Abstract
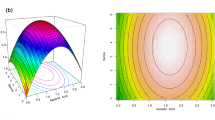
Bacterial Cellulose (BC) offers a wide range of applications across various industries, including food, biomedical, and textiles, owing to its distinctive properties. Its unique 3D reticulated network of cellulose nanofibers, imparts excellent mechanical qualities, a high water-holding capacity, and thermal stability. Additionally, it possesses remarkable biocompatibility, biodegradability, high crystallinity, and purity. These attributes have offered significant interest in BC within both academic and industrial sectors. However, BC production is associated with high costs due to the use of expensive growth media and low yields. The study reports the potential of our indigenous isolate, Komagataeibacter saccharivorans BC-G1, as BC producer. Statistical optimization of BC production was carried out using Placket-Burman design and Central composite design, by selecting different parameters. Eight significant factors such as temperature, pH, glucose, yeast, peptone, acetic acid, incubation time and % inoculum were studies using ANOVA-based response surface methodology. Results showed that BC yield (8.5 g/L) with 1.8-fold after optimization of parameters. Maximum cellulose production (8.5 ± 1.8 g/L) was obtained using 2% glucose, 0.3% yeast extract, 0.3% peptone, 0.75% (v/v) acetic acid at pH 7.0 for 10 days of incubation with 4% inoculum at 25 °C under static culture. Main effect graph showed incubation time and acetic acid concentration as the most significant parameters affecting BC production in our study. The physicochemical characterization of produced BC was done using FTIR, XRD and SEM techniques.







Similar content being viewed by others
References
Srivastava S, Mathur G(2022)bacterial cellulose: a multipurpose biomaterial for manmade world. Curr Appl Sci. https://doi.org/10.55003/cast.2022.03.23.014
Lin S, Singajaya S, Lo T, Santoso SP, Hsu H, Cheng K (2023) Evaluation of porous bacterial cellulose produced from foam templating with different additives and its application in 3D cell culture. Int J Biol Macromol 234:123680. https://doi.org/10.1016/j.ijbiomac.2023.123680
Lin S, Huang S, Ting Y, Hsu H, Cheng K (2022) Evaluation of detoxified sugarcane bagasse hydrolysate by atmospheric cold plasma for bacterial cellulose production. Int J Biol Macromol 204:136–143. https://doi.org/10.1016/j.ijbiomac.2022.01.186
Wahid F, Huang LH, Zhao XQ, Li WC, Wang YY, Jia SR, Zhong C (2021) Bacterial cellulose and its potential for biomedical applications. Biotechnol Adv 53:107856. https://doi.org/10.1016/j.biotechadv.2021.107856
Chen C, Ding W, Zhang H, Zhang L, Huang Y, Fan M, Yang J, Sun D (2022) Bacterial cellulose-based biomaterials: from fabrication to application. Carbohydr Polym 278:p118995. https://doi.org/10.1016/j.carbpol.2021.118995
Stumpf TR, Yang X, Zhang J, Cao X (2018) In situ and ex situ modifications of bacterial cellulose for applications in tissue engineering. Mater Sci Eng 82:372–383. https://doi.org/10.1016/j.msec.2016.11.121
Carvalho T, Guedes G, Sousa FL, Freire CS, Santos HA (2019) Latest advances on bacterial cellulose-based materials for wound healing, delivery systems, and tissue engineering. Biotechnol J 14(12):p1900059. https://doi.org/10.1002/biot.201900059
El-Gendi H, Salama A, El-Fakharany EM, Saleh AK (2023) Optimization of bacterial cellulose production from prickly pear peels and its ex situ impregnation with fruit byproducts for antimicrobial and strawberry packaging applications. Carbohydr Polym 302:120383. https://doi.org/10.1016/j.carbpol.2022.120383
Bae S, Shoda M (2005) Statistical optimization of culture conditions for bacterial cellulose production using Box-Behnken design. Biotechnol Bioeng 90(1):20–28. https://doi.org/10.1002/bit.20325
Rastogi A, Banerjee R (2020) Statistical optimization of bacterial cellulose production by Leifsonia soli and its physico-chemical characterization. Process Biochem 91:297–302. https://doi.org/10.1016/j.procbio.2019.12.021
Sievers M, Swings J (2005) Family II. Acetobacteraceae. In: Brenner DJ, Krieg NR, Staley JT, Garrity GM, editors. Bergey’s manual® of systematic bacteriology, vol. 2, The proteobacteria. Part C, The alpha-, beta-, delta-, and epsilon proteobacteria. New York, NY, USA: Springer; 41–95
Srivastava Sand Mathur G (2022) Komagataeibacter saccharivorans strain BC-G1: an alternative strain for production of bacterial cellulose. Biologia 77:3657–3668. https://doi.org/10.1007/s11756-022-01222-4
Tang W, Jia S, Jia Y, Yang H (2010) The influence of fermentation conditions and post-treatment methods on porosity of bacterial cellulose membrane. World J Microbiol Biotechnol 26 L:125–131. https://doi.org/10.1007/s11274-009-0151-y
Ul-Islam M, Khattak WA, Kang M, Kim SM, Khan T, Park JK (2013) Effect of post-synthetic processing conditions on structural variations and applications of bacterial cellulose. Cellulose 20:253–263. https://doi.org/10.1007/s10570-012-9799-9
Plackett RL, Burman JP (1946) The design of optimum multifactorial experiments. Biometrika 305–325. https://doi.org/10.2307/2332195
Box GE, Behnken DW (1960) Some new three level designs for the study of quantitative variables. Technometrics 2(4):455–475
Yatsyshyn VY, Fedorovych DV, Sibirny AА (2010) Medium optimization for production of flavin mononucleotide by the recombinant strain of the yeast Candida famata using statistical designs. Biochem Eng J 49(1):52–60. https://doi.org/10.1016/j.bej.2009.11.010
Embuscado ME, Marks JS, BeMiller JN (1994) Bacterial cellulose. II. Optimization of cellulose production by Acetobacter xylinum through response surface methodology. Food Hydrocoll 8(5):419–430. https://doi.org/10.1016/S0268-005X(09)80085-4
Ruan C, Zhu Y, Zhou X, Abidi N,Catcmark JM (2016) Effect of cellulose crystallinity on bacterial cellulose assembly. Cellulose 23:3417–3427. https://doi.org/10.1007/s10570-016-1065-0
Park S, Baker JO, Himmel ME, Parilla PA, Johnson DK (2010) Cellulose crystallinity index: measurement techniques and their impact on interpreting cellulase performance. Biotechnol Biofuels 3:10. https://doi.org/10.1186/1754-6834-3-10
Scherrer P (1918) Bestimmung der Größe und der inneren Struktur von Kolloidteilchen mittels Röntgenstrahlen. Nachrichten von der Gesellschaft der Wissenschaften zu Göttingen, Mathematisch-Physikalische Klasse, 98–100. https://eudml.org/doc/59018
Zeng X, Small DP, Wan W (2011) Statistical optimization of culture conditions for bacterial cellulose production by Acetobacter Xylinum BPR 2001 from maple syrup. Carbohydr Polym 85(3):506–513. https://doi.org/10.1016/j.carbpol.2011.02.034
Hegde S, Bhadri G, Narsapur K, Koppal S, Oswal P, Turmuri N, Jumnal V, Hungund B (2013) Statistical optimization of medium components by response surface methodology for enhanced production of bacterial cellulose by Gluconacetobacter Persimmonis. J Bioprocess Biotech 4(1):1–5. https://doi.org/10.4172/2155-9821.1000142
Mohite BV, Kamalja KK, Patil SV (2012) Statistical optimization of culture conditions for enhanced bacterial cellulose production by Gluconoacetobacter Hansenii NCIM 2529. Cellulose 19:1655–1666. https://doi.org/10.1007/s10570-012-9760-y
Anand Vera B (1993) Computational methods for the representation of contour lines. Computer-Aided Civil and Infrastructure Engineering 8. no. 2: 119–128. https://doi.org/10.1111/j.1467-8667.1993.tb00199.x
Saleh AK, Farrag AA, Soliman NA, Ibrahim MM, El-Shinnawy NA, Abdel Fattah YR (2019) Evaluation of culture requirements for cellulose production by Egyptian local isolate alongside reference strain Gluconacetobacter hansenii ATCC 23769. Pak J Biotechnol 16(2):69–80. https://doi.org/10.34016/pjbt.2019.16.1.12
Santoso SP, Chou CC, Lin S, Soetaredjo FE, Ismadji S, Hsieh C, Cheng K (2020) Enhanced production of bacterial cellulose by Komactobacter intermedius using statistical modeling. Cellulose 27(5):2497–2509. https://doi.org/10.1007/s10570-019-02961-5
Akintunde MO, Adebayo-Tayo BC, Ishola MM, Zamani A, Horváth IS (2022) Bacterial cellulose production from agricultural residues by two Komagataeibacter Sp. Strains Bioengineered 13(4):10010–10025. https://doi.org/10.1080/21655979.2022.2062970
Ling Z, Chen S, Zhang X, Takabe K, Xu F (2017) Unraveling variations of crystalline cellulose induced by ionic liquid and their effects on enzymatic hydrolysis. Scientific 7(1). https://doi.org/10.1038/s41598-017-09885-9
Shao M, Shi Z, Zhai B, Zhang X, Li Z (2022) Preparation and performance analysis of bacterial cellulose-based Composite hydrogel based on scanning electron microscope. https://doi.org/10.1155/2022/8750394. Scanning,1–7
Funding
Not applicable.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Luis Henrique Souza Guimaraes.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Srivastava, S., Mathur, G. Statistical optimization of bioprocess parameters for enhanced production of bacterial cellulose from K. saccharivorans BC-G1. Braz J Microbiol (2024). https://doi.org/10.1007/s42770-024-01397-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42770-024-01397-9