Abstract
Background
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) for the treatment of type 2 diabetes mellitus are known to delay gastric emptying (GE). The potential effect of the GLP-1 RA dulaglutide on the pharmacokinetics (PK) of four orally administered drugs and on the pharmacodynamic (PD) effect of warfarin was investigated.
Methods
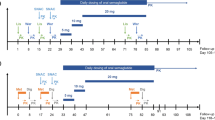
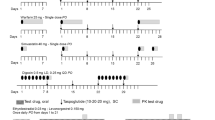
In four separate clinical pharmacology studies, digoxin, warfarin, atorvastatin and Ortho-Cyclen® were orally administered to healthy subjects with and without a subcutaneous dose of dulaglutide 1.5 mg. The effect of dulaglutide coadministration was assessed based on the PK parameters of key analytes. For warfarin PD, the effect of dulaglutide on the international normalized ratio (INR) was evaluated.
Results
Areas under the concentration–time curves (AUCs) with and without dulaglutide were similar for all analytes except atorvastatin, where it was reduced by 21%. Maximum concentrations (C max) were generally lower following coadministration with dulaglutide, with statistically significant reductions (90% confidence intervals of geometric least squares means ratios outside 0.80–1.25) for all analytes except R-warfarin. For all analytes, there was a general trend for the time to C max (t max) to increase following coadministration with dulaglutide. For warfarin, dulaglutide coadministration had no statistically significant effect on the maximum INR (INRmax); however, a 2% increase in area under the INR curve (AUCINR) was observed.
Conclusions
Dulaglutide did not affect the absorption of the tested medications to a clinically relevant degree. Based on the PK and PD evaluations, no dose adjustments for digoxin, warfarin, atorvastatin and Ortho-Cyclen® are recommended when coadministered with dulaglutide.
Clinical trial registration numbers
NCT01458210, NCT01436201, NCT01432938, and NCT01250834.







Similar content being viewed by others
Notes
AUC during one dosing interval (AUCτ) for digoxin.
References
Trulicity (prescribing information). Lilly USA, LLC, Indianapolis; 2014. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2014/125469Orig1s000Lbl.pdf. Accessed 1 Sep 2016.
Trulicity (summary of product characteristics). Eli Lilly Nederland BV, Utrecht; 2014. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/002825/WC500179470.pdf. Accessed 21 Oct 2016.
Faraj J, Melander O, Sundkvist G, Olsson R, Thorsson O, Ekberg O, et al. Oesophageal dysmotility, delayed gastric emptying and gastrointestinal symptoms in patients with diabetes mellitus. Diabet Med. 2007;24(11):1235–9.
Jones KL, Horowitz M, Wishart JM, Maddox AF, Harding PE, Chatterton BE. Relationships between gastric emptying, intragastric meal distribution and blood glucose concentrations in diabetes mellitus. J Nucl Med. 1995;36(12):2220–8.
Stacher G, Lenglinger J, Bergmann H, Schneider C, Brannath W, Festa A, et al. Impaired gastric emptying and altered intragastric meal distribution in diabetes mellitus related to autonomic neuropathy. Dig Dis Sci. 2003;48(6):1027–34.
Woodyatt RT, Sansum WD, Wilder RM. Prolonged and accurately timed intravenous injection of sugar. JAMA. 1915;65(24):2067–70.
Gonlachanvit S, Hsu CW, Boden GH, Knight LC, Maurer AH, Fisher RS, et al. Effect of altering gastric emptying on postprandial plasma glucose concentrations following a physiologic meal in type-II diabetic patients. Dig Dis Sci. 2003;48(3):488–97.
Thompson DG, Wingate DL, Thomas M, Harrison D. Gastric emptying as a determinant of the oral glucose tolerance test. Gastroenterology. 1982;82(1):51–5.
O’Donovan DG, Doran S, Feinle-Bisset C, Jones KL, Meyer JH, Wishart JM, et al. Effect of variations in small intestinal glucose delivery on plasma glucose, insulin, and incretin hormones in healthy subjects and type 2 diabetes. J Clin Endocrinol Metab. 2004;89:3431–5.
Flint A, Raben A, Astrup A, Holst JJ. Glucagon-like peptide 1 promotes satiety and suppresses energy intake in humans. J Clin Invest. 1998;101(3):515–20.
Naslund E, King N, Mansten S, Adner N, Holst JJ, Gutniak M, et al. Prandial subcutaneous injections of glucagon-like peptide-1 cause weight loss in obese human subjects. Br J Nutr. 2004;91:439–46.
Meier JJ, Gallwitz B, Salmen S, Goetze O, Holst JJ, Schmidt WE, et al. Normalization of glucose concentrations and deceleration of gastric emptying after solid meals during intravenous glucagon-like peptide 1 in patients with type 2 diabetes. J Clin Endocrinol Metab. 2003;88(6):2719–25.
Linnebjerg H, Park S, Kothare PA, Trautmann ME, Mace K, Fineman M, et al. Effect of exenatide on gastric emptying and relationship to postprandial glycemia in type 2 diabetes. Regul Pept. 2008;151(1–3):123–9.
Meier JJ, Rosenstock J, Hincelin-Méry A, Roy-Duval C, Delfolie A, Coester HV, et al. Contrasting effects of lixisenatide and liraglutide on postprandial glycemic control, gastric emptying, and safety parameters in patients with type 2 diabetes on optimized insulin glargine with or without metformin: a randomized, open-label trial. Diabetes Care. 2015;38(7):1263–73.
Trujillo JM, Nuffer W. Albiglutide: a new GLP-1 receptor agonist for the treatment of type 2 diabetes. Ann Pharmacother. 2014;48(11):1494–501.
Loghin C, de la Peña A, Cui X, Chien J. Gastric emptying effects of once weekly dulaglutide in patients with type 2 diabetes mellitus (poster). Presented at the American Association of Clinical Endocrinologists, 23rd annual scientific and clinical congress, 14–18 May 2014, Las Vegas.
Meier JJ. GLP-1 receptor agonists for individualized treatment of type 2 diabetes mellitus. Nat Rev Endocrinol. 2012;8:728–42.
Lanoxin (prescribing information). Covis Pharmaceuticals Inc.; 2015. http://www.accessdata.fda.gov/drugsatfda_docs/label/2015/009330s030lbl.pdf.Accessed 21 Oct 2016.
Coumadin (prescribing information). Bristol-Myers Squibb Company; 2016. http://www.accessdata.fda.gov/drugsatfda_docs/label/2016/009218s116lbl.pdf. Accessed 21 Oct 2016.
O’Reilly RA, Aggeler PM, Leong LS. Studies on the coumarin anticoagulant drugs: the pharmacodynamics of warfarin in man. J Clin Invest. 1963;42(10):1542–51.
Soon D, Kothare PA, Linnebjerg H, Park S, Yuen E, Mace KF, et al. Effect ofexenatideon the pharmacokinetics and pharmacodynamics of warfarin in healthy Asian men. J Clin Pharmacol. 2006;46(10):1179–87.
Takahashi H, Echizen H. Pharmacogenetics of warfarin elimination and its clinical implications. Clin Pharmacokinet. 2001;40(8):587–603.
Kaminsky LS, Zhang ZY. Human P450 metabolism of warfarin. Pharmacol Ther. 1997;73(1):67–74.
Lipitor (prescribing information). Pfizer Inc.; 2015. http://www.accessdata.fda.gov/drugsatfda_docs/label/2015/020702s065lbl.pdf. Accessed 21 Oct 2016.
Ortho-Cyclen (prescribing information). Janssen Pharmaceuticals, Inc.; 2015. http://www.accessdata.fda.gov/drugsatfda_docs/label/2015/019653s056,019697s052,021690s001lbl.pdf. Accessed 21 Oct 2016.
Steinijans VW, Sauter R, Hauschke D, Diletti E, Schall R, Luus HG, et al. Reference tables for the intrasubject coefficient of variation in bioequivalence studies. Int J Clin Pharmacol Ther. 1995;33(8):427–30.
Stern RH, Smithers JA, Olsen SC. Atorvastatin does not produce a clinically significant effect on the pharmacokinetics of terfenadine. J Clin Pharmacol. 1998;38(8):753–7.
Kearney BP, Mathias A. Lack of effect of tenofovirdisoproxilfurmarate on pharmacokinetics of hormonal contraceptives. Pharmacotherapy. 2009;29(8):924–9.
Kothare PA, Soon DK, Linnebjerg H, Park S, Chan C, Yeo A, et al. Effect of exenatide on the steady-state pharmacokinetics of digoxin. J Clin Pharmacol. 2005;45(9):1032–7.
Narwal R, Akhlaghi F, Asberg A, Hermann M, Rosenbaum SE. Development of a population pharmacokinetic model for atorvastatin acid and its lactone metabolite. Clin Pharmacokinet. 2010;49(10):693–702.
Schmitt-Hoffmann AH, Roos B, Sauer J, Schleimer M, Schoetzau A, Leese PT, et al. Influence of alitretinoin on the pharmacokinetics of the oral contraceptive ethinylestradiol/norgestimate. Clin Exp Dermatol. 2011;36(2):4–11.
Kothare PA, Seger ME, Northrup J, Mace K, Mitchell MI, Linnebjerg H. Effect of exenatide on the pharmacokinetics of a combination oral contraceptive in healthy women: an open-label, randomised, crossover trial. BMC Clin Pharmacol. 2012;12(8):1–9.
US FDA. Guidance for industry. Drug interaction studies—study design, data analysis, implications for dosing, and labeling recommendations. US FDA; 2012.
Bush M, Scott R, Watanalumlerd P, Zhi H, Lewis E. Effects of multiple doses of albiglutide on the pharmacokinetics, pharmacodynamics, and safety of digoxin, warfarin, or a low-dose oral contraceptive. Postgrad Med. 2012;124(6):55–72.
Hurren KM, Pinelli NR. Drug–drug interactions with glucagon-like peptide-1 receptor agonists. Ann Pharmacother. 2012;46(5):710–7.
Acknowledgements
The authors thank the trial investigators, trial staff, and trial participants for their contributions. They would also like to express their gratitude to Dr. Helen Salter and Dr. Mark Stead for providing medical writing support, and Dr. Jenny Chien for providing scientific review and technical support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was sponsored by Eli Lilly and Company.
Conflict of interest
Amparo de la Peña, Xuewei Cui, Jeanne Geiser, and Corina Loghin are employees and shareholders of Eli Lilly and Company.
Scientific meeting presentation
This work was presented in part at the annual meetings of the American Association of Clinical Endocrinologists (AACE), Nashville, TN, USA, 13–17 May 2015, and the American Society for Clinical Pharmacology and Therapeutics (ASCPT), New Orleans, LA, USA, 3–7 March 2015.
Rights and permissions
About this article
Cite this article
de la Peña, A., Cui, X., Geiser, J. et al. No Dose Adjustment is Recommended for Digoxin, Warfarin, Atorvastatin or a Combination Oral Contraceptive When Coadministered with Dulaglutide. Clin Pharmacokinet 56, 1415–1427 (2017). https://doi.org/10.1007/s40262-017-0531-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40262-017-0531-7