Abstract
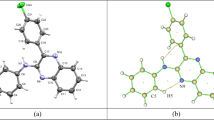
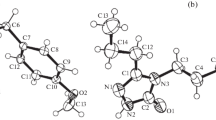
X-ray crystal structure analysis and quantum chemical calculations based on the density functional theory (DFT) were used for structural and electronic characterizations of three 2,3-dihydroquinazolin-4(1H)-ones (DHQZs). The occurrence of the C2-stereocenter in the heterocyclic ring causes the formations of both R- and S-enantiomers. X-ray diffraction technique indicates that these compounds exist as a racemic mixture in the crystal structure, and the enantiomers are orientated to each other via hydrogen bonding between the potential hydrogen donor (N3–H) and acceptor species (C4 = O group) in each layer under the formation of an enantio-syndio packing. Additional intermolecular and intramolecular interactions affect the orientations of the molecules adopted in the crystal packing, especially the orientation of the ring substitution. Most computational data, including the bond lengths and angles, are well in agreement with the experimental data. Dihedral angle scanning elucidates the effect of the nature and the location of the additional substituent on the aryl group at C2-position on the total energy content of the molecule.







Similar content being viewed by others
References
N.B. Patel, J.C. Pael, Med. Chem. Res. 20, 511 (2001)
N.D. Amnerkar, K.P.D. Bhusari, Eur. J. Med. Chem. 45, 149 (2010)
A. Kamal, E.V. Bharathi, J.S. Reddy, M.J. Ramaiah, D. Dastagiri, M.K. Reddy, A. Viswanath, T.L. Reddy, T.B. Shaik, S.N.C.V.L. Pushpavalli, M.P. Bhadra. Eur. J. Med. Chem. 46, 691 (2011)
H. Wu, X.L. **e, G. Liu, J. Comb. Chem. 12, 346 (2010)
A.M. Farghaly, R. Soliman, M.A. Khalil, A.A. Bekhit, A. El-Din, A. Bekhit, Boll. Chem. Farm. 141, 372 (2002)
H.R. Memarian, M. Abdoli-Senejani, Ultrasonics Sonochem. 15, 110 (2008)
H.R. Memarian, H. Sabzyan, M. Abdoli-Senejani, J. Mol. Struct. (Theochem) 813, 39 (2007)
H.R. Memarian, A. Farhadi, H. Sabzyan, M. Soleymani, J. Photochem. Photobiol. A: Chem. 209, 95 (2010)
H.R. Memarian, H. Sabzyan, A. Farhadi, Monatsh. Chem. 141, 1203 (2010)
H.R. Memarian, H. Sabzyan, M. Soleymani, M.H. Habibi, T. Suzuki, J. Mol. Struct. 998, 91 (2011)
H.R. Memarian, M. Soleymani, H. Sabzyan, J. Iran. Chem. Soc. 9, 805 (2012)
H.R. Memarian, M. Ranjbar, J. Mol. Catal. A 356, 46 (2012)
H.R. Memarian, H. Sabzyan, M. Ranjbar, Struct. Chem. 25, 85 (2014)
H.R. Memarian, S. Ebrahimi, J. Photochem. Photobiol. A: Chem. 271, 8 (2013)
H.R. Memarian, S. Ebrahimi, Monatsh. Chem. 145, 1545 (2014)
M. Abdollahi-Alibeik, E. Shabani, Chin. Chem. Lett. 22, 1163 (2011)
M.J. Frisch, G.W. Trucks, H.B. Schlegel, G.E. Scuseria, M.A. Robb, J.R. Cheeseman, V.G. Zakrzewski, J.A. Montgomery, Jr., R.E. Stratmann, J.C. Burant, S. Dapprich, J.M. Millam, A.D. Daniels, K.N. Kudin, M.C. Strain, O. Farkas, J. Tomasi, V. Barone, M. Cossi, R. Cammi, B. Mennucci, C. Pomelli, C. Adamo, S. Clifford, J. Ochterski, G.A. Petersson, P.Y. Ayala, Q. Cui, K. Morokuma, D.K. Malick, A.D. Rabuck, K. Raghavachari, J.B. Foresman, J. Ciolowski, J.V. Ortiz, B.B. Stefanov, G, Liu, A. Liashenko, P. Piskorz, I. Komaromi, R, Gomperts, R.L. Martin, D.J. Fox, T. Keith, M.A. Al-Laham, C.Y. Peng, A. Nanayakkara, C. Gonzalez, M. Challacombe, P.M.W. Gill, B. Johnson, W. Chen, M.W. Wong, J.L. Andres, M. Head-Gordon, E.S. Replogle, J.A. Pople, Gaussian 98 (Gaussian Inc., Pittsburg PA, 1998)
J.E. Carpenter, F. Weinhold, J. Mol. Struct. (Theochem) 169, 41 (1988)
A.E. Reed, L.A. Curtiss, F. Weinhold, Chem. Rev. 88, 899 (1988)
A.E. Reed, R.B. Weinstock, F. Weinhold, J. Chem. Phys. 83, 735 (1985)
L. **ao-Hong, T. Zheng-**n, Z. **an-Zhou, J. Mol. Struct. (Theochem) 900, 50 (2009)
Acknowledgments
We are thankful to the Research Council and Office of Graduate Studies of the University of Isfahan for their financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Memarian, H.R., Ebrahimi, S., Amiri Rudbari, H. et al. Inter- and intramolecular interactions in 2,3-dihydroquinazolin-4(1H)-ones: molecular structure and conformational analysis. J IRAN CHEM SOC 13, 1395–1404 (2016). https://doi.org/10.1007/s13738-016-0854-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-016-0854-6