Abstract
Background
Autophagy plays an important role in cancer cells. Targeting autophagy in cancer can provide new opportunities for drug development.
Methods
In this study we tested four Schiff base Cu(II) complexes against human breast cancer cells (MCF-7) and human non-cancerous cells (HEK-293T). We have tested their cytotoxic effect by evaluating IC50 using MTT test. To detect morphological changes of the actin fibers we have used fluorescent microscopy. To determine the type of cell death we used electrophoretic analysis and western blot analysis (protein LC3).
Results
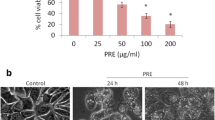
IC50 values of the complexes increased with time of their influence, indicating acquired resistance of MCF-7 to the complexes. Healthy cells HEK-293T were not sensitive to the Cu(II) complexes. Compared with the control cells (cells without Cu(II) complexes) which were without morphological changes of actin fibers, Cu(II) complexes induced condensation and asymmetric conformational changes in actin filaments. To examine the type of cell death induced by the Cu(II) complexes we treated MCF-7 cells with Cu(II) complexes (1, 10, 50 and 100 μmol/L) during a 72 h incubation period. By electrophoresis we have not detected any DNA fragmentation. To determine whether Cu(II) complexes induced autophagy or necrotic cell death we used the western blot analysis. MCF-7 cells influenced with tested Cu(II) complexes produced LC3 protein after their 72 h incubation indicating autophagy in MCF-7 cancer cells.
Conclusions
Tested Schiff base copper (II) complexes have antiproliferative activity against cancer cells but not against healthy cells. They have induced autophagy in the cancer cell line MCF-7.
Similar content being viewed by others

References
Lowe SW, Lin WA. Apoptosis in cancer. Cancerogenesis 2000;2(3)485–95 1.
Hajrezaie M, Paydar M, Moghadamtousi SZ, Hassandarvish P, Gwaram NS, Zahedifard ME, et al. Schiff base-derived copper (II) complex is a potent inducer of apoptosis in colon cancer cells by activating the intrinsic pathway. Sci World J 2014;2014(2014):12.
Liang C, **a J, Lei D, Li X, Yao Q, Gao J. Synthesis, in vitro and in vivo antitumor activity of symmetrical bis-Schiff base derivatives of isatin. Eur J Med Chem 2014;74:742–50.
Zhong W, Zhu H, Sheng F, Tian Y, Zhou J, Chen Y, et al. Activation of the MAPK11/12/13/14 (p38 MAPK) pathway regulates the transcription of autophagy genes in response to oxidative stress induced by a novel copper complex in HeLa cells. Autophagy 2014;10(7):1285–300.
Farber E. Programmed cell death: necrosis versus apoptosis. Mod Pathol 1994;7(5):605–9.
Langer V, Gyepesová D, Scholtzová E, Mach P, Kohútová M, Valent A, et al. Crystal and electronic structure of aqua(N-salicylidene-methylester-L-glutamato)Cu(II) monohydrate. Z Kristallogr 2004;219(2004):112–6.
Langer V, Scholtzová E, Gyepesová D, Kohútová M, Valent A. (N-salicylidene-D, L-glutamato)(2-methylimidazole)copper(II). Acta Cryst 2004;0:129–32.
Langer V, Gyepesová D, Kohútová M, Valent A. Dimeric (isoquinoline)(N-salicylidene-D,L-glutamato)copper(II) ethanol solvate. Acta Cryst 2009; C65:208–10.
Langer V, Mach P, Gyepesová D, Andrezálová L, Kohútová M. X-ray and DFT studies of a mono- and binuclear copper(II) ionic compound containing a Schiff base. Acta Cryst 2012;C68:326–8.
Vyas A, Patitungkho S, Jamadar A, Adsule S, Padhyea S, Ahmad A. ATRA-hydrazonate derivatives and their copper complexes against hormone-dependent (MCF-7), hormone-independent (MDA-MB-231 and BT-20) breast cancer and androgen-independent (PC3) prostate cancer cell lines. Inorg Chem Commun 2012;23:17–20.
Francisco D, Peddi P, Hair JM, Flood BA, Cecil AM, Kalogerinis PT. Induction and processing of complex DNA damage in human breast cancer cells MCF-7 and nonmalignant MCF-10A cells. Free Radic Biol Med 2009;44(4):558–69.
Santini C, Pellei M, Gandin V, Porchia M, Tisato F, Marzano C. Advances in copper complexes as anticancer agents. Chem Rev 2014;114(1):815–62.
Frias González SE, Anguiano EA, Herrera AM, Calzada DE, Pichardo CO. Cytotoxic, pro-apoptotic, pro-oxidant, and non-genotoxic activities of a novel copper(II) complex against human cervical cancer. Toxicology 2013;314 (1):155–6.
Singh R, Jadeja RN, Thounaojam MC, Patel T, Devkar RV, Chakraborty D. Synthesis, DNA binding and antiproliferative activity of ternary copper complexes of moxifloxacin and gatifloxacin against lung cancer cells. Inorg Chem Commun 2012;23:78–84.
Vyas MV, Jadeja RN, Patel D, Devkar RV, Gupta VK. Effect of ligand substitution in pyrazolone based binary and ternary Cu(II) complexes on DNA binding, protein binding and anti-cancer activity on A549 lung carcinoma cell lines. Polyhedron 2014;80:20–33.
Wen-jie G, Si-si Y, Ning C, **g G, Qiu-yun Q. ROS-mediated autophagy was involved in cancer cell death induced by novel copper(II) complex. Exp Toxicol Pathol 2010;62(5):577–82.
Bhumika T, Noble A, Creaven BS, Walsh M, Kavanagh K, Egan DA. Apoptotic cell death: a possible key event in mediating the in vitro anti-proliferative effect of a novel copper(II) complex, [Cu(4-Mecdoa)(phen)2] (phen = phenanthroline, 4-Mecdoa = 4-methylcoumarin-6,7-dioxactetate), in human malignant cancer cells. Eur J Pharmacol 2007;569(1–2):16–8.
Milacic V, Chen D, Giovagnini L, Diez A, Fregona D, Dou QP. Pyrrolidine dithiocarbamate-zinc(II) and -copper(II) complexes induce apoptosis in tumor cells by inhibiting the proteasomal activity. Toxicol Appl Pharmacol 2008;231(1):24–33.
De Vizcaya-Ruiz A, Rivero-Muller A, Ruiz-Ramirez L, Kass GE, Kelland LR, Orr RM, et al. Induction of apoptosis by a novel copper-based anticancer compound, casiopeina II, in L1210 murine leukaemia and CH1 human ovarian carcinoma cells. Toxicol In Vitro 2000;14(1):1–5.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koňariková, K., Perdikaris, G.A., Gbelcová, H. et al. Autophagy in MCF-7 cancer cells induced by copper complexes. Pharmacol. Rep 68, 1221–1224 (2016). https://doi.org/10.1016/j.pharep.2016.07.011
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2016.07.011