Abstract
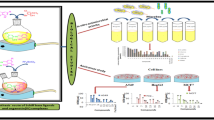
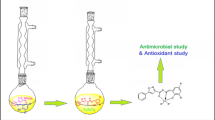
In this study, firstly, 3-bromopropanenitrile(2a), ethyl bromoacetate(2b), and 2-bromoethan-1-amine(2c) were added to 5,6-dimethylbenzimidazole(1) and symmetrically-connected N-heterocyclic carbenes(NHC)[(3a–3c)] were synthesized. Then, the NHC precursor compounds were reacted with PdCl2 and Pd (II)-NHC complexes[(4a—4c)] were obtained. The synthesized NHC precursors(3a–3c) and Pd( II)-NHC complexes(4a—4c) were characterized by different spectroscopic methods. The antibacterial activities of these products were measured against bacteria and fungus with the disc diffusion method. Pd(II)-NHC complexes had higher antibacterial activity than the NHC precursors. The synthesized compounds showed antimicrobial activity with different properties from each other. In vitro biological activities of salts and metal complexes on selected bacteria were determined by the broth dilution method based on Clinical and Laboratory Standards Institute guidelines. 1,3-Bis-(2-hydroxyethyl) imidazolinium bromide and 3-(2-ethoxy-2-oxoethyl)-1-(3-aminopropyl)-1H-imidazole-3-ium bromide had activity against Escherichia coli.

Similar content being viewed by others
References
Glorius F., N-Heterocyclic Carbenes in Transition Metal Catalysis, Springer-Verlag, Berlin, 2006
Wurtz S., Glorius F., Acc. Chem. Res., 2008, 41, 1523
Hashmi A. S. K., Lothschuetz C., Graf K., Haeffner T., Schuster A., Rominger F., Adv. Synth. and Catal., 2011, 353(9), 1407
Oehninger L., Rubbiani R., Ott I., Dalton Trans., 2013, 42(16), 3269
Cisnetti F., Gibard C., Gautier A., J. Organomet. Chem., 2015, 782, 22
Fortman G. C., Nolan S. P., Chem. Soc. Rev., 2011, 40(10), 5151
Nemcsok D., Wichmann K., Frenking G., Organometallics, 2004, 23(15), 3640
Gaillard S., Nun P., Slawin A. M. Z., Nolan S. P., Organometallics, 2010, 29(21), 5402
Li W. S., Wang C. H., Ko S., Chang T. T., Jen Y. C., Yao C. F., More S. V., Jao S. C., Journal of Medicinal Chemistry, 2012, 55(4), 1583
Peris E., Crabtree R. H., Coordination Chemistry Reviews, 2004, 248(21–24), 2239
Ray S., Mohan R., Singh J. K., Samantaray M. K., Shaikh M. M., Panda D., Ghosh P., J. Am. Chem. Soc., 2007, 129(48), 15042
Kleinman E.F., The Bimolecular Aliphatic Mannich and Related Reactions in Comprehensive Organic Synthesis, Pergamon Press, Oxford, 1991
Fekri A., Keshk E. M., Khalil A. G. M., Taha I., Mol. Divers, 2022, 26, 781
Scattolin T., Bortolamiol E., Palazzolo S., Caligiuri I., Perin T., Canzonieri V., Demitri N., Rizzolio F., Cavallo L., Dereli B., Mane M. V., Nolan S. P., Visentin F., Chem. Commun., 2020, 56, 12238
Liu J., Zhou X., Rao H., **ao F., Li C. J., Chemistry: A European Journal, 2011, 17(29), 7996
Ofele K., Journal of Organometallic Chemistry, 1968, 12, 42
Wanzlick H. W., Schönherr H. J., Angewandte Chemie International Edition, 1968, 7(2), 141
Peris E., Chemical Reviews, 2018, 118(19), 9988
Yang J., J. Coord. Chem., 2017, 70, 441
Liang R. X., Jia Y. X., Acc. Chem. Res., 2022, 55(5), 734
Ehrentraut A., Zapf A., Beller M., Synlett., 2000, 11, 1589
**ao J., Wang L., Zhang H., Ma N., Tao M., Zhang W., Chemical Engineering Science, 2022, 247(16), 117053
Samiee S., Shiralinia A., Hoveizi E., Gable R. W., Inorganica Chimica Acta, 2022, 538, 120964
Gu S., Chen C., Qiu H., Chen W., Current Organic Chemistry, 2011, 15, 3291
Guisado-Barrios G., Hiller J., Peris E., Chem. Eur. J., 2013, 19, 10405
Parmar D., Sugiono E., Raja S., Rue** M., Chem. Rev., 2014, 114, 9047
Burrows A. D., Sci. Prog., 2002, 85, 199
Crabtree R. H., New J. Chem., 2011, 35, 18
Dahm G., Borre E., Guichard G., Bellemin-Laponnaz S., Eur. J. Inorg. Chem., 2015, 2015(10), 1665
Simic D., Zaric M., Nikolic I., Zivkovic-Zaric R., Canovic P., Kocovic A., Radojevic I., Rakovic I., Jovicic S., Petrovic D., Stojkovic D., Vukovic N., Kacaniova M., Vukic M., Jevtic V., Dalton Transactions, 2022, 51, 1191
Clinical and Laboratory Standards Institute, Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts, Approved standard—Second Edition, NCCLS Document M27-A2(ISBN 1-56238-469-4), Wayne, Pennsylvania, 2002
Clinical and Laboratory Standards Institute, Performance Standards for Antimicrobial Susceptibility Testing, Twenty-second Informational Supplement, CLSI Document M100-S22, Wayne, Pennsylvania, 2012
Kaya E., Ozbilge H., Turk. J. Med. Sci., 2012, 42(2), 325
Altun Ö., Koçer M. Ö., J. Mol. Struc., 2021, 1224(129242), 1
Cajiao N., Mora H., Palencia M., Journal of Science with Technological Applications, 2016, 1(1), 77
Cai W., Wang J., Chu C., Chen W., Wu C., Liu G., Advenced Science, 2018, 6(1), 1801526
Ma S., Zhou Z., Ran G., **e J., Luo X., Li Y., Wang L., Journal of Hazardous Materials, 2022, 422, 126785
Geary W. J., Coord. Chem. Rev., 1971, 7(1), 81
Kettle S. F. A., Coordination Compounds, Thomas Nelson and Sons, London, 1975
Dyer J. R., Application of Absorption Spectroscopy of Organic Compounds, Prentice-Hall, New Jersey, 1965
Lever A. B. P., Inorganic Electronic Spectroscopy, Elsevier, New York, 1984
Kang J. G., Cho D. H., Park C., Kang S. K., Kim I. T., Lee S. W., Lee H. H., Lee Y. N., Lim D. W., Lee S. J., Kim S. H., Bae Y. J., Bull. Korean Chem. Soc., 2008, 29(3), 599
Jambi S. M. S., Kandil S. S., J. Mater. Environ. Sci., 2012, 3(3), 591
Lambert J. B., Shurwell H. F., Verbit L., Cooks R. G., Stout G. H., Organic Structural Analysis, MacMillan, New York, 1976
Nakamato K., Infrared and Raman Spectra of Inorganic and Coordination Compounds, John Wiley and Sons, New York, 1997
Socrates G., Infrared Characterization Group Frequencies: Tables and Charts, 3rd Edition, John Wiley and Sons, London, 2004
Giovagnini L., Ronconi L., Aldinucci D., Lorenzon D., Sitran S., Fregona D., J. Med. Chem., 2005, 48(5), 1588
Salem M. A., Bakr E. A., El-Attar H. G., Spectrochim. Acta, Part A, 2018, 188, 155
Warren B. E., X-Ray Diffraction, Dover, New York, 1990
Barbosa H. F. G., Attjioui M., Ferreira A. P. G., Dockal E. R., El Gueddari N. E., Moerschbacher B. M., Cavalheiro E. T. G., Molecules, 2017, 22(1987), 1
Kavitha K., Reddy L., Arab. J. Chem., 2016, 9(5), 640
Yılmaz V. T., Ertem A., Guney E., Buyukgungor O., Z. Anorg. Allg. Chem., 2010, 636(3/4), 610
Maskovic J. M., Maskovic P. Z., Koval’chuk T. V., Loginova N. V., Trifunovic S. R., Der Chemica Sinica, 2018, 9(1), 535
Al-Hamdani A. A. S., Balkhı A. M., Falah A., Shaker S. A., J. Chil. Chem. Soc., 2015, 60(1), 2774
Pahontu E., Paraschivescu C., Ilies D. C., Poirier D., Oprean C., Paunescu V., Gulea A., Rosu T., Bratu O., Molecules, 2016, 21(5), 1
Kumar V. A., Sarala Y., Siddikha A., Vanitha S., Babu S., Reddy A. V., J. Appl. Pharm. Sci., 2018, 8(4), 071
Mihalache M., Negreanu-Pirjol T., Dumitraşu F., Draghici C., Calinescu M., J. Serb. Chem. Soc., 2018, 83(3), 271
Acknowledgements
This work was supported by the Trakya University Research Fund(No. TUBAP-2014-106).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflicts of interest.
Rights and permissions
About this article
Cite this article
Türkyilmaz, M., Dönmez, M. & Altun, Ö. Synthesis, Spectral Analysis and Antimicrobial Activity of New Pd (II) Complexes Involving 5,6-Dimethylbenzimidazole. Chem. Res. Chin. Univ. 39, 968–975 (2023). https://doi.org/10.1007/s40242-023-2355-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-023-2355-3