Abstract
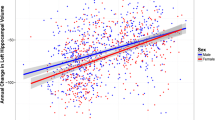
Tau proteins accumulation and their spreading pattern were affected by gender in cognitive impairment patients, especially in the progression of Alzheimer’s disease (AD). However, it was unclear whether the gender effects for tau deposition influenced by amyloid deposition. The aim of this study was to investigate gender differences for tau depositions in Aβ positive (A+) subjects. In this study, tau and amyloid positron emission tomography images, structural magnetic resonance imaging images, and demographic information were collected from 179 subjects in Alzheimer’s Disease Neuroimaging Initiative (ADNI) database and 63 subjects from Huashan Hospital. Subjects were classified as T+ or T− according to the presence or absence of tau (T) biomarkers. We used two-sample t test and one-way analysis of variance test to analyze the effect of gender with adjusting for age, years of education, and Minimum Mental State Examination. In the ADNI cohort, we found differences in Tau deposition in fusiform gyrus, inferior temporal gyrus, middle temporal gyrus and parahippocampal gyrus between the female T+ (FT+) and male T+ (MT+) groups (p < 0.05). Tau deposition did not differ significantly between female T− (FT−) and male T− (MT−) subjects (p > 0.05). In the Huashan Hospital cohort, there was no difference in Tau deposition between FT+ and MT+ (p > 0.05). The results show that tau depositions significantly increased in females in above brain regions. Our findings suggest that tau deposition is influenced by gender in the A+ subjects. This result has important clinical implications for the development of gender-guided early interventions for patients with both Tau and Amyloid depositions.





Similar content being viewed by others
Data Availability
Data from the ADNI cohort are available on the website (https://adni.loni.usc.edu/). Data from the Huashan cohort were used under license for the current study, and so are not publicly available. Data are, however, available from the authors upon reasonable request and with permission of Huashan Hospital.
Code Availability
Not applicable.
References
Ardekani BA, Convit A, Bachman AH (2016) Analysis of the MIRIAD data shows sex differences in hippocampal atrophy progression. J Alzheimers Dis JAD 50(3):847–857. https://doi.org/10.3233/jad-150780
Barnes LL, Wilson RS, Bienias JL, Schneider JA, Evans DA, Bennett DA (2005) Sex differences in the clinical manifestations of Alzheimer disease pathology. Arch Gen Psychiatry 62(6):685–691. https://doi.org/10.1001/archpsyc.62.6.685
Braak H, Alafuzoff I, Arzberger T, Kretzschmar H, Del Tredici K (2006) Staging of Alzheimer disease-associated neurofibrillary pathology using paraffin sections and immunocytochemistry. Acta Neuropathol 112(4):389–404. https://doi.org/10.1007/s00401-006-0127-z
Chen K, Roontiva A, Thiyyagura P, Lee W, Liu X, Ayutyanont N, Protas H, Luo JL, Bauer R, Reschke C, Bandy D, Koeppe RA, Fleisher AS, Caselli RJ, Landau S, Jagust WJ, Weiner MW, Reiman EM (2015) Improved power for characterizing longitudinal amyloid-β PET changes and evaluating amyloid-modifying treatments with a cerebral white matter reference region. J Nucl Med off Publ Soc Nucl Med 56(4):560–566. https://doi.org/10.2967/jnumed.114.149732
Chini M, Pöpplau JA, Lindemann C, Carol-Perdiguer L, Hnida M, Oberländer V, Xu X, Ahlbeck J, Bitzenhofer SH, Mulert C, Hanganu-Opatz IL (2020) Resolving and rescuing developmental miswiring in a mouse model of cognitive impairment. Neuron 105(1):60-74.e67. https://doi.org/10.1016/j.neuron.2019.09.042
Chotipanich C, Nivorn M, Kunawudhi A, Promteangtrong C, Boonkawin N, Jantarato A (2020) Evaluation of imaging windows for tau PET imaging using (18)F-PI2620 in cognitively normal individuals, mild cognitive impairment, and Alzheimer’s disease patients. Mol Imaging 19:1536012120947582. https://doi.org/10.1177/1536012120947582
Demetrius LA, Eckert A, Grimm A (2021) Sex differences in Alzheimer’s disease: metabolic reprogramming and therapeutic intervention. Trends Endocrinol Metab 32(12):963–979. https://doi.org/10.1016/j.tem.2021.09.004
Ferretti MT, Iulita MF, Cavedo E, Chiesa PA, Schumacher Dimech A, Santuccione Chadha A, Baracchi F, Girouard H, Misoch S, Giacobini E, Depypere H, Hampel H, Women’s Brain Project, the Alzheimer Precision Medicine Initiative (2018) Sex differences in Alzheimer disease—the gateway to precision medicine. Nat Rev Neurol 14(8):457–469. https://doi.org/10.1038/s41582-018-0032-9
Gonzalez-Escamilla G, Lange C, Teipel S, Buchert R, Grothe MJ (2017) PETPVE12: an SPM toolbox for Partial Volume Effects correction in brain PET—application to amyloid imaging with AV45-PET. Neuroimage 147:669–677. https://doi.org/10.1016/j.neuroimage.2016.12.077
Goyal MS, Blazey TM, Su Y, Couture LE, Durbin TJ, Bateman RJ, Benzinger TLS, Morris JC, Raichle ME, Vlassenko AG (2019) Persistent metabolic youth in the aging female brain. Proc Natl Acad Sci USA 116(8):3251–3255. https://doi.org/10.1073/pnas.1815917116
Holland D, Desikan RS, Dale AM, McEvoy LK (2013) Higher rates of decline for women and apolipoprotein E epsilon4 carriers. AJNR Am J Neuroradiol 34(12):2287–2293. https://doi.org/10.3174/ajnr.A3601
Hu YT, Boonstra J, McGurran H, Stormmesand J, Sluiter A, Balesar R, Verwer R, Swaab D, Bao AM (2021) Sex differences in the neuropathological hallmarks of Alzheimer’s disease: focus on cognitively intact elderly individuals. Neuropathol Appl Neurobiol 47(7):958–966. https://doi.org/10.1111/nan.12729
Hua X, Hibar DP, Lee S, Toga AW, Jack CR Jr, Weiner MW, Thompson PM (2010) Sex and age differences in atrophic rates: an ADNI study with n = 1368 MRI scans. Neurobiol Aging 31(8):1463–1480. https://doi.org/10.1016/j.neurobiolaging.2010.04.033
Jack CR, Knopman DS, Jagust WJ, Petersen RC, Weiner MW, Aisen PS, Shaw LM, Vemuri P, Wiste HJ, Weigand SD, Lesnick TG, Pankratz VS, Donohue MC, Trojanowski JQ (2013) Tracking pathophysiological processes in Alzheimer’s disease: an updated hypothetical model of dynamic biomarkers. Lancet Neurol 12(2):207–216. https://doi.org/10.1016/S1474-4422(12)70291-0
Jack CR Jr, Wiste HJ, Weigand SD, Therneau TM, Knopman DS, Lowe V, Vemuri P, Mielke MM, Roberts RO, Machulda MM, Senjem ML, Gunter JL, Rocca WA, Petersen RC (2017a) Age-specific and sex-specific prevalence of cerebral β-amyloidosis, tauopathy, and neurodegeneration in cognitively unimpaired individuals aged 50–95 years: a cross-sectional study. Lancet Neurol 16(6):435–444. https://doi.org/10.1016/s1474-4422(17)30077-7
Jack CR, Wiste HJ, Weigand SD, Therneau TM, Lowe VJ, Knopman DS, Gunter JL, Senjem ML, Jones DT, Kantarci K, Machulda MM, Mielke MM, Roberts RO, Vemuri P, Reyes DA, Petersen RC (2017b) Defining imaging biomarker cut points for brain aging and Alzheimer’s disease. Alzheimers Dement 13(3):205–216. https://doi.org/10.1016/j.jalz.2016.08.005
Jack CR Jr, Bennett DA, Blennow K, Carrillo MC, Dunn B, Haeberlein SB, Holtzman DM, Jagust W, Jessen F, Karlawish J, Liu E, Molinuevo JL, Montine T, Phelps C, Rankin KP, Rowe CC, Scheltens P, Siemers E, Snyder HM, Sperling R (2018) NIA-AA Research Framework: toward a biological definition of Alzheimer’s disease. Alzheimers Dement 14(4):535–562. https://doi.org/10.1016/j.jalz.2018.02.018
Jansen WJ, Ossenkoppele R, Knol DL, Tijms BM, Scheltens P, Verhey FR, Visser PJ, Aalten P, Aarsland D, Alcolea D, Alexander M, Almdahl IS, Arnold SE, Baldeiras I, Barthel H, van Berckel BN, Bibeau K, Blennow K, Brooks DJ, van Buchem MA, Camus V, Cavedo E, Chen K, Chetelat G, Cohen AD, Drzezga A, Engelborghs S, Fagan AM, Fladby T, Fleisher AS, van der Flier WM, Ford L, Förster S, Fortea J, Foskett N, Frederiksen KS, Freund-Levi Y, Frisoni GB, Froelich L, Gabryelewicz T, Gill KD, Gkatzima O, Gómez-Tortosa E, Gordon MF, Grimmer T, Hampel H, Hausner L, Hellwig S, Herukka SK, Hildebrandt H, Ishihara L, Ivanoiu A, Jagust WJ, Johannsen P, Kandimalla R, Kapaki E, Klimkowicz-Mrowiec A, Klunk WE, Köhler S, Koglin N, Kornhuber J, Kramberger MG, Van Laere K, Landau SM, Lee DY, de Leon M, Lisetti V, Lleó A, Madsen K, Maier W, Marcusson J, Mattsson N, de Mendonça A, Meulenbroek O, Meyer PT, Mintun MA, Mok V, Molinuevo JL, Møllergård HM, Morris JC, Mroczko B, Van der Mussele S, Na DL, Newberg A, Nordberg A, Nordlund A, Novak GP, Paraskevas GP, Parnetti L, Perera G, Peters O, Popp J, Prabhakar S, Rabinovici GD, Ramakers IH, Rami L, Resende de Oliveira C, Rinne JO, Rodrigue KM, Rodríguez-Rodríguez E, Roe CM, Rot U, Rowe CC, Rüther E, Sabri O, Sanchez-Juan P, Santana I, Sarazin M, Schröder J, Schütte C, Seo SW, Soetewey F, Soininen H, Spiru L, Struyfs H, Teunissen CE, Tsolaki M, Vandenberghe R, Verbeek MM, Villemagne VL, Vos SJ, van Waalwijk van Doorn LJ, Waldemar G, Wallin A, Wallin Å K, Wiltfang J, Wolk DA, Zboch M, Zetterberg H, (2015) Prevalence of cerebral amyloid pathology in persons without dementia: a meta-analysis. JAMA 313(19):1924–1938. https://doi.org/10.1001/jama.2015.4668
Jia L, Quan M, Fu Y, Zhao T, Li Y, Wei C, Tang Y, Qin Q, Wang F, Qiao Y, Shi S, Wang YJ, Du Y, Zhang J, Zhang J, Luo B, Qu Q, Zhou C, Gauthier S, Jia J (2020) Dementia in China: epidemiology, clinical management, and research advances. Lancet Neurol 19(1):81–92. https://doi.org/10.1016/s1474-4422(19)30290-x
Kaczkurkin AN, Raznahan A, Satterthwaite TD (2019) Sex differences in the develo** brain: insights from multimodal neuroimaging. Neuropsychopharmacology 44(1):71–85. https://doi.org/10.1038/s41386-018-0111-z
La Joie R, Visani AV, Baker SL, Brown JA, Bourakova V, Cha J, Chaudhary K, Edwards L, Iaccarino L, Janabi M, Lesman-Segev OH, Miller ZA, Perry DC, O’Neil JP, Pham J, Rojas JC, Rosen HJ, Seeley WW, Tsai RM, Miller BL, Jagust WJ, Rabinovici GD (2020) Prospective longitudinal atrophy in Alzheimer’s disease correlates with the intensity and topography of baseline tau-PET. Sci Transl Med 12(524):eaau5732. https://doi.org/10.1126/scitranslmed.aau5732
Li L, Liu FT, Li M, Lu JY, Sun YM, Liang X, Bao W, Chen QS, Li XY, Zhou XY, Guan Y, Wu JJ, Yen TC, Jang MK, Luo JF, Wang J, Zuo C (2021) Clinical utility of (18) F-APN-1607 tau PET imaging in patients with progressive supranuclear palsy. Mov Disord off J Mov Disord Soc 36(10):2314–2323. https://doi.org/10.1002/mds.28672
Long JM, Holtzman DM (2019) Alzheimer disease: an update on pathobiology and treatment strategies. Cell 179(2):312–339. https://doi.org/10.1016/j.cell.2019.09.001
Lu J, Bao W, Li M, Li L, Zhang Z, Alberts I, Brendel M, Cumming P, Lu H, **ao Z, Zuo C, Guan Y, Zhao Q, Rominger A (2020) Associations of [(18)F]-APN-1607 tau PET binding in the brain of Alzheimer’s disease patients with cognition and glucose metabolism. Front Neurosci 14:604. https://doi.org/10.3389/fnins.2020.00604
Lundeen TF, Seibyl JP, Covington MF, Eshghi N, Kuo PH (2018) Signs and artifacts in amyloid PET. Radiographics 38(7):2123–2133. https://doi.org/10.1148/rg.2018180160
Mattsson N, Lönneborg A, Boccardi M, Blennow K, Hansson O (2017) Clinical validity of cerebrospinal fluid Aβ42, tau, and phospho-tau as biomarkers for Alzheimer’s disease in the context of a structured 5-phase development framework. Neurobiol Aging 52:196–213. https://doi.org/10.1016/j.neurobiolaging.2016.02.034
McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM (1984) Clinical diagnosis of Alzheimer’s disease: report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 34(7):939–944. https://doi.org/10.1212/wnl.34.7.939
Medeiros AM, Silva RH (2019) Sex Differences in Alzheimer’s disease: where do we stand? J Alzheimers Dis JAD 67(1):35–60. https://doi.org/10.3233/jad-180213
Meeker KL, Wisch JK, Hudson D, Coble D, **ong C, Babulal GM, Gordon BA, Schindler SE, Cruchaga C, Flores S, Dincer A, Benzinger TL, Morris JC, Ances BM (2021) Socioeconomic status mediates racial differences seen using the AT(N) framework. Ann Neurol 89(2):254–265. https://doi.org/10.1002/ana.25948
Mielke MM, Vemuri P, Rocca WA (2014) Clinical epidemiology of Alzheimer’s disease: assessing sex and gender differences. Clin Epidemiol 6:37–48. https://doi.org/10.2147/clep.S37929
Ossenkoppele R, Schonhaut DR, Schöll M, Lockhart SN, Ayakta N, Baker SL, O’Neil JP, Janabi M, Lazaris A, Cantwell A, Vogel J, Santos M, Miller ZA, Bettcher BM, Vossel KA, Kramer JH, Gorno-Tempini ML, Miller BL, Jagust WJ, Rabinovici GD (2016) Tau PET patterns mirror clinical and neuroanatomical variability in Alzheimer’s disease. Brain J Neurol 139(Pt 5):1551–1567. https://doi.org/10.1093/brain/aww027
Ossenkoppele R, Rabinovici GD, Smith R, Cho H, Schöll M, Strandberg O, Palmqvist S, Mattsson N, Janelidze S, Santillo A, Ohlsson T, Jögi J, Tsai R, La Joie R, Kramer J, Boxer AL, Gorno-Tempini ML, Miller BL, Choi JY, Ryu YH, Lyoo CH, Hansson O (2018) Discriminative accuracy of [18F]flortaucipir positron emission tomography for Alzheimer disease vs other neurodegenerative disorders. JAMA 320(11):1151–1162. https://doi.org/10.1001/jama.2018.12917
Oveisgharan S, Arvanitakis Z, Yu L, Farfel J, Schneider JA, Bennett DA (2018) Sex differences in Alzheimer’s disease and common neuropathologies of aging. Acta Neuropathol 136(6):887–900. https://doi.org/10.1007/s00401-018-1920-1
Petersen RC (2004) Mild cognitive impairment as a diagnostic entity. J Intern Med 256(3):183–194. https://doi.org/10.1111/j.1365-2796.2004.01388.x
Salehi A, Gonzalez Martinez V, Swaab DF (1998) A sex difference and no effect of ApoE type on the amount of cytoskeletal alterations in the nucleus basalis of Meynert in Alzheimer’s disease. Neurobiol Aging 19(6):505–510. https://doi.org/10.1016/s0197-4580(98)00106-7
Samuel BG (1995) Diagnostic and statistical manual of mental disorders, 4th edn. (DSM-IV). 152(8):1228–1228. https://doi.org/10.1176/ajp.152.8.1228
Shadlen MF, Larson EB, Gibbons L, McCormick WC, Teri L (1999) Alzheimer’s disease symptom severity in blacks and whites. J Am Geriatr Soc 47(4):482–486. https://doi.org/10.1111/j.1532-5415.1999.tb07244.x
Shi Z, Fu LP, Zhang N, Zhao X, Liu S, Zuo C, Cai L, Wang Y, Gao S, Ai L, Guan YH, Xu B, Ji Y (2020) Amyloid PET in dementia syndromes: a Chinese Multicenter Study. J Nucl Med off Publ Soc Nucl Med 61(12):1814–1819. https://doi.org/10.2967/jnumed.119.240325
Shinohara M, Murray ME, Frank RD, Shinohara M, DeTure M, Yamazaki Y, Tachibana M, Atagi Y, Davis MD, Liu CC, Zhao N, Painter MM, Petersen RC, Fryer JD, Crook JE, Dickson DW, Bu G, Kanekiyo T (2016) Impact of sex and APOE4 on cerebral amyloid angiopathy in Alzheimer’s disease. Acta Neuropathol 132(2):225–234. https://doi.org/10.1007/s00401-016-1580-y
Sowell ER, Peterson BS, Kan E, Woods RP, Yoshii J, Bansal R, Xu D, Zhu H, Thompson PM, Toga AW (2007) Sex differences in cortical thickness mapped in 176 healthy individuals between 7 and 87 years of age. Cereb Cortex (new York, NY: 1991) 17(7):1550–1560. https://doi.org/10.1093/cercor/bhl066
Sundermann EE, Panizzon MS, Chen X, Andrews M, Galasko D, Banks SJ (2020) Sex differences in Alzheimer’s-related Tau biomarkers and a mediating effect of testosterone. Biol Sex Differ 11(1):33. https://doi.org/10.1186/s13293-020-00310-x
Tang MX, Stern Y, Marder K, Bell K, Gurland B, Lantigua R, Andrews H, Feng L, Tycko B, Mayeux R (1998) The APOE-epsilon4 allele and the risk of Alzheimer disease among African Americans, whites, and Hispanics. JAMA 279(10):751–755. https://doi.org/10.1001/jama.279.10.751
Weuve J, Barnes LL, Mendes de Leon CF, Rajan KB, Beck T, Aggarwal NT, Hebert LE, Bennett DA, Wilson RS, Evans DA (2018) Cognitive aging in Black and White Americans: cognition, cognitive decline, and incidence of Alzheimer disease. Dementia 29(1):151–159. https://doi.org/10.1097/ede.0000000000000747
Wolters EE, Golla SSV, Timmers T, Ossenkoppele R, van der Weijden CWJ, Scheltens P, Schwarte L, Schuit RC, Windhorst AD, Barkhof F, Yaqub M, Lammertsma AA, Boellaard R, van Berckel BNM (2018) A novel partial volume correction method for accurate quantification of [18F] flortaucipir in the hippocampus. EJNMMI Res 8(1):79. https://doi.org/10.1186/s13550-018-0432-2
**ong C, Luo J, Schindler SE, Fagan AM, Benzinger T, Hassenstab J, Balls-Berry JE, Agboola F, Grant E, Moulder KL, Morris JC (2022) Racial differences in longitudinal Alzheimer’s disease biomarkers among cognitively normal adults. Alzheimers Dement. https://doi.org/10.1002/alz.12608
Yan S, Zheng C, Paranjpe MD, Li J, Benzinger TLS, Lu J, Zhou Y (2020) Association of sex and APOE ε4 with brain tau deposition and atrophy in older adults with Alzheimer’s disease. Theranostics 10(23):10563–10572. https://doi.org/10.7150/thno.48522
Yan S, Zheng C, Paranjpe MD, Li Y, Li W, Wang X, Benzinger TLS, Lu J, Zhou Y (2021) Sex modifies APOE ε4 dose effect on brain tau deposition in cognitively impaired individuals. Brain J Neurol 144(10):3201–3211. https://doi.org/10.1093/brain/awab160
Zhao Q, Liu M, Ha L, Zhou Y (2019) Quantitative (18)F-AV1451 brain tau PET imaging in cognitively normal older adults, mild cognitive impairment, and Alzheimer’s disease patients. Front Neurol 10:486. https://doi.org/10.3389/fneur.2019.00486
Zhu D, Montagne A, Zhao Z (2021) Alzheimer’s pathogenic mechanisms and underlying sex difference. Cell Mol Life Sci CMLS 78(11):4907–4920. https://doi.org/10.1007/s00018-021-03830-w
Acknowledgements
This work was supported by the National Natural Science Foundation of China (82020108013, 81971641, 82071200, 82021002), the research project of Shanghai Health Commission (2020YJZX0111), the Shanghai Aging and Maternal and Child Health Research Special Project (Grant 2020YJZX0111) and the Clinical Research Plan of Shanghai Hospital Development Center (Grants SHDC2020CR1038B and SHDC2020CR4007). Also, special thanks to HWZ, who helped us with the visual assessment of the images of the Huashan cohort.
Author information
Authors and Affiliations
Contributions
All authors significantly contributed to planning, writing, and editing of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
The study was approved by the Ethics Committee of Huashan Hospital, Fudan University, Shanghai, China.
Consent to participate
All subjects from Huashan Hospital provided written informed consent.
Consent for publication
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Y., Lu, J., Wang, M. et al. Influence of Gender on Tau Precipitation in Alzheimer’s Disease According to ATN Research Framework. Phenomics 3, 565–575 (2023). https://doi.org/10.1007/s43657-022-00076-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43657-022-00076-9