Abstract
Background
Familial non-autoimmune hyperthyroidism is a rare disease caused by germline activating variants in the thyroid-stimulating hormone receptor (TSHR) gene. The c.1856A > G (p.Asp619Gly) pathogenic variant has been described in cases of toxic adenoma but never before, to our knowledge, in a case of familial non-autoimmune hyperthyroidism.
Patient findings
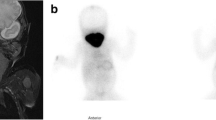
A 3-year-old boy was admitted for acute gastroenteritis presenting with goiter and tall stature. Laboratory findings revealed peripheral hyperthyroidism and negativity for thyroid autoantibodies. Antithyroid drug treatment was effective, but relapses occurred shortly after attempts to decrease the drug dose. As the boy’s father and paternal grandmother also experienced relapsing hyperthyroidism manifesting in early childhood, genetic testing of TSHR was indicated. The c.1856A > G (p.Asp619Gly) pathogenic variant was found in all three affected family members. Functional in vitro characterization of the variant verified that it enhances constitutional activation of the receptor, leading to increased production of cyclic adenosine monophosphate. Total thyroidectomy was indicated in the boy due to an unsatisfactory prognosis. Due to persistent positive thyroglobulin serum concentration, a diagnostic radioiodine scan was performed approximately 2 years later. Residual thyroid tissue was revealed; therefore, radioiodine ablative therapy was performed. Despite adequate thyroxine substitution over a long period of follow-up, TSH remained suppressed.
Conclusions
Unlike Graves’ disease, familial non-autoimmune hyperthyroidism cases present with antithyroid drug-dependence. Not ultrasound but positive thyroglobulin serum concentration indicated residual thyroid tissue. Early detection of residual thyroid tissue and radioiodine ablation prevented the subject from experiencing relapsing hyperthyroidism and undergoing unnecessary repeated surgery. Life-long hormone substitution should be adjusted to free thyroxine rather than TSH serum concentrations.



Similar content being viewed by others
Data availability
Maximum care was taken to thoroughly describe the methodology and provide all relevant data to enable reproduction of our observations. All data generated or analyzed during this study are included in this published article or its Supplementary Material file.
Code availability
Not applicable.
References
Hanley P, Lord K, Bauer AJ (2016) Thyroid disorders in children and adolescents: a review. JAMA Pediatr 170:1008–1019. https://doi.org/10.1001/jamapediatrics.2016.0486
Lavard L, Ranlov I, Perrild H, Andersen O, Jacobsen BB (1994) Incidence of juvenile thyrotoxicosis in Denmark, 1982–1988. A nationwide study Eur J Endocrinol 130:565–568. https://doi.org/10.1530/eje.0.1300565
Paschke R, Niedziela M, Vaidya B, Persani L, Rapoport B, Leclere J (2012) 2012 European thyroid association guidelines for the management of familial and persistent sporadic non-autoimmune hyperthyroidism caused by thyroid-stimulating hormone receptor germline mutations. Eur Thyroid J 1:142–147. https://doi.org/10.1159/000342982
Kleinau G, Neumann S, Gruters A, Krude H, Biebermann H (2013) Novel insights on thyroid-stimulating hormone receptor signal transduction. Endocr Rev 34:691–724. https://doi.org/10.1210/er.2012-1072
Stephenson A, Lau L, Eszlinger M, Paschke R (2020) The thyrotropin receptor mutation database update. Thyroid 30:931–935. https://doi.org/10.1089/thy.2019.0807
Ferraz C, Paschke R (2017) Inheritable and sporadic non-autoimmune hyperthyroidism. Best Pract Res Clin Endocrinol Metab 31:265–275. https://doi.org/10.1016/j.beem.2017.04.005
Delange F, Benker G, Caron P, Eber O, Ott W, Peter F, Podoba J, Simescu M, Szybinsky Z et al (1997) Thyroid volume and urinary iodine in European schoolchildren standardization of values for assessment of iodine deficiency. Eur J Endocrinol 136:180–187. https://doi.org/10.1530/eje.0.1360180
Zimmermann MB, Hess SY, Molinari L, De Benoist B, Delange F, Braverman LE, Fujieda K, Ito Y, Jooste PL et al (2004) New reference values for thyroid volume by ultrasound in iodine-sufficient schoolchildren: a World Health Organization/Nutrition for Health and Development Iodine Deficiency Study Group Report. Am J Clin Nutr 79:231–237. https://doi.org/10.1093/ajcn/79.2.231
Tanner JM, Goldstein H, Whitehouse RH (1970) Standards for children’s height at ages 2–9 years allowing for heights of parents. Arch Dis Child 45:755–762. https://doi.org/10.1136/adc.45.244.755
Barstow C, Rerucha C (2015) Evaluation of short and tall stature in children. Am Fam Physician 92:43–50
Wong FC, Au EY, Ip RW, Yau HC, Lit LC (2018) False-positive TSH receptor antibody results in an infant with activating TSHR mutation. Endocr J 65:793–794. https://doi.org/10.1507/endocrj.EJ18-0161
Giuliani C, Saji M, Bucci I, Napolitano G (2016) Bioassays for TSH receptor autoantibodies from FRTL-5 cells to TSH receptor-LH/CG Receptor chimeras the contribution of Leonard D Kohn. Front Endocrinol (Lausanne) 7:103. https://doi.org/10.3389/fendo.2016.00103
Nwosu BU, Gourgiotis L, Gershengorn MC, Neumann S (2006) A novel activating mutation in transmembrane helix 6 of the thyrotropin receptor as cause of hereditary nonautoimmune hyperthyroidism. Thyroid 16:505–512. https://doi.org/10.1089/thy.2006.16.505
Laurberg P, Wallin G, Tallstedt L, Abraham-Nordling M, Lundell G, Torring O (2008) TSH-receptor autoimmunity in Graves’ disease after therapy with anti-thyroid drugs surgery or radioiodine: a 5-year prospective randomized study. Eur J Endocrinol 158:69–75. https://doi.org/10.1530/EJE-07-0450
Parma J, Duprez L, Van Sande J, Cochaux P, Gervy C, Mockel J, Dumont J, Vassart G (1993) Somatic mutations in the thyrotropin receptor gene cause hyperfunctioning thyroid adenomas. Nature 365:649–651. https://doi.org/10.1038/365649a0
Kleinau G, Worth CL, Kreuchwig A, Biebermann H, Marcinkowski P, Scheerer P, Krause G (2017) Structural-functional features of the thyrotropin receptor: a class a G-protein-coupled receptor at work. Front Endocrinol (Lausanne) 8:86. https://doi.org/10.3389/fendo.2017.00086
Nishihara E, Nagayama Y, Amino N, Hishinuma A, Takano T, Yoshida H, Kubota S, Fukata S, Kuma K et al (2007) A novel thyrotropin receptor germline mutation (Asp617Tyr) causing hereditary hyperthyroidism. Endocr J 54:927–934. https://doi.org/10.1507/endocrj.k07-088
Claeysen S, Govaerts C, Lefort A, Van Sande J, Costagliola S, Pardo L, Vassart G (2002) A conserved Asn in TM7 of the thyrotropin receptor is a common requirement for activation by both mutations and its natural agonist. FEBS Lett 517:195–200. https://doi.org/10.1016/s0014-5793(02)02620-0
Huth S, Jaeschke H, Schaarschmidt J, Paschke R (2014) Controversial constitutive TSHR activity patients, physiology, and in vitro characterization. Horm Metab Res 46:453–461. https://doi.org/10.1055/s-0034-1375651
Nishihara E, Tsugawa M, Ozaki Y, Nagayama Y, Fukata S, Hirokawa M, Ito M, Nishikawa M, Nakamura H et al (2018) Long-term follow-up of a patient with sporadic nonautoimmune hyperthyroidism due to a thyrotropin-receptor mutation (D619G). AACE Clin Case Rep 4:e84–e89. https://doi.org/10.4158/EP171919
Kahaly GJ, Bartalena L, Hegedus L, Leenhardt L, Poppe K, Pearce SH (2018) 2018 European thyroid association guideline for the management of graves’ hyperthyroidism. Eur Thyroid J 7:167–186. https://doi.org/10.1159/000490384
Andersen SL, Olsen J, Laurberg P (2016) Antithyroid drug side effects in the population and in pregnancy. J Clin Endocrinol Metab 101:1606–1614. https://doi.org/10.1210/jc.2015-4274
Jaeschke H, Eszlinger M, Lueblinghoff J, Coslovsky R, Paschke R (2011) Prolonged inappropriate TSH suppression during hypothyroidism after thyroid ablation in a patient with nonautoimmune familial hyperthyroidism. Horm Metab Res 43:500–504. https://doi.org/10.1055/s-0031-1277184
Alexander C, Bader JB, Schaefer A, Finke C, Kirsch CM (1998) Intermediate and long-term side effects of high-dose radioiodine therapy for thyroid carcinoma. J Nucl Med 39:1551–1554
Acknowledgements
We acknowledge the family for giving us permission to publish their case.
Funding
This work was partially supported by the Project for the Conceptual Development of Research Organization, Motol University Hospital (Ministry of Health, Czech Republic; 00064203) and Operational Programme Research and Development for Innovation (Ministry of Education, Youth and Sports, Czech Republic, co-financed by the EU; CZ.1.05/4.1.00/16.0337). Mutation analyses were further supported by the Ministry of Health of the Czech Republic (grant NU20-03–00285). The funding bodies had no impact on the study design, analyses, or data interpretation.
Author information
Authors and Affiliations
Contributions
Martin Bezdicka performed the functional study and drafted the Supplementary file. P.K. and Mariana Borecka performed the molecular genetics analyses of the participants. J.S. revealed hyperthyroidism in the boy and treated him until referral to the university center. D.S. and M.R. performed the flow cytometry. E.E. and Z.S. reviewed the case and gave important clinical hints and suggestions. J.M. supervised the site-directed mutagenesis and transformation procedures. O.S. treated the patient in the referral center, designed the study, and drafted the manuscript. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethical approval
This research was performed according to the appropriate ethical guidelines and the Declaration of Helsinki.
Consent to participate and for publication
The patient as well as his parents gave written informed consent to have their case published, including the pictures kindly provided by the family.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bezdicka, M., Kleiblova, P., Soucek, J. et al. Novel presentation of the c.1856A > G (p.Asp619Gly) TSHR gene-activating variant: relapsing hyperthyroidism in three subsequent generations manifesting in early childhood and an in vitro functional study. Hormones 20, 803–812 (2021). https://doi.org/10.1007/s42000-021-00299-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42000-021-00299-x