Abstract
Background
The hyperglycemic condition in diabetes induces cellular senescence in vascular endothelial cells and causes cardiovascular complications. Alpha-mangostin is a xanthone found in Garcinia mangostana, and has shown protective effects in metabolic syndrome.
Objective
In this study, the anti-senescence effects of alpha-mangostin in the hyperglycemic condition are investigated.
Methods
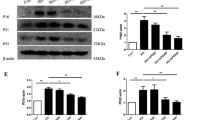
HUVECs were incubated with high glucose for 6 days and co-treated by metformin or alpha-mangostin. After 6 days, cell viability, reactive oxygen species, the percentage of senescent cells, secretory interleukin-6, and the expression of SIRT1, AMPK, p53 and p21 were measured.
Results
High glucose (60 mM) significantly decreased cellular viability and increased reactive oxygen species and cellular senescence through the reduction of senescence-associated β-galactosidase activity. Moreover, high glucose increased the protein levels of p53, acetyl-p53 and p21. The protein levels of SIRT1 and total AMPK were decreased by high glucose. High glucose increased the secretion of IL-6. Alpha-mangostin (1.25 μM) and metformin (50 μM) reversed the toxic effects of high glucose in HUVECs.
Conclusion
These results show that alpha-mangostin, similar to metformin, has anti-senescence effects in high-glucose conditions, which is probably due to its antioxidant activity through the SIRT1 pathway. Alpha-mangostin has previously shown anti-inflammatory effects and metabolic status improvement in animal and clinical studies. Therefore, this natural agent can be considered as a supplement to prevent vascular complications caused by high glucose in patients with diabetes.

Graphical abstract













Similar content being viewed by others
Abbreviations
- AGEs:
-
Advanced glycation end products
- Akt:
-
Protein kinase B
- alpha-MG:
-
Alpha-mangostin
- AMPK:
-
5’-AMP-activated protein kinase
- eNOS:
-
Endothelial nitric oxide synthase
- FOXO1:
-
Forkhead box O1
- FoxO3a:
-
Forkhead box O3
- HG:
-
High glucose
- HO-1:
-
Heme oxygenase 1
- HUVECs:
-
Human umbilical vein endothelial cells
- IL-6:
-
Interleukin-6
- JNK:
-
c-Jun N-terminal kinases
- LPS:
-
Lipopolysaccharides
- NF-κB:
-
Nuclear factor kappa-light-chain-enhancer of activated B cells
- NO:
-
Nitric oxide
- Nrf2:
-
Nuclear factor erythroid 2-related factor 2
- p38-MAPK:
-
P38 mitogen-activated protein kinases
- PGC-1α:
-
Peroxisome proliferator-activated receptor gamma coactivator 1-alpha
- PI3K:
-
Phosphatidylinositol-4,5-bisphosphate 3-kinase
- PPAR:
-
Peroxisome proliferator-activated receptor
- ROS:
-
Reactive oxygen species
- SASP:
-
Senescence-associated secretory phenotype
- SA-β-GAL:
-
Senescence-associated beta galactosidase
- SIRT1:
-
Sirtuin 1
- TNF-α:
-
Tumor necrosis factor alpha
References
Cade WT. Diabetes-related microvascular and macrovascular diseases in the physical therapy setting. Phys Ther. 2008;88(11):1322–35. https://doi.org/10.2522/ptj.20080008.
Stolar M. Glycemic control and complications in type 2 diabetes mellitus. Am J Med. 2010;123(3 Suppl):S3–11. https://doi.org/10.1016/j.amjmed.2009.12.004.
Burton DGA, Faragher RGA. Obesity and type-2 diabetes as inducers of premature cellular senescence and ageing. Biogerontology. 2018;19(6):447–59. https://doi.org/10.1007/s10522-018-9763-7.
Goligorsky MS. Vascular endothelium in diabetes. American journal of physiology Renal physiology. 2017;312(2):F266–f75. https://doi.org/10.1152/ajprenal.00473.2016.
Muller FL, Lustgarten MS, Jang Y, Richardson A, Van Remmen H. Trends in oxidative aging theories. Free Radic Biol Med. 2007;43(4):477–503.
Harman D. Aging: overview. Ann N Y Acad Sci. 2001;928(1):1–21.
Shin DH, Lee SJ, Kim JS, Ryu JH, Kim JS. Synergistic effect of immunoliposomal gemcitabine and bevacizumab in glioblastoma stem cell-targeted therapy. J Biomed Nanotechnol. 2015;11(11):1989–2002.
Signer RAJ, Morrison SJ. Mechanisms that regulate stem cell aging and life span. Cell Stem Cell. 2013;12(2):152–65. https://doi.org/10.1016/j.stem.2013.01.001.
Brunet A, Sweeney LB, Sturgill JF, Chua KF, Greer PL, Lin Y et al. Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Science (New York, NY). 2004;303(5666):2011–5. https://doi.org/10.1126/science.1094637.
Arunachalam G, Samuel SM, Marei I, Ding H, Triggle CR. Metformin modulates hyperglycaemia-induced endothelial senescence and apoptosis through SIRT1. Br J Pharmacol. 2014;171(2):523–35. https://doi.org/10.1111/bph.12496.
Bumrungpert A, Kalpravidh RW, Chitchumroonchokchai C, Chuang CC, West T, Kennedy A, et al. Xanthones from mangosteen prevent lipopolysaccharide-mediated inflammation and insulin resistance in primary cultures of human adipocytes. J Nutr. 2009;139(6):1185–91. https://doi.org/10.3945/jn.109.106617.
Jariyapongskul A, Areebambud C, Suksamrarn S, Mekseepralard C. Alpha-mangostin attenuation of hyperglycemia-induced ocular hypoperfusion and blood retinal barrier leakage in the early stage of type 2 diabetes rats. Biomed Res Int. 2015;2015:785826. https://doi.org/10.1155/2015/785826.
Choi Y. al. e. α-Mangostin regulates hepatic Steatosis and obesity through SirT1-AMPK and PPARγ pathways in high-fat diet-induced obese mice. J Agric Food Chem. 2015;63:8399–406.
Chae HS, Kim YM, Bae JK, Sorchhann S, Yim S, Han L, et al. Mangosteen extract attenuates the metabolic disorders of high-fat-fed mice by activating AMPK. J Med Food. 2016;19(2):148–54. https://doi.org/10.1089/jmf.2015.3496.
Taher M, Tg Zakaria TM, Susanti D, Zakaria ZA. Hypoglycaemic activity of ethanolic extract of Garcinia mangostana Linn in normoglycaemic and streptozotocin-induced diabetic rats. BMC complementary and alternative medicine 2016;16:135. https://doi.org/10.1186/s12906-016-1118-9.
Zhang E, Guo Q, Gao H, Xu R, Teng S, Wu Y. Metformin and resveratrol inhibited high glucose-induced metabolic memory of endothelial senescence through SIRT1/p300/p53/p21 pathway. PLoS One. 2015;10(12):e0143814. https://doi.org/10.1371/journal.pone.0143814.
Rameshrad M, Imenshahidi M, Razavi BM, Iranshahi M, Hosseinzadeh H. Bisphenol a vascular toxicity: protective effect of Vitis vinifera (grape) seed extract and resveratrol. Phytotherapy research : PTR. 2018. https://doi.org/10.1002/ptr.6175.
Wang H, Joseph JA. Quantifying cellular oxidative stress by dichlorofluorescein assay using microplate reader. Free Radic Biol Med. 1999;27(5–6):612–6.
Tong YF, Liu Y, Hu ZX, Li ZC. A a. Protocatechuic aldehyde inhibits TNF-alpha-induced fibronectin expression in human umbilical vein endothelial cells via a c-Jun N-terminal kinase dependent pathway. Experimental and therapeutic medicine. 2016;11(1):277–82. https://doi.org/10.3892/etm.2015.2896.
Tousian Shandiz H, Razavi BM, Hosseinzadeh H. Review of Garcinia mangostana and its xanthones in metabolic syndrome and related complications. Phytother Res. 2017;31(8):1173–82. https://doi.org/10.1002/ptr.5862.
Detaille D, Guigas B, Chauvin C, Batandier C, Fontaine E, Wiernsperger N, et al. Metformin prevents high-glucose-induced endothelial cell death through a mitochondrial permeability transition-dependent process. Diabetes. 2005;54(7):2179–87.
Ding A-J, Zheng S-Q, Huang X-B, **ng T-K, Wu G-S, Sun H-Y, et al. Current perspective in the discovery of anti-aging agents from natural products. Natural products and bioprospecting. 2017;7(5):335–404. https://doi.org/10.1007/s13659-017-0135-9.
Jittiporn K. Moongkarndi p, samer j, suvitayavat w. protective effect of α-Mangostin on high glucose induced endothelial cell apoptosis. Walailak J Sci & Tech. 2017;15(8):579–87.
Kitada M, Ogura Y, Koya D. The protective role of Sirt1 in vascular tissue: its relationship to vascular aging and atherosclerosis. Aging. 2016;8(10):2290–307. https://doi.org/10.18632/aging.101068.
Olmos Y, Sánchez-Gómez FJ, Wild B, García-Quintans N, Cabezudo S, Lamas S, et al. SirT1 regulation of antioxidant genes is dependent on the formation of a FoxO3a/PGC-1α complex. Antioxid Redox Signal. 2013;19(13):1507–21. https://doi.org/10.1089/ars.2012.4713.
D'Onofrio N, Servillo L, Balestrieri ML. SIRT1 and SIRT6 signaling pathways in cardiovascular disease protection. Antioxid Redox Signal. 2018;28(8):711–32. https://doi.org/10.1089/ars.2017.7178.
Huo J, Xu Z, Hosoe K, Kubo H, Miyahara H, Dai J, et al. Coenzyme Q10 prevents senescence and dysfunction caused by oxidative stress in vascular endothelial cells. Oxidative Med Cell Longev. 2018;2018:3181759. https://doi.org/10.1155/2018/3181759.
Olivieri F, Lazzarini R, Babini L, Prattichizzo F, Rippo MR, Tiano L, et al. Anti-inflammatory effect of ubiquinol-10 on young and senescent endothelial cells via miR-146a modulation. Free Radic Biol Med. 2013;63:410–20. https://doi.org/10.1016/j.freeradbiomed.2013.05.033.
Mohan S, Syam S, Abdelwahab SI, Thangavel N. An anti-inflammatory molecular mechanism of action of alpha-mangostin, the major xanthone from the pericarp of Garcinia mangostana: an in silico, in vitro and in vivo approach. Food Funct. 2018;9(7):3860–71. https://doi.org/10.1039/c8fo00439k.
Acknowledgements
This study was financially supported by Mashhad University of Medical Sciences (Grant No. 327 941389).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors state that there are no actual or potential conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tousian, H., Razavi, B.M. & Hosseinzadeh, H. Alpha-mangostin decreased cellular senescence in human umbilical vein endothelial cells. DARU J Pharm Sci 28, 45–55 (2020). https://doi.org/10.1007/s40199-019-00305-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40199-019-00305-z