Abstract
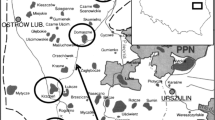
In semi-arid regions, eutrophic waters and stable climatic conditions favor the dominance of cyanobacteria, with frequent blooms. In this study, we hypothesized that the biomass of cyanobacteria will be higher during the dry period and assessed toxic cyanobacteria and cyanotoxins in the Argemiro de Figueiredo Reservoir (Paraíba, Brazil). We conducted 22 samplings between August 2007 and July 2009 at three sites (PC, confluence of tributaries; PNC, net cages; and PD, dam site). Significant seasonal and spatial differences were not observed for abiotic variables; however, reduction in pH, electrical conductivity, alkalinity, and dissolved oxygen occurred only during overflow events in the reservoir. Filamentous cyanobacteria were dominant mainly in the dry period, accounting for 90 % of the total biomass; six of eight dominant or co-dominant species of cyanobacteria were observed to be potential producers of toxins. Planktothrix agardhii (Gomont) Anagnostidis & Komárek was dominant at PC, PNC, and PD and formed perennial blooms, and microcystin-LR was identified in 14 of 16 blooming events. Thus, we suggest that stable conditions, availability of nutrients, and a reduced light regime during the dry period favors blooms of cyanobacteria.




Similar content being viewed by others
References
Azevedo SMFO (1998) Toxinas de Cianobactérias: Causas e consequências para a saúde pública. Med Online 3:1–19
Barbosa JEL, Mendes JS (2005) Estrutura da comunidade fitoplanctônica e aspetos físicos e químicos das águas do reservatório de Acauã-semiárido paraibano. Anais da X Reunião Brasileira de Ficologia (Sociedade Brasileira de Ficologia, coord). Museu Nacional, Rio de Janeiro, pp 339–360
Bláha L, Bláhová L, Kohoutek J, Adamovský O, Babica P, Maršálek B (2010) Temporal and spatial variability of cyanobacterial toxins microcystins in three interconnected freshwater reservoirs. J Serb Chem Soc 75:1303–1312
Bonilla S, Aubriot L, Soares MCS, Gonzalez-Piana M, Fabre A, Huszar VL, Lürling M, Antoniades D, Padisák J, Kruk C (2012) What drives the distribution of the bloom-forming cyanobacteria Planktothrix agardhii and Cylindrospermopsis raciborskii? FEMS Microbiol Ecol 79:594–607
Bouvy MA, Molica R, Oliveira S, Marinho M, Beker B (1999) Dynamics of a toxic cyanobacterial bloom (Cylindrospermopsis raciborskii) in a shallow reservoir in the semi-arid region of northeast Brazil. Aquat Microb Ecol 20:285–297
Carmichael WW (1996) Liver failure and human deaths at a haemodialysis centre in Brazil: microcystins as a major contributing factor. Harmful algal news: 11. IOC UNESCO, Paris
Carmichael WW, Gorhan PR (1981) The mosaic nature of toxic blooms of cyanobacteria. In: Carmichael WW (ed) The water environment: algal toxins and health. Plenum Press, New York, pp 161–172
Chellappa NT, Costa MAM (2003) Dominant and co-existing species of cyanobacteria from a eutrophicated reservoir of Rio Grande do Norte State, Brazil. Acta Oecol 124:3–10
Chellappa NT, Chellappa SL, Chellappa S (2008a) Harmful phytoplankton blooms and fish mortality in a eutrophicated reservoir of Northeast Brazil. Braz Arch Biol Technol 51:833–841
Chellappa NT, Borba JM, Rocha O (2008b) Phytoplankton community and physical–chemical characteristics of water in the public reservoir of Cruzeta, RN, Brazil. Braz J Biol 68:477–494
Clesceri LS, Eaton AD, Rice EW, Greenberg AE (2005) Standard methods for the examination of water and wastewater, 21st edn. American Public Health Association, Washington, DC
Cole GA (1994) Textbook of limnology. Waveland Press, Lake Zurich
Costa IAS, Azevedo SMFO, Senna PAC, Bernardo RR, Costa SM, Chellappa NT (2006) Occurrence of toxin-producing cyanobacterial blooms in a Brazilian semiarid reservoir. Braz J Biol 66:211–219
Costa IAS, Cunha SRS, Panosso RF, Araújo MFF, Melo JLS, Eskinazi-Sant’Anna EM (2009) Dinâmica de cianobactérias em reservatórios eutróficos do semi-árido do Rio Grande do Norte. Oecol Bras 13:382–401
Dantas EW, Moura NA, Bittencourt-Oliveira MC, Neto JDTA, Cavalcanti ADC (2008) Temporal variation of the phytoplankton community at short sampling intervals in the Mundaú Reservoir, Northeastern Brazil. Acta Bot Bras 22:970–982
Davis TW, Berry DL, Boyer GL, Gobler CJ (2009) The effects of temperature and nutrients on the growth and dynamics of toxic and non-toxic strains of Microcystis during cyanobacteria blooms. Harmful Algae 8:715–725
Fernandes VO, Cavati B, Oliveira LB, Souza BD (2009) Ecologia de cianobactérias: fatores promotores e consequências das florações. Oecol Bras 13:247–258
Figueredo CC, Giani A (2009) Phytoplankton community in the Tropical Lake of Lagoa Santa (Brazil): conditions favoring a persistent bloom of Cylindrospermopsis raciborskii. Limnologica 39:264–272
Golterman HL, Clymo RS, Ohnstad MAM (1978) Methods for physical and chemical analysis of fresh waters, 2nd edn. Blackwell Scientific Publications, Oxford, p 213
Hillebrand H, Dürselen C-D, Kirschtel D, Pollingher U, Zohary T (1999) Biovolume calculation for pelagic and benthic microalgae. J Phycol 35:403–424
Hudnell KH, Dortch Q (2008) A synopsis of research needs identified at the interagency (ISOC-HAB). In: Hudnell HK (ed) Advances in experimental medicine and biology. Springer, New York, pp 17–35
Huszar VLM, Silva LHS, Marinho M, Domingos P, Sant’Anna CL (2000) Cyanoprokaryote assemblages in eight productive tropical Brazilian waters. Hydrobiologia 424:67–77
Jensen JP, Jeppesen E, Olrik K, Kristensen P (1994) Impact of nutrients and physical factors on the shift from cyanobacterial to chlorophyte dominance in shallow Danish lakes. Can J Fish Aquat Sci 51:1692–1699
Jochimsen EM, Carmichael WW, An J, Cardo DM, Cookson ST, Holmes CEM, Antunes BC, Melo FDA, Lyra TM, Barreto VST, Azevedo SMFO, Jarvis WR (1998) Liver failure and death after exposure to microcystins at a hemodialysis center in Brazil. N Engl J Med 338:873–878
Kardinaal WEA, Tonk L, Janse I, Hol S, Slot P, Huisman J, Visser PM (2007) Competition for light between toxic and nontoxic strains of the harmful cyanobacterium Microcystis. Appl Environ Microbiol 73:2939–2946
Kotak BG, Kenefick SL, Fritz DL, Rousseaux CG, Prepas EE, Hrudey SE (1993) Occurrence and toxicological evaluation of cyanobacterial toxins in Alberta lakes and farm dugouts. Water Res 27:495–506
Kuiper-Goodman T, Falconer I, Fitzgerald J (1999) Human health aspects. In: Chorus I, Bartram J (eds) A guide to their public health consequences, monitoring and management. E and FN Spon, London, pp 113–153
Kwang-Guk A, Jones JR (2000) Factors regulating bluegreen dominance in a reservoir directly influenced by the Asian monsoon. Hydrobiologia 432:37–48
Lund JWG, Kipling C, Lecren ED (1958) The inverted microscope method of estimating algal numbers and the statistical basis of estimation by counting. Hydrobiologia 11:143–170
Magalhães VF, Soares RM, Azevedo SMFO (2001) Microcystin contamination in fish from the Jacarepaguá Lagon (Rio de Janeiro, Brazil): ecological implication and human health risk. Toxicon 39:1077–1085
Meriluoto J, Spoof L (2005) Solid phase extraction of microcystin in water samples. In: Meriluoto J, Codd G (eds) Toxic cyanobacterial monitoring and cyanotoxin analysis. Abo Akademi University Press, Turku
Moura AN, Bittencourt-Oliveira MC, Dantas EW, Arruda-Neto JDT (2007) Phytoplanktonic associations: a tool to understand dominance events in a tropical Brazilian reservoir. Acta Bot Bras 21:641–648
Ozawa K, Fujioka H, Muranaka M, Yokoyama A, Katagami Y, Homma T, Ishikawa K, Tsujimura S, Kumagai M, Watanabe MF, Park HD (2005) Spatial distribution and temporal variation of Microcystis species composition and microcystin concentration in Lake Biwa. Environ Toxicol 20:270–276
Padisák J, Reynolds CS (1998) Selection of phytoplankton associations in Lake Balaton, Hungary, in response to eutrophication and restoration measures, with special reference to cyanoprokaryotes. Hydrobiologia 384:41–53
Padisák J, Crossetti LO, Naselli-Flores L (2009) Use and misuse in the application of the phytoplankton functional classification: a critical review with updates. Hydrobiologia 621:1–19
Paerl HW, Tucker CS (1995) Ecology of blue-green algae in aquaculture ponds. J World Aquac Soc 26:109–131
Panosso R, Costa IAS, Souza NR, Attayde JL, Cunha SRS, Gomes FCF (2007) Cianobactérias e cianotoxinas em reservatórios do Estado do Rio Grande do Norte e o potencial controle das florações pela tilápia do Nilo (Oreochromis niloticus). Oecol Bras 11:433–449
Park HD, Iwami C, Watanabe MF, Harada KI, Tokio O, Hidetake H (1998) Temporal variabilities of the concentration of intra- and extracellular microcystins and toxic microcystins species in a hypertrophic lake, Lake Suwa, Japan. Environ Toxicol Water Qual 13:61–72
Pawlik-Skowrońska B, Pirszel J, Kornijów R (2008) Spatial and temporal variation in microcystin concentrations during perennial bloom of Planktothrix agardhii in a hypertrophic lake. Ann Limnol Int J Limnol 44:145–150
Ros J (1979) Prácticas de ecología. Editorial Omega, Barcelona
Scheffer M (1998) Ecology of shallow lakes. Chapman and Hall, London
Scheffer M, Rinaldi S, Gragnani A, Mur LR, van Nes EH (1997) On the dominance of filamentous cyanobacteria in shallow, turbid lakes. Ecology 78:272–282
Semarh (2007) State plan for water resources of the Paraíba-PERH-PB. Executive Review and Atlas, João Pessoa
Silva DF, Sousa FAZ, Kayano MT, Araujo LE (2008) Acompanhamento climático das bacias hidrográficas do rio Mundú (AL e PE) e do rio Paraíba (PB). Eng Amb 5:79–93
Sivonen K (1990) Effect of light, temperature, nitrate, orthophosphate and bacteria on growth of and hepatotoxin production by Oscillatoria agardhii strains. Appl Environ Microbiol 56:2658–2666
Soares MCS, Huszar VLM, Miranda MN, Mello MM, Roland F, Lürling M (2013) Cyanobacterial dominance in Brazil: distribution and environmental preferences. Hydrobiologia 717:1–12
Sun J, Liu D (2003) Geometric models for calculating cell biovolume and surface area for phytoplankton. J Plankton Res 25:1331–1346
Talling JF (1987) The phytoplankton of Lake Victoria (East Africa). Arch Hydrobiol 25:229–256
Ter Braak CJF (1986) Canonical correspondence analysis: a new eigenvector technique for multivariate direct gradient analysis. Ecology 67:1167–1179
Toledo Jr AP, Talarico M, Chinez SJ, Agudo EG (1983) Aplicação de modelos simplificados para a avaliação do processo da eutrofização em lagos e reservatórios tropicais. In: 12 Congresso Brasileiro de Engenharia Sanitária e Ambiental. Anais, Camboriú
Utermöhl H (1958) Zur Vervollkommung der quantitativen Phytoplankton-Methodik. Mitt int Ver Limnol Stuttg 9:1–38
Vincent WF (1989) Cyanobacterial growth and dominance in two eutrophic lakes: review and synthesis. Arch Hydrobiol Beih Ergeb Limnol 32:239–254
Watanabe MF, Oishi S (1985) Effects of environmental factors on toxicity of a cyanobacterium (Microcystis aeruginosa) under culture conditions. Appl Environ Microbiol 49:1342–1344
Wetzel RG, Likens GE (2000) Limnological analyses. Springer, New York
Wiedner C, Visser PM, Fastner J, Metcalf JS, Codd GA, Mur LR (2003) Effects of light on the microcystin content of Microcystis strain PCC 7806. Appl Environ Microbiol 69:1475–1481
Zurawell RW, Kotak BG, Prepas EE (1999) Influence of lake trophic status on the occurrence of microcystin-LR in the tissue of pulmonate snails. Freshw Biol 42:707–718
Acknowledgments
The authors would like to thank the National Council for Scientific and Technological Development for the financial support through Universal Project MCT/CNPq O2/2006 and for Granting a PhD Scholarship to the first author (MCT/CNPq/CT-Hidro Public Notice 040/2006); the Sanitation Laboratory of the Federal University of Campina Grande; and the Microplankton Laboratory of the Marine Resources Study and Research Center of the Federal University of Paraíba.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lins, R.P.M., Barbosa, L.G., Minillo, A. et al. Cyanobacteria in a eutrophicated reservoir in a semi-arid region in Brazil: dominance and microcystin events of blooms. Braz. J. Bot 39, 583–591 (2016). https://doi.org/10.1007/s40415-016-0267-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40415-016-0267-x