Abstract
While research concerning brain structural biomarkers of major depressive disorder (MDD) is continuously progressing, our state of knowledge regarding biomarkers of specific clinical profiles of MDD is still limited. The aim of the present study was to investigate brain structural correlates of social anhedonia as a cardinal symptom of MDD. In a cross-sectional study, we investigated n = 166 patients with MDD and n = 166 matched healthy controls (HC) using structural magnetic resonance imaging (MRI). Social anhedonia was assessed using the Chapman Scales for Social Anhedonia (SAS). An anhedonia x group ANCOVA was performed in a region of interest approach of the dorsal and ventral striatum (bilateral caudate nucleus, putamen, nucleus accumbens respectively) as well as on whole-brain level. The analyses revealed a significant main effect for social anhedonia: higher SAS-scores were associated with reduced gray matter volume in the bilateral caudate nucleus in both the MDD-group (pFWE = 0.002) and the HC-group (pFWE = 0.032). The whole-brain analysis confirmed this association (left: pFWE = 0.036, right: pFWE = 0.047). There was no significant main effect of group and no significant anhedonia x group interaction effect. This is the first study providing evidence for volumetric aberrations in the reward system related to social anhedonia independently of diagnosis, depression severity, medication status, and former course of disease. These results support the hypothesis that social anhedonia has a brain biomarker serving as a possible endophenotype of depression and possibly providing an alternative approach for a more precise and effective treatment.
Similar content being viewed by others
Introduction
Major depressive disorder (MDD) as one of the most frequent and disabling psychiatric diseases [1, 2] has been examined extensively. There are numerous studies showing structural and functional brain alterations in depressive individuals [3, 4]. Among the most consistent findings are alterations of the limbic-cortical brain circuit, namely hyperreactivity of the limbic system to negative stimuli [5,6,7] as well as reductions of gray matter volume in emotion-processing areas like the anterior cingulate cortex, hippocampus, dorsolateral prefrontal cortex, orbitofrontal cortex, and basal ganglia [3, 4, 8,9,10,11]. However, investigating MDD as one homogenous disorder carries the risk of attributing specific biomarkers to the diagnosis of MDD in general even if they might rather be associated with specific symptoms. Considering the variety of symptoms defining the diagnosis of MDD — of which five have to be fulfilled according to DSM-5 [12] — it is likely that the vast phenotypic heterogeneity within this diagnosis is also reflected by different neurobiological patterns. Hence, for a better understanding of the characteristics of MDD, a more detailed approach investigating the specific properties on a symptom-level is needed.
Anhedonia — a cardinal symptom of MDD — is defined as the loss of experiencing pleasure in a variety of behaviors [12]. According to Chapman et al. [13], anhedonia can occur in both physical experiences like touching, eating and movements (physical anhedonia) as well as in interpersonal experiences like making new friends, social interaction, being close to someone (social anhedonia). Anhedonia is not ubiquitous in MDD: it affects only half of patients with current MDD [14], and thus it has been suggested as a potential phenotype of depression [15]. Moreover, it is associated with a poorer outcome in terms of chronicity, relapse, treatment-resistance, and elevated suicide rates in patients with MDD [16, 17]. Anhedonia also occurs in healthy subjects, representing a precursor of MDD [18, 19].
Recent neuroimaging research has mainly focused on anhedonia in general and its physical dimension. In healthy controls, a diminished responsiveness to positive and rewarding stimuli in the nucleus accumbens and the anterior cingulate [19, 20], as well as a reduced nucleus accumbens and caudate nucleus volume [18, 20, 21] have been shown to be associated with (physical) anhedonia. Furthermore, baseline volume of the putamen predicted the severity of anhedonia in healthy adolescents in a three months follow-up investigation [21]. In patients with MDD, imaging studies revealed associations of (physical) anhedonia with reduced gray matter volume in caudate nucleus [22] and reduced brain functional reactivity on positive stimuli in the amygdala, ventral and dorsal striatum and in the insula, while activity of the ventromedial prefrontal cortex was increased [23,24,25].
None of the present studies has examined brain structural associations of the social dimension of anhedonia. Therefore, this is the first study investigating whether social anhedonia itself is associated with specific structural brain alterations in patients with MDD as well as in healthy controls. This could underpin the hypothesis of anhedonia as a possible endophenotype of depression and thereby disclose systematic differences in the courses and treatment outcomes of patients with MDD with different degrees of anhedonia. We hypothesized a reduced volume of the dorsal and ventral striatum as part of the reward system to represent such a biomarker of social anhedonia. As actual depression severity [26], treatment with antidepressive medication [27] and former course of illness [28, 29] have already been shown to have an impact on brain structure, we aimed to clarify if the effect within the subgroup with MDD exists independently of these parameters.
Thus, our study aimed to test the following hypotheses:
-
(1)
Higher social anhedonia is associated with reduced gray matter volume in the caudate nucleus, putamen, and nucleus accumbens in patients with MDD as well as in healthy subjects.
-
(2)
In patients with MDD, these brain alterations are specific for social anhedonia, existing independently of acute depressive symptom severity, actual medication status, and former course of illness.
Materials and Methods
Participants and study design
The sample is based on the Münster neuroimaging cohort as described earlier [29, 30]. Patients were recruited from the inpatient service of the Department of Psychiatry, University of Muenster. Diagnoses were verified employing the structured clinical interview for DSM-IV (SCID-I, [31]). All patients suffered from a current major depressive episode and fulfilled the criteria of MDD. Inclusion criterion was a fully completed questionnaire for anhedonia [32]. Exclusion criteria for all participants were any neurological abnormalities or previous traumatic head injury, chronic medical diseases, or MRI contraindications. Further exclusion criteria for patients with MDD were a diagnosis of bipolar or psychotic disorder, eating disorders, post-traumatic stress disorder, substance or alcohol abuse and dependence as well as any former electroconvulsive therapy. The final sample for our analyses comprised n = 166 patients with MDD with available SAS questionnaire data. Most of the patients (n = 111) did not exhibit any comorbidities (see Table 1 for details), and all but 10 patients were receiving antidepressant treatment.
As a control group, we selected n = 166 healthy controls (HC) matched for age (p = 0.96), sex (p = 0.58) and years of education (p = 0.93). Healthy controls were recruited through public notices and newspaper announcements. Further exclusion criterion for HC was any life-time psychiatric disorder according to the SCID-I [31]. For sample characteristics see Table 1.
All participants completed structural MRI, SCID-I, as well as questionnaires for depression severity (Beck Depression Inventory, BDI-II; [33]), social and physical anhedonia measured by a validated German version of the Chapman Scales for Physical and Social Anhedonia (SASPAS; German version: [32]; Original version: [13]). This self-report questionnaire comprises 26 statements describing pleasure in experiencing various social life events (e.g. “Getting together with old friends has been one of my greatest pleasures.”) as well as 17 items concerning physical pleasures (e.g. “I have had very little desire to try new kinds of foods.”). Participants were instructed to express either approval or rejection to these statements on a dichotomous scale. Patients with MDD also underwent structured interviews to assess the course of illness before baseline (e.g. number of depressive episodes, number and duration of inpatient treatments). We recorded the type and dose of psychopharmacological treatment of the patients with MDD and computed a medication load index as described earlier [8, 34].
The experimental procedure was approved by the local Institutional Review Board (IRB). Written informed consent was obtained from all participants and all participants received a financial compensation.
Structural MRI
Structural data were obtained using a 3-Tesla-MRI (“Gyroscan Intera 3 T” Philips Medical Systems, Best, NL) in cooperation with the Translational Radiology Research Imaging Center (TRIC) of the University Hospital Muenster, Germany. Structural images were preprocessed using the VBM8-toolbox (http://www.neuro.uni-jena.de/vbm/download/) using default parameters as described in our previous works [8, 51, 52]. For a detailed description of data acquisition and preprocessing, see Supplementary Methods.
Statistical analyses
Statistical analyses were performed using statistical parametric map** software (SPM12, Welcome Department of Cognitive Neurology, London, UK; http://www.fil.ion.ucl.ac.uk/spm).
In order to address our first hypothesis that social anhedonia would be associated with reduced gray matter volume in the caudate nucleus, a social anhedonia (SAS-score) x group (MDD, HC) ANCOVA was performed to analyze potential interaction and main effects followed by separate regression analyses for each sample (MDD and HC). We performed an additional physical anhedonia (PAS-score) x group (MDD, HC) ANCOVA in order to examine if the effects were also present for the physical dimension of anhedonia. To investigate our second hypothesis, we further conducted a separate regression analysis of the SAS-score on gray matter volume within the MDD sample including BDI, medication load index and number of psychiatric inpatient treatments as covariates of no interest. We performed region of interest (ROI)-analyses of the caudate nucleus, the putamen and the nucleus accumbens according to our hypotheses followed by subsequent whole-brain analyses. The masks for the bilateral caudate nucleus and for the bilateral putamen were created using the Wake Forest University PickAtlas [37] according to the Automated Anatomical Labeling (AAL)-atlas definitions [38] while the mask for the nucleus accumbens was created according to the IBASPM atlas (http://www.fil.ion.ucl.ac.uk/spm/ext/#IBASPM; [39]). Significance thresholds for multiple testing were obtained at the cluster-level by threshold-free cluster enhancement as a non-parametric approach, which is implemented in the TFCE-toolbox (http://dbm.neuro.uni-jena.de/tfce, Version 138). We consequently established a conservative family wise error (FWE)-corrected threshold of p < .05 obtained by 5000 permutations per test.
Results
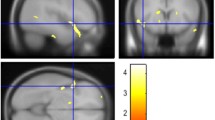
The ROI-analyses of our ANCOVA showed a significant main effect of social anhedonia. Higher scores of social anhedonia (SAS-score) were associated with less gray matter volume in the bilateral caudate nucleus (left: x = −8, y = 8, z = −2, t(328) = 3.83, TFCE = 416.47, k = 1404, pFWE = 0.002, r = −0.176; right: x = 15, y = 6, z = 9, t(328) = 3.34, TFCE = 310.45, k = 1376, pFWE = 0.006, r = −0.168; see Table 2 and Fig. 1) as well as in the left putamen (x = −12, y = 9, z = −3, t(328) = 3.59, TFCE = 201.97, k = 165, pFWE = 0.027, r = −0.177) and in the bilateral nucleus accumbens (left: x = −12, y = 8, z = −8, t(328) = 2.87, TFCE = 36.84, k = 5, pFWE = 0.043, r = −0.174; right: x = 10, y = 15, z = −12, t(328) = 2.49, TFCE = 46.14, k = 101, pFWE = 0.029, r = −0.155). Post-hoc analyses revealed that only for the caudate nucleus, this association was present in both the HC sample and the MDD sample, while for the putamen the association was only present within the MDD sample but not within the HC sample. The analysis of the nucleus accumbens showed no significance for subsamples (see Table 2). The whole-brain analysis confirmed the significant main effect for social anhedonia, showing a significant single cluster located in the left and right caudate nucleus, respectively (left: x = −9, y = 4, z = −2, t(328) = 3.99, TFCE = 1231.30, k = 625, pFWE = 0.036, r = −0.204; right: x = 12, y = 3, z = 0, t(328) = 3.75, TFCE = 1134.54, k = 75, pFWE = 0.047, r = −0.184). No other significant clusters were found at the whole-brain level. There was no significant main effect of physical anhedonia (PAS) neither in the ROI-analyses of the caudate nucleus (pFWE = 0.196), of the putamen (pFWE = 0.264) or of the nucleus accumbens (pFWE = 0.202) nor on whole-brain level (pFWE = 0.646). When entering physical anhedonia as a covariate of no interest in the SAS x group ANCOVA, the ROI-analysis of the caudate nucleus still revealed a significant effect for social anhedonia within the whole sample (left: x = −8, y = 8, z = 1, t(326) = 4.18, TFCE = 476.03, k = 1271, pFWE = 0.001, r = −0.170; right: x = 9, y = 6, z = −2, t(326) = 3.53, TFCE = 281.52, k = 818, pFWE = 0.008, r = −0.163) as well as within the MDD subgroup (left: x = −8, y = 14, z = 10, t(326) = 3.64, TFCE = 366.02, k = 1006, pFWE = 0.004, r = −0.252; right: x = 14, y = 4, z = 10, t(326) = 3.12, TFCE = 228.35, k = 574, pFWE = 0.0015, r = −0.220) and within the HC subgroup (left: x = −8, y = 8, z = 0, t(326) = 3.50, TFCE = 190.12, k = 210, pFWE = 0.019, r = −0.174).
Scatterplot depicting total score of social anhedonia (SAS-score) correlated with gray matter volume of the left caudate nucleus separated by group (mean cluster value at x = −8, y = 8, z = −2). Continuous lines: regression slopes, separated by group. HC healthy control sample, MDD sample with major depressive disorder
The ROI-analysis for the caudate nucleus of the negative relationship between social anhedonia scores and gray matter volume in the MDD sample remained significant after accounting for BDI, medication load index, and total number of psychiatric inpatient treatments (left: x = −18, y = 16, z = 10, t(161) = 3.46, TFCE = 265.31, k = 681, pFWE = 0.011, r = −0.250; right: x = 9, y = 18, z = 12, t(161) = 2.77, TFCE = 143.72, k = 84, pFWE = 0.043, r = −0.196; see Fig. 2). Further analyses excluding patients with anxiety comorbidity still revealed a significant association of reduced caudate nucleus volume and social anhedonia (see Supplementary Results 1). For the putamen, the regression analysis within the subgroup with MDD accounting for BDI, medication load index, and total number of psychiatric inpatient treatments did not reveal a significant association with social anhedonia (pFWE = 0.089).
Axial (z = 9) and sagittal (x = −6) view of the ROI analysis of the caudate nucleus regressing social anhedonia (SAS-score) on gray matter volume within the sample with major depressive disorder (MDD-sample) including BDI, medication load index, and number of inpatient treatments as covariates. Color bar depicts p-value (FWE-correction at pFWE = 0.05 after threshold-free cluster enhancement)
Including age as covariate of no interest did not alter the results of the social anhedonia x group ANCOVA in the ROI analysis of the caudate nucleus (see Supplementary Results 2). The association of social anhedonia and gray matter volume of the caudate nucleus was present both within the male subgroup (see Supplemental Figure S1) as well as within the female subgroup (see Supplemental Figure S2). For results of regression analyses separated by gender, see Supplementary Results 2 and Table S2.
There was no significant main effect for the factor group and no significant effect of the anhedonia x group interaction on gray matter volume neither in the ROI-analyses nor on whole-brain level surviving our rigorous alpha correction procedure for the entire brain volume. However, an exploratory whole-brain analysis at punc < 0.005 comparing patients with MDD and HC using a t-test revealed less gray matter volume in patients with MDD compared to HC in several areas including the inferior frontal gyrus, insula, and hippocampus as already described in the literature ([3, 4]; see Table S1).
Discussion
To our knowledge, this is the first study revealing a specific neurobiological substrate of social anhedonia in both MDD patients and healthy controls. In sum, two major findings emerged: First, these data support our hypothesis of a negative association between social anhedonia and striatal volume, independently from diagnosis. Second, for patients with MDD, this association was not due to differences in current depression severity, medication status and former course of disease. We did not find an association of physical anhedonia with any gray matter volume reductions in the dorsal or ventral striatum.
Our results provide evidence that higher levels of social anhedonia, but not physical anhedonia, were associated with smaller volumes in the ventral and dorsal striatum. However, only for the caudate nucleus, this association was present in both the healthy subgroup as well as in the subgroup with major depression. The caudate nucleus is part of the dorsal striatum. Besides its role in motor control and psychomotricity [40], it is especially involved in reward-based learning [41], reward anticipation and the initiation of goal-directed behaviors, while the ventral striatum (nucleus accumbens) is rather involved in the hedonic experience, so in the reward-processing per se [18, 22]. Thus, reduced volumes in the caudate nucleus may stand in association with difficulties in the (motivational and motoric) initiation and execution of goal-directed actions, described by social anhedonia.
In the healthy subgroup, we could only confirm a correlation between social anhedonia and reduced gray matter volume for the caudate nucleus, but neither for the putamen nor for the nucleus accumbens. Also within the MDD subgroup, under control for actual depression severity, medication load index and former inpatient treatments, the association of social anhedonia and reduced gray matter volume was only significant for the caudate nucleus. These results suggest that the brain structural correlate of social anhedonia is restricted to the caudate nucleus as a region involved in the anticipation of reward. Though, it is possible that alterations of putamen and nucleus accumbens may be associated with social anhedonia on functional rather than on structural level. Investigating the anticipatory as well as the consummatory facets of social anhedonia in a functional neuroimaging paradigm could finally improve the understanding of the regions actually involved in social anhedonia.
In our study, we did not find any association of physical anhedonia with striatal volume reductions. We showed that the relationship of social anhedonia and reduced caudate volume was still significant when controlling for physical anhedonia. Even if these results may firstly suggest that volume reductions of the caudate nucleus are specifically associated to social anhedonia, this stands in contrast to earlier studies showing reduced striatal volumes in association with general or physical anhedonia in healthy adolescents and adults [18, 20, 21] as well as in patients with MDD [22]. One explanation for this conflicting finding may be the restricted variance of the PAS-scores within our sample (see Table 1) as we used a 17-item version of the PAS [32] in contrast to the 61-item version implemented e.g. by the study of Harvey et al. [18]. It is possible that reduced gray matter volume of the caudate nucleus may represent a brain structural correlate of both physical and social anhedonia rather than of one subscale only. Future studies are needed to clarify the specificity of the relationship of social anhedonia and reduced caudate nucleus volume.
Within the vast clinical heterogeneity of MDD [14], anhedonia represents one distinctive feature between melancholic and atypical depression and is associated with the melancholic subtype [12]. Our findings show an association of caudate volume reductions with social anhedonia, and thus may be more prominent in depression with melancholic features. Since accounting for the BDI and course of illness parameters as covariates did not affect our results substantially, we infer from our findings that reduced caudate volume is more likely representing a correlate of (social) anhedonia rather than of MDD in general. Therefore, the caudate nucleus possibly reflects a biomarker suitable for distinguishing different phenotypes of MDD. Recent research describes anhedonia as a promising endophenotype in MDD as it meets the following criteria: specificity, state independence, heritability, familial association, co-segregation, and biological and clinical plausibility [15, 42]. By revealing the caudate as a neurobiological substrate of (social) anhedonia, the present study provides further evidence regarding the biological plausibility for anhedonia as an endophenotype and offers a more differentiated perspective on the neurobiological alterations in MDD.
Moreover, beyond its relevance regarding depression phenoty**, social anhedonia can occur in healthy subjects and is a common precursor to depression in healthy adolescents [43]. In our study, we found a neurobiological substrate of social anhedonia not only in acutely depressed subjects but also in healthy controls. Regarding the debate about whether anhedonia is a state of acute depression or represents a general trait marker [14], our results support a consideration of social anhedonia as a trait-like characteristic associated with structural alterations irrespective of depressive state. This assumption is in line with previous studies showing structural and/or functional alterations in the caudate nucleus in a group of acutely depressed patients [22,23,24], in a group of patients recovered from depression [44] and in healthy subjects [18]. To summarize, we present a neurobiological substrate of social anhedonia, which has the potential to represent: (1) a possible endophenotype of depression itself and (2) a risk biomarker for MDD.
Reduced caudate volume may carry additional clinical relevance, as it is also related to schizophrenia [45], obsessive-compulsive disorder [46], as well as Huntington’s and Parkinson’s disease [47, 48]. Although all of these diseases are comorbid with depressive symptoms, especially (social) anhedonia [49], the findings of reduced caudate volumes may not only represent a specific brain structural correlate of social anhedonia but also of anhedonia in general [18, 22] or of other psychological and neurological features. Future studies are needed in order to investigate (social) anhedonia in a transdiagnostic neuroimaging approach.
This study possibly provides important implications for clinical practice in terms of indication and execution of antidepressive therapies. Due to its contribution to the dopaminergic brain reward system, a reduced caudate volume may provide an explanation for an unfavorable course and treatment resistance seen in patients with MDD suffering from pronounced social anhedonia [17]. Most first-line pharmacological treatments (selective serotonin reuptake inhibitors, serotonin-norepinephrine reuptake inhibitors etc.) primarily focus on the modulation of the serotonergic and/or noradrenergic system, thus the effects on regions such as the caudate involved in the dopaminergic system may be marginal. Consequently, the specific neurobiological alterations of patients suffering from social anhedonia may remain unaffected. However, medications that more directly act on the dopaminergic system, like monoamine oxidase inhibitors (MAOIs), norepinephrine-dopamine reuptake inhibitors (NDRIs), serotonin-norepinephrine-dopamine reuptake inhibitors (SNDRIs), or even psychostimulants, may have the potential to influence striatal structure and/or function and thus relieve anhedonic symptoms [40]. This approach is also transferable to other forms of treatment: in fact there is evidence that cognitive behavioral therapy including behavioral activation increases the striatal activity during reward anticipation [50]. In contrast, other approaches in psychotherapy like cognitive therapy or psychodynamic therapy, which do not directly address anhedonic symptoms, do not seem to have these effects on the reward system [51, 52]. Electroconvulsive therapy, which represents an effective treatment in up to 60–80% of patients with treatment-resistant depression [53], has also been shown to increase gray matter volume of basal ganglia [54]. This confirms the relevance of a treatment-approach addressing social anhedonia including its neural substrate more directly, e.g. by choosing dopaminergic-acting antidepressants or behavioral activation rather than the usual first-line treatment.
A major strength of the present study is the sample size. Above, it is the first study revealing a structural brain correlate of social anhedonia in patients with MDD as well as in healthy controls. The investigation of (social) anhedonia as a possible endophenotype is a novel and innovative aspect in the literature of neuroimaging in MDD. Furthermore, it is the only study up to date showing that the association of (social) anhedonia with striatal volume reductions is not explained by diagnostic status, acute depression severity, former disease course, or medication. Above, in order to examine anhedonia as a specific symptom of depression, we excluded participants with comorbidities related to the reward system like post-traumatic stress disorder, eating disorders, substance or alcohol abuse and dependence. Thus, most of the participants did not have any comorbidities. Furthermore, the analysis excluding those patients with anxiety comorbidities confirms our results pointing out that the relationship of social anhedonia and reduced caudate nucleus volume is present in patients with MDD independently of comorbid anxiety disorders.
Some limitations should be mentioned. First, all but 10 participants were receiving medical treatment. To quantify the influence of psychotropic medication, we computed an index measuring the load of individual medication by quantity and dosage of medication. However, in computing the medication load index we did not differentiate the different classes of antidepressant medication (dopaminergic vs. non-dopaminergic) and we could not quantify the possible effects of the intake of neuroleptics (anti-dopaminergic). Second, as the patient sample has been recruited from the inpatient service, all the patients were suffering from severe MDD. To make further conclusions about the state-or-trait-debate of social anhedonia it would be illuminating to investigate patients remitted from a depressive episode as well. Third, by excluding all patients and healthy controls with substance related disorders, we did not consider nicotine abuse or dependence. As nicotine dependence is shown to be associated with striatal volume alterations [55], we cannot rule out the influence of smoking status on our results. Thus, smoking status may be a possible confounder that should be taken into account in future studies. Fourth, males and females differ in the degree of social anhedonia as well as in gray matter volumes. Although our analyses show that the association of social anhedonia and reduced caudate nucleus volume is present in both the female and male subgroup, gender effects cannot completely ruled out. Finally, we only focused on structural correlates of social anhedonia and cannot make any evidenced conclusions about functional associations.
For future works, we propose a more detailed specific investigation of the different symptoms and phenotypes in MDD. This may be important to gain a better knowledge about differences in the diagnosis, prognosis, and treatment of MDD.
Conclusions
Our data provide evidence for volumetric aberrations in the reward system related to social anhedonia both in depressed and healthy individuals. Namely, social anhedonia is associated with gray matter volume reductions in the caudate nucleus. The association existed independently of diagnosis, depression severity, medication status and former course of disease. These results support the hypothesis that social anhedonia has a brain biomarker and thus serves as a possible endophenotype of depression and provides an alternative approach for a more precise treatment and an improvement of treatment outcomes.
Funding and disclosure
The study was supported by grants of the Innovative Medizinische Forschung (RE111604 and RE111722 to R.R.), and the German Research Foundation (DFG, grant FOR2107 DA1151/5-1 and DA1151/5-2 to U.D.; SFB-TRR58, Projects C09 and Z02 to U.D.) and the Interdisciplinary Center for Clinical Research (IZKF) of the medical faculty of Münster (grant Dan3/012/17 to U.D.). All authors state that they have no actual or potential conflict of interest to declare, including any financial, personal or other relationships with other people or organizations within three years of beginning the submitted work that could influence or bias their work.
References
Murray CJL, Vos T, Lozano R, Naghavi M, Flaxman AD, Michaud C, et al. Disability-adjusted life years (DALYs) for 291 diseases and injuries in 21 regions, 1990-2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380:2197–223.
Wittchen H-U, Jacobi F. Size and burden of mental disorders in Europe - A critical review and appraisal of 27 studies. Eur Neuropsychopharmacol. 2005;15:357–76.
Arnone D, McIntosh AM, Ebmeier KP, Munafò MR, Anderson IM. Magnetic resonance imaging studies in unipolar depression: systematic review and meta-regression analyses. Eur Neuropsychopharmacol. 2012;22:1–16.
Bora E, Fornito A, Pantelis C, Yücel M. Gray matter abnormalities in Major Depressive Disorder: a meta-analysis of voxel based morphometry studies. J Affect Disord. 2012;138:9–18.
Redlich R, Opel N, Bürger C, Dohm K, Grotegerd D, Förster K, et al. The limbic system in youth depression: brain structural and functional alterations in adolescent in-patients with severe depression. Neuropsychopharmacology. 2018;43:546–54.
Stuhrmann A, Suslow T, Dannlowski U. Facial emotion processing in major depression: a systematic review of neuroimaging findings. Biol Mood Anxiety Disord. 2011;1:1–17.
Suslow T, Konrad C, Kugel H, Rumstadt D, Zwitserlood P, Schöning S, et al. Automatic mood-congruent amygdala responses to masked facial expressions in major depression. Biol Psychiatry. 2010;67:155–60.
Redlich R, Almeida JR, Grotegerd D, Opel N, Kugel H, Heindel W, et al. Brain morphometric biomarkers distinguishing unipolar and bipolar depression: a voxel-based morphometry-pattern classification approach. JAMA Psychiatry. 2014;71:1222–30.
Redlich R, Opel N, Grotegerd D, Dohm K, Zaremba D, Burger C, et al. Prediction of individual response to electroconvulsive therapy via machine learning on structural magnetic resonance imaging data. JAMA Psychiatry. 2016;73:557–64.
Schmaal L, Hibar DP, Sämann PG, Hall GB, Baune BT, Jahanshad N, et al. Cortical abnormalities in adults and adolescents with major depression based on brain scans from 20 cohorts worldwide in the ENIGMA Major Depressive Disorder Working Group. Mol Psychiatry. 2017;22:900–9.
Stratmann M, Konrad C, Kugel H, Krug A, Schöning S, Ohrmann P, et al. Insular and hippocampal gray matter volume reductions in patients with major depressive disorder. PLoS One. 2014;9:e102692.
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5). Arlington, VA: American Psychiatric Publishing; 2013.
Chapman LJ, Chapman JP, Raulin ML. Scales for physical and social anhedonia. J Abnorm Psychol. 1976;85:374–82.
Pelizza L, Pupo S, Ferrari A. Anhedonia in schizophrenia and major depression: state or trait? Review of the literature. J Psychopathol. 2012;18:145–55.
Hasler G, Drevets WC, Manji HK, Charney DS. Discovering endophenotypes for major depression. Neuropsychopharmacology. 2004;29:1765–81.
McMakin DL, Olino TM, Porta G, Dietz LJ, Emslie G, Clarke G, et al. Anhedonia predicts poorer recovery among youth with selective serotonin reuptake inhibitor treatmentresistant depression. J Am Acad Child Adolesc Psychiatry. 2012;51:404–11.
Spijker J, Bijl RV, Graaf R, de, Nolen WA. Determinants of poor 1-year outcome of DSM-III-R major depression in the general population: results from the Netherlands Mental Health Survey and Incidence Study (NEMESIS). Acta Psychiatr Scand. 2001;103:122–30.
Harvey PO, Pruessner J, Czechowska Y, Lepage M. Individual differences in trait anhedonia: a structural and functional magnetic resonance imaging study in non-clinical subjects. Mol Psychiatry. 2007;12:767–75.
Keller J, Young CB, Kelley E, Prater K, Levitin DJ, Menon V. Trait anhedonia is associated with reduced reactivity and connectivity of mesolimbic and paralimbic reward pathways. J Psychiatr Res. 2013;47:1319–28.
Wacker J, Dillon DG, Pizzagalli DA. The role of the nucleus accumbens and rostral anterior cingulate cortex in anhedonia: integration of resting EEG, fMRI, and volumetric techniques. Neuroimage. 2009;46:327–37.
Auerbach RP, Pisoni A, Bondy E, Kumar P, Stewart JG, Yendiki A, et al. Neuroanatomical prediction of anhedonia in adolescents. Neuropsychopharmacology. 2017;42:2087–95.
Pizzagalli DA, Holmes AJ, Dillon DG, Goetz EL, Birk JL, Bogdan R, et al. Reduced caudate and nucleus accumbens response to rewards in unmedicated subjects with major depressive disorder. Am J Psychiatry. 2009;166:702–10.
Epstein J, Pan H, Kocsis JH, Yang Y, Butler T, Chusid J, et al. Lack of ventral striatal response to positive stimuli in depressed versus normal subjects. Am J Psychiatry. 2006;163:1784–90.
Keedwell P, Andrew C, Williams SCR, Brammer MJ, Phillips ML. The neural correlates of anhedonia in major depressive disorder. Biol Psychiatry. 2005;58:843–53.
Stuhrmann A, Dohm K, Kugel H, Zwanzger P, Redlich R, Grotegerd D, et al. Mood-congruent amygdala responses to subliminally presented facial expressions in major depression: associations with anhedonia. J Psychiatry Neurosci. 2013;38:249–58.
Vakili K, Pillay SS, Lafer B, Fava M, Renshaw PF, Bonello-Cintron CM, et al. Hippocampal volume in primary unipolar major depression: a magnetic resonance imaging study. Biol Psychiatry. 2000;47:1087–90.
Arnone D, McKie S, Elliott R, Juhasz G, Thomas EJ, Downey D, et al. State-dependent changes in hippocampal grey matter in depression. Mol Psychiatry. 2013;18:1265–72.
Zaremba D, Dohm K, Redlich R, Grotegerd D, Strojny R, Meinert S, et al. Association of brain cortical changes with relapse in patients with major depressive disorder. JAMA Psychiatry. 2018a;75:484–92.
Zaremba D, Enneking V, Meinert S, Förster K, Bürger C, Dohm K et al. Effects of cumulative illness severity on hippocampal gray matter volume in major depression: a voxel-based morphometry study. Psychol Med 2018b;48: 1–8.
Dannlowski U, Kugel H, Grotegerd D, Redlich R, Suchy J, Opel N, et al. NCAN cross-disorder risk variant is associated with limbic gray matter deficits in healthy subjects and major depression. Neuropsychopharmacology. 2015;40:2510–6.
Wittchen H-U, Wunderlich U, Gruschwitz S, Zaudig M. Strukturiertes Klinisches interview für DSM-IV: SKID. Achse I: Psychische Störungen. Interviewheft und Beurteilungsheft. Eine deutschsprachige, erweiterte Bearbeitung der amerikanischen Originalversion des SCID-I. Göttingen: Hogrefe; 1997.
Burgdörfer G, Hautzinger M. Physische und soziale Anhedonie - Die Evaluation eines Forschungsinstruments zur Messung einer psychopathologischen Basisstörung. Eur Arch Psychiatry Neurol Sci. 1987;236:223–9.
Beck AT, Ward CH, Mendelson M, Mock J, Erbaugh J. An inventory for measuring depression. Arch Gen Psychiatry. 1961;4:561–71.
Redlich R, Dohm K, Grotegerd D, Opel N, Zwitserlood P, Heindel W, et al. Reward processing in unipolar and bipolar depression: a functional MRI study. Neuropsychopharmacology. 2015;40:2623–31.
Dannlowski U, Stuhrmann A, Beutelmann V, Zwanzger P, Lenzen T, Grotegerd D, et al. Limbic scars: long-term consequences of childhood maltreatment revealed by functional and structural magnetic resonance imaging. Biol Psychiatry. 2012;71:286–93.
Opel N, Zwanzger P, Redlich R, Grotegerd D, Dohm K, Arolt V, et al. Differing brain structural correlates of familial and environmental risk for major depressive disorder revealed by a combined VBM/pattern recognition approach. Psychol Med. 2016;46:277–90.
Maldjian JA, Laurienti PJ, Kraft RA, Burdette JH. An automated method for neuroanatomic and cytoarchitectonic atlas-based interrogation of fMRI data sets. Neuroimage. 2003;19:1233–9.
Tzourio-Mazoyer N, Landeau B, Papathanassiou D, Crivello F, Etard O, Delcroix N, et al. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage. 2002;15:273–89.
Alemán-Gómez Y, Melie-Garcia L, Valdes-Hernandez P. IBASPM: toolbox for automatic parcellation of brain structures. 12th Annual Meeting of the Organization for Human Brain Map**. June 11–15, 2006. Florence, Italy. 2006.
Treadway MT, Zald DH. Reconsidering anhedonia in depression: lessons from translational neuroscience. Neurosci Biobehav Rev. 2011;35:537–55.
Haruno M, Kuroda T, Kenji D, Toyama K, Kimura M, Samejima K, et al. A neural correlate of reward-based behavioral learning in caudate nucleus: a functional magnetic resonance imaging study of a stochastic decision task. J Neurosci. 2004;24:1660–65.
Goldstein BL, Klein DN. A review of selected candidate endophenotypes for depression. Clin Psychol Rev. 2014;34:417–27.
Pan PM, Sato JR, Salum GA, Rohde LA, Gadelha A, Zugman A, et al. Ventral striatum functional connectivity as a predictor of adolescent depressive disorder in a longitudinal community-based sample. Am J Psychiatry. 2017;174:1112–9.
McCabe C, Cowen PJ, Harmer CJ. Neural representation of reward in recovered depressed patients. Psychopharmacol (Berl). 2009;205:667–77.
Ebdrup BH, Glenthøj B, Rasmussen H, Aggernaes B, Langkilde AR, Paulson OB, et al. Hippocampal and caudate volume reductions in antipsychotic-naive first-episode schizophrenia. J Psychiatry Neurosci. 2010;35:95–104.
Robinson D. Reduced caudate nucleus volume in obsessive-compulsive disorder. Arch Gen Psychiatry. 1995;52:393.
Jurgens CK, Van De Wiel L, Van Es AC, Grimbergen YM, Witjes-Ané MN, Van Der Grond J, et al. Basal ganglia volume and clinical correlates in “preclinical” Huntington’s disease. J Neurol. 2008;255:1785–91.
Nagano-Saito A, Washimi Y, Arahata Y, Kachi T, Lerch JP, Evans AC, et al. Cerebral atrophy and its relation to cognitive impairment in Parkinson disease. Neurology. 2005;64:224–9.
Nilsson FM, Kessing LV, Sorensen TM, Andersen PK, Bolwig TG. Major depressive disorder in Parkinson’s disease: a register-based study. Acta Psychiatr Scand. 2002;106:202–11.
Dichter GS, Felder JN, Petty C, Bizzell J, Ernst M, Smoski MJ. The effects of psychotherapy on neural responses to rewards in major depression. Biol Psychiatry. 2009;66:886–97.
Yoshimura S, Okamoto Y, Onoda K, Matsunaga M, Okada G, Kunisato Y, et al. Cognitive behavioral therapy for depression changes medial prefrontal and ventral anterior cingulate cortex activity associated with self-referential processing. Soc Cogn Affect Neurosci. 2014;9:487–93.
Buchheim A, Viviani R, Kessler H, Kächele H, Cierpka M, Roth G et al. Changes in prefrontal-limbic function in major depression after 15 months of long-term psychotherapy. PLoS ONE. 2012;7:e33745.
Haq AU, Sitzmann AF, Goldman ML, Maixner DF, Mickey BJ. Response of depression to electroconvulsive therapy: a meta-analysis of clinical predictors. J Clin Psychiatry. 2015;76:1374–84.
Wade BS, Joshi SH, Njau S, Leaver AM, Vasavada M, Woods RP, et al. Effect of electroconvulsive therapy on striatal morphometry in major depressive disorder. Neuropsychopharmacology. 2016;5:1–30.
Das D, Cherbuin N, Anstey KJ, Sachdev PS, Easteal S. Lifetime cigarette smoking is associated with striatal volume measures. Addict Biol. 2012;17:817–25.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Enneking, V., Krüssel, P., Zaremba, D. et al. Social anhedonia in major depressive disorder: a symptom-specific neuroimaging approach. Neuropsychopharmacol. 44, 883–889 (2019). https://doi.org/10.1038/s41386-018-0283-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41386-018-0283-6
- Springer Nature Switzerland AG
This article is cited by
-
Social Anhedonia: (f)MRI Studies
Neuroscience and Behavioral Physiology (2023)
-
Anhedonia in Depression: Neurobiological and Genetic Aspects
Neuroscience and Behavioral Physiology (2022)
-
I, robot: depression plays different roles in human–human and human–robot interactions
Translational Psychiatry (2021)
-
Neuroanatomical Correlates of Social Intelligence Measured by the Guilford Test
Brain Topography (2021)
-
A novel construct of anhedonia revealed in a Chinese sample via the Revised Physical and Social Anhedonia Scales
BMC Psychiatry (2020)