 32–46.
Demidenok, O.I. and Goncharenko, A.V., Bacterial toxin–antitoxin systems and perspectives for their application in medicine, Appl. Biochem. Microbiol., 2013, vol. 49, no. 6, pp. 535–541.
Déziel, E., Comeau, Y., and Villemur, R., Initiation of biofilm formation by Pseudomonas aeruginosa 57RP correlates with emergence of hyperpiliated and highly adherent phenotypic variants deficient in swimming, swarming, and twitching motilities, J. Bacteriol., 2001, vol. 183, pp. 1195–1204.
Fernández, L. and Hancock, R.E., Adaptive and mutational resistance: role of porins and efflux pumps in drug resistance, Clin. Microbiol. Rev., 2012, vol. 25, pp. 661–681.
Gutierrez, A., Laureti, L., Crussard, S., Abida, H., Rodríguez-Rojas, A., Blázquez, J., Baharoglu, Z., Mazel, D., Darfeuille, F., Vogel, J., and Matic, I., b-Lactam antibiotics promote bacterial mutagenesis via an RpoS-mediated reduction in replication fidelity, Nature Commun., 2013. doi 10.1038/ncomms2607
Kirisits, M.J., Prost, L., Starkey, M., and Parsek, M.R., Characterization of colony morphology variants isolated from Pseudomonas aeruginosa biofilms, Appl. Environ. Microbiol., 2005, vol. 71, pp. 4809–4821.
Kivisaar, M., Mechanisms of stationary-phase mutagenesis in bacteria: mutational processes in pseudomonads, FEMS Microbiol. Lett., 2010, vol. 312, pp. 1–14.
Koorits, L., Tegova, R., Tark, M., Tarassova, K., Tover, A., and Kivisaar, M., Study of involvement of ImuB and DnaE2 in stationary-phase mutagenesis in Pseudomonas putida, DNA Repair, 2007, vol. 6, pp. 863–868.
Layton, J.C. and Foster, P.L., Error-prone DNA polymerase IVis controlled by the stress-response sigma factor, RpoS, in Escherichia coli, Mol. Microbiol., 2003, vol. 50, pp. 549–561.
Magdanova, L.A. and Golyasnaya, N.V., Genetic and morphotypic heterogeneity of swimming pool bacterial populations, Ecol. Genetics, 2011, vol. 9, no. 2, pp. 24–33.
Mamun, A.A.M., Gautam, S., and Humayun, M.Z., Hypermutagenesis in mutA cells is mediated by mistranslational corruption of polymerase, and is accompanied by replication fork collapse, Mol. Microbiol., 2006, vol. 62, pp. 1752–1763.
Mulyukin, A.L., Suzina, N.E., Mel’nikov, V.G., Gal’-chenko, V.F., and El’-Registan, G.I., Dormant state and phenotypic variability of Staphylococcus aureus and Corynebacterium pseudodiphtheriticum, Microbiology (Moscow), 2014, vol. 83, nos. 1–2, pp. 77–84.
Murray, T.S., Ledizet, M., and Kazmierczak, B.I., Swarming motility, secretion of type 3 effectors and biofilm formation phenotypes exhibited within a large cohort of Pseudomonas aeruginosa clinical isolates, J. Med. Microbiol., 2010, vol. 59, pp. 511–520.
Nelson, L.K., Stanton, M.M., Elphinstone, R.E.A., Helwerda, J., Turner, R.J., and Ceri, H., Phenotypic diversification in vivo: Pseudomonas aeruginosa gacS-strains generate small colony variants in vivo that are distinct from in vitro variants, Microbiology (UK), 2010, vol. 156, no. 12, pp. 3699–3709.
Plakunov, V.K., Strelkova, E. A., and Zhurina, M.V., Persistence and adaptive mutagenesis in biofilms, Microbiology (Moscow), 2010, vol. 79, no. 4, pp. 424–434.
Sidorenko, J., Jatsenko, T., Saumaa, S., Teras, R., Tark-Dame, M., Hõrak, R., and Kivisaar, M., Involvement of specialized DNA polymerases Pol II, Pol IV and DnaE2 in DNA replication in the absence of Pol I in Pseudomonas putida, Mutat. Res., 2011, vol. 714, nos. 1–2, pp. 63–77.
Smania, A.M., Segura, I., Pezza, R.J., Becerra, C., Albesa, I., and Argaran, C.E., Emergence of phenotypic variants upon mismatch repair disruption in Pseudomonas aeruginosa, Microbiology (UK), 2004, vol. 150, pp. 1327–1338.
Smith, E.E., Buckley, D.G., Wu, Z., Saenphimmachak, C., Hoffman, L.R., D’Argenio, D.A., Miller, S.I., Ramsey, B.W., Speert, D.P., Moskowitz, S.M., Burns, J.L., Kaul, R., and Olson, M.V., Genetic adaptation by Pseudomonas aeruginosa to the airways of cystic fibrosis patients, Proc. Natl. Acad. Sci. U. S. A., 2006, vol. 103, pp. 8487–8492.
Toymentseva, A.A. and Sharipova, M.R., Genetic mechanisms of bacilli adaptation, Microbiology (Moscow), 2013, vol. 82, no. 3, pp. 257–270.
Tsvetkova, N.A., Guzacheva, I.M., Golyasnaya, N.V., and Belyaeva, L.A., Study of frequensy adaptive mutants and population heterogeneity of cells strains Pseudomonas aeruginosa ATCC 27853 and with a phenotype of small colonies as a result of ciprofloxacin, Fundamental Research, 2013, no. 11, pp. 706–710.
Wei, Q., Tarighi, S., Dötsch, A., Häussler, S., Müsken, M., Wright, V., Cámara, M., Williams, P., Haenen, S., Boerjan, B., Bogaerts, A., Vierstraete, E., Verleyen, P., Schoofs, L., Willaert, R., et al., Phenotypic and genomewide analysis of an antibiotic-resistant small colony variant (SCV) of Pseudomonas aeruginosa, PLoS ONE, 2011, vol. 6, e29276. 10.1371/journalpone.0029276.(12):e29276doi 10.1371.
Weigand, M.R. and Sundin, G.W., General and inducible hypermutation facilitate parallel adaptation in Pseudomonas aeruginosa despite divergent mutation spectra, Proc. Natl. Acad. Sci. U. S. A., 2012, vol. 109, pp. 13680–13685.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © N.A. Tsvetkova, N.V. Golyasnaya, L.A. Belyaeva, I.M. Guzacheva, 2016, published in Mikrobiologiya, 2016, Vol. 85, No. 3, pp. 275–282.
Rights and permissions
About this article
Cite this article
Tsvetkova, N.A., Golyasnaya, N.V., Belyaeva, L.A. et al. Strategies for adaptation to antibiotics in wild type Pseudomonas aeruginosa and in the strains with small colony phenotype. Microbiology 85, 295–301 (2016). https://doi.org/10.1134/S0026261716030140
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026261716030140