Abstract
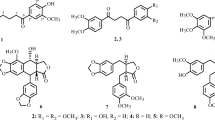
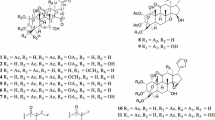
Phytochemical investigation of Bauhinia acuruana Moric., Fabaceae, resulted in the isolation of sixteen constituents, including two new compounds 2′-hydroxy-2,3,5-trimethoxybibenzyl (1), (2R,3S)-2-(3,4′-dihydroxyphenyl)-5-methoxy-6-methylchroman-3,7-diol (2), together with fourteen known ones (3–16). The structures of the compounds were established by spectroscopic analysis including HR-ESIMS, 1D and 2D NMR data, followed by comparison with previously reported data from the literature. Compounds 1, 2, 6, 7, 8 and 9 were evaluated for their cytotoxicity, which turned out to be marginal in a panel of six human cancer cell lines.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Anjaneyulu, A.S.R., Reddy, A.V.R., Reddy, D.S.K., Ward, R.S., Adhikesavalu, D., Cameron, T.S., 1984. Pacharin: a new dibenzo(2,3-6,7)oxepin derivative from Bauhinia racemosa Lank. Tetrahedron 40, 4245–4252.
Apisantiyakom, S., Kittakoop, P., Manyum, T., Kirtikara, K., Bremner, J.B., Thebtaranonth, Y., 2004. Novel biologically active bibenzyls from Bauhinia saccocalyx PIERRE. Chem. Biodivers. 1, 1694–1701.
Bezerra, G.P., Góis, R.W.S., De Brito, T.S., De Lima, F.J.B., Bandeira, M.A.M., Romero, N.R., Magalhães, P.J.C., Santiago, G.M.P., 2013. Phytochemical study guided by the myorelaxant activity of the crude extract, fractions and constituent from stem bark of Hymenaea courbaril L. J. Ethnopharmacol. 149, 62–69.
Boonphong, S., Puangsombat, P., Baramee, A., Mahidol, C., Ruchirawat, S., Kittakoop, P., 2007. Bioactive compounds from Bauhinia purpurea possessing antimalarial, antimycobacterial, antifungal, anti-inflammatory, and cytotoxic activities. J. Nat. Prod. 70, 795–801.
Cechinel Filho, V., 2009. Chemical composition and biological potential of plants from the genus Bauhinia. Phytother. Res. 23, 1347–1354.
Chen, X.-J., Mei, W.-L., Cai, C.-H., Guo, Z.-K., Song, X.-Q., Dai, H.-F., 2014. Four new bibenzyl derivatives from Dendrobium sinense. Phytochem. Lett. 9, 107–112.
Cren-Olivé, C., Lebrun, S., Rolando, C., 2002. An efficient synthesis of the four mono methylated isomers of (+)-catechin including the major metabolites and of some dimethylated and trimethylated analogues through selective protection of the catechol ring. J. Chem. Soc., Perkin Trans. 16, 821–830.
Danielsen, K., Aksnes, D.W., Francis, G.W., 1992. NMR study of some anthraquinones from Rhubarb. Mag. Reson. Chem. 30, 359–363.
Da Silva, F.M.A., Koolen, H.H.F., Barison, A., De Souza, A.D.L., Pinheiro, M.L.B., 2012. Steroids and triterpene from the bark of Unonopsis guatterioides R.E. Fr. (Anonnaceae). Int. J. Pharm. Pharm. Sci. 4, 522–523.
De Sousa, L.M., De Carvalho, J.L., Da Silva, H.C., Lemos, T.L.G., Arriaga, A.M.C., Braz-Filho, R., Militão, G.C.G., Silva, T.D.S., Ribeiro, P.R.V., Santiago, G.M.P., 2016. New cytotoxic bibenzyl and other constituents from Bauhinia ungulata L. (Fabaceae). Chem. Biodivers. 13, 1630–1635.
Gois, R.W.S., De Sousa, L.M., Lemos, T.L.G., Arriaga, A.M.C., Andrade-Neto, M., Santiago, G.M.P., Ferreira, Y.S., Alves, P.B., De Jesus, H.C.R., 2011. Chemical composition and larvicidal effects of essential oil from Bauhinia acuruana (Moric) against Aedes aegypti. J. Essent. Oil Res. 23, 59–62.
Góis, R.W.S., De Sousa, L.M., Santiago, G.M.P., Romero, N.R., Lemos, T.L.G., Arriaga, A.M.C., Braz-Filho, R., 2013. Larvicidal activity against Aedes aegypti of pacharin from Bauhinia acuruana. Parasitol. Res. 112, 2753–2757.
Imai, T., Inoue, S., Ohdaira, N., Matsushita, Y., Suzuki, R., Sakurai, M., De Jesus, J.M.H., Ozaki, S.K., Finger, Z., Fukushima, K., 2008. Heartwood extractives from the Amazonian trees Dipteryx odorata, Hymenaea courbaril, and Astronium lecointei and their antioxidant activities. J. Wood Sci. 54, 470–475.
Imam, S., Azhar, I., Hasan, M.M., Ali, M.S., Ahmed, S.W., 2007. Two triterpenes lupanone and lupeol isolated and identified from Tamarindus indica Linn. Pak. J. Pharm. Sci. 20, 125–127.
Kittakoop, P., Kirtikara, K., Tanticharoen, M., Thebtaranonth, Y., 2000. Antimalarial preracemosols A and B, possible biogenetic precursors of racemosol from Bauhinia malabarica Roxb. Phytochemistry 55, 349–352.
Lendl, A., Werner, I., Glasl, S., Kletter, C., Mucaji, P., Presser, A., Reznicek, G., Jurenitsch, J., Taylor, D.W., 2005. Phenolic and terpenoid compounds from Chione venosa (SW.) Urban var. venosa (Bois Bandé). Phytochemistry 66, 2381–2387.
Li, Y.-K., Zhou, B., Ye, Y.-Q., Du, G., Niu, D.-Y., Meng, C.-Y., Gao, X.-M., Hu, Q.-F., 2013. Two new diphenylethylenes from Arundina graminifolia and their cytotoxicity. Bull. Korean Chem. Soc. 34, 3257–3260.
Meira, M., David, J.M., David, J.P., Araújo, S.V., Regis, T.L., Giulietti, A.M., De Queiróz, L.P., 2008. Constituintes químicos de Ipomoea subincana Meisn. (Convolvulaceae). Quim. Nova 31, 751–754.
Mosmann, T., 1983. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. Immunol. Methods 65, 55–63.
Moujir, L.M., Seca, A.M.L., Araujo, L., Silva, A.M.S., Barreto, M.C., 2011. A new natural spiro heterocyclic compound and the cytotoxic activity of the secondary metabolites from Juniperus brevifolia leaves. Fitoterapia 82, 225–229.
Pettit, G.R., Numata, A., Iwamoto, C., Usami, Y., Yamada, T., Ohishi, H., Cragg, G.M., 2006. Antineoplasic agents. 551. Isolation and structures of bauhiniastatins 1–4 from Bauhinia purpurea. J. Nat. Prod. 69, 323–327.
Slowing, K., Sollhuber, M., Carretero, E., Villar, A., 1994. Flavonoids glycosides from Eugenia jambos. Phytochemistry 37, 255–258.
Soares, P.K., Scarminio, I.S., 2008. Multivariate chromatographic fingerprint preparation and authentication of plant material from the genus Bauhinia. Phytochem. Anal. 19, 78–85.
Takasugi, M., Kawashima, S., Monde, K., Katsui, N., Masamune, T., Shirata, A., 1987. Antifungal compounds from Discorea batatas inoculated with Pseudomonas cichorii. Phytochemistry 26, 371–375.
Vaz, A.M.S.F., Tozzi, A.M.G.A., 2003. Bauhinia ser. Cansenia (Leguminosae: Caesalpinioideae) no Brasil. Rodriguésia 54, 55–143.
Xu, F.-Q., Xu, F.-C., Hou, B., Fan, W.-W., Zi, C.-T., Li, Y., Dong, F.-W., Liu, Y.-Q., Sheng, J., Zuo, Z.-L., Hu, J.-M., 2014. Cytotoxic bibenzyl dimers from the stems of Dendrobium fimbriatum Hook. Bioorg. Med. Chem. Lett. 24, 5268–5273.
Yang, D.-S., Wei, J.-G., Peng, W.-B., Wang, S.-M., Sun, C., Yang, Y.-P., Liu, K.-C., Li, X.-L., 2014. Cytotoxic prenylated bibenzyls and flavonoids from Macaranga kurzii. Fitoterapia 99, 261–266.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors’ contributions
RWSG (PhD student) did the compound isolation procedure and contributed in compound identification by NMR and literature search. LMS, HCS, FEFS and ATAP contributed in carrying out the laboratory work, and interpretation of the spectroscopic data. MASL, AMCA, TLGL and RBF contributed in the interpretation of the spectroscopic data. GCCG and PBNS contributed to biological assays. FJTG contributed to plant collection and confection of herbarium. GMPS designed the study, supervised the laboratory work and wrote the manuscript. LMS contributed to critical reading of the manuscript. All the authors have read the final manuscript and approved the submission.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Góis, R.W.S., de Sousa, L.M., da Silva, H.C. et al. Chemical constituents from Bauhinia acuruana and their cytotoxicity. Rev. Bras. Farmacogn. 27, 711–715 (2017). https://doi.org/10.1016/j.bjp.2017.09.002
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2017.09.002