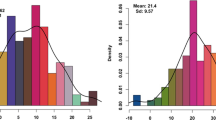
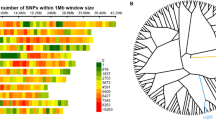
Abstract
Dwarfism in crops is a useful breeding trait. In this study, we aimed to identify a candidate gene controlling semi-dwarfism in watermelon (Citrullus lanatus) using a combination of genetic map** and quantitative trait loci (QTL)-seq. We evaluated phenotypes using an F2 and F2:3 population derived from a cross between a “Bush Sugar Baby” (BSB, semi-dwarf type) and a PCL-J1 (normal type) cultivar. Results indicated that a single recessive gene confers semi-dwarfism in BSB. We constructed a genetic map using 180 F2 plants and 336 single-nucleotide polymorphisms (SNPs), detected using genoty**-by-sequencing, and mapped the semi-dwarfism locus, sdw-1, between the SNP markers linked by 9.6 cM (0.99 Mb) on Chr. 9. QTL analysis pointed to the same genomic location for sdw-1 using single-marker analysis. Further, based on the QTL-seq, we identified a significant genomic region for sdw-1 that matched with the sdw-1-flanking region in the genetic linkage map. This 1-Mb region was narrowed down by map** three SNP markers developed from the QTL-seq data. The sdw-1 locus was mapped to the 0.44-Mb genomic region, which harbored 13 genes. One of the 13 genes, ClCG09G018320 (ATP-binding cassette (ABC) transporter B family member 19), showed the SNPs in its coding sequence, and cleaved amplified APS markers developed from those SNPs co-segregated with sdw-1 in fine map** using 620 F2 plants. Our results implied that a frameshift mutation in the ABC transporter gene and the resulting alteration in auxin transportation are the most likely to be responsible for semi-dwarfism in BSB.





Similar content being viewed by others
References
Arikit S, Wanchana S, Khanthong S, Saensuk C, Thianthavon T, Vanavichit A, Too**da T (2019) QTL-seq identifies cooked grain elongation QTLs near soluble starch synthase and starch branching enzymes in rice (Oryza sativa L.). Sci Rep 9:1–10. https://doi.org/10.1038/s41598-019-44856-2
Barboza L, Effgen S, Alonso-Blanco C, Kooke R, Keurentjes JJ, Koornneef M, Alcázar R (2013) Arabidopsis semidwarfs evolved from independent mutations in GA20ox1, ortholog to green revolution dwarf alleles in rice and barley. Proc Natl Acad Sci USA 110:15818–15823. https://doi.org/10.1073/pnas.1314979110
Branham SE, Wechter WP, Lambel S, Massey L, Ma M, Fauve J, Farnham MW, Levi A (2018) QTL-seq and marker development for resistance to Fusarium oxysporum f. sp. niveum race 1 in cultivated watermelon. Mol Breed 38:139. https://doi.org/10.1007/s11032-018-0896-9
Chen K et al (2009) BreakDancer: an algorithm for high-resolution map** of genomic structural variation. Nat Methods 6:677–681
Choe S (2006) Brassinosteroid biosynthesis and inactivation. Physiol Plant 126:539–548. https://doi.org/10.1111/j.1399-3054.2006.00681.x
Chomicki G, Renner SS (2015) Watermelon origin solved with molecular phylogenetics including L innaean material: another example of museomics. New Phytol 205:526–532. https://doi.org/10.1111/nph.13163
Clevenger J, Chu Y, Chavarro C, Botton S, Culbreath A, Isleib TG, Holbrook CC, Ozias-Akins P (2018) Map** late leaf spot resistance in peanut (Arachis hypogaea) using QTL-seq reveals markers for marker-assisted selection. Front Plant Sci 9:83. https://doi.org/10.3389/fpls.2018.00083
Crienen J, Reuling G, Segers B, van de Wal M (2009) New cucumber plants with a compact growth habit. Patent. International publication number WO 59777:A1
Danisman S (2016) TCP transcription factors at the interface between environmental challenges and the plant’s growth responses. Front Plant Sci 7:1930. https://doi.org/10.3389/fpls.2016.01930
Denna D (1963) Morphology of the bush and vine habits and the allelism of the bush genes in Cucurbita maxima and C. pepo squash. Proc Am Soc Hort Sci 82:370–377
Denna DW (1962) A study of the genetic, morphological and physiological basis of the bush and vine habit of several cucurbits. Cornell University, June
Dong W, Wu D, Li G, Wu D, Wang Z (2018) Next-generation sequencing from bulked segregant analysis identifies a dwarfism gene in watermelon. Sci Rep 8:1–7. https://doi.org/10.1038/s41598-018-21293-1
Dyutin K, Afanas’eva E (1987) Inheritance of the short vine trait in watermelon. Cytol Genet 21:71–73
Elshire RJ, Glaubitz JC, Sun Q, Poland JA, Kawamoto K, Buckler ES, Mitchell SE (2011) A robust, simple genoty**-by-sequencing (GBS) approach for high diversity species. PLoS ONE 6:e19379. https://doi.org/10.1371/journal.pone.0019379
Fukino N, Ohara T, Sugiyama M, Kubo N, Hirai M, Sakata Y, Matsumoto S (2012) Map** of a gene that confers short lateral branching (slb) in melon (Cucumis melo L.). Euphytica 187:133–143. https://doi.org/10.1007/s10681-012-0768-z
Gomes RF, Santos LdS, Braz LT, Andrade FLdN, Monteiro SMF (2019) Number of stems and plant density in mini watermelon grown in a protected environment. Pesqui Agropecu Trop. https://doi.org/10.1590/1983-40632019v4954196
Gordon A, Hannon GJ (2010) FASTX-Toolkit. FASTQ/A short-reads pre-processing tools. http://hannonlab.cshl.edu/fastx_toolkit/
Guo S, Zhang J, Sun H, Salse J, Lucas WJ, Zhang H, Zheng Y, Mao L, Ren Y et al (2013) The draft genome of watermelon (Citrullus lanatus) and resequencing of 20 diverse accessions. Nat Genet 45:51–58. https://doi.org/10.1038/ng.2470
Hedden P (2003) The genes of the Green Revolution. Trends Genet 19:5–9. https://doi.org/10.1016/S0168-9525(02)00009-4
Hexun H, **aoqi Z, Zhencheng W, Qinghuai L, ** L (1998) Inheritance of male-sterility and dwarfism in watermelon [Citrullus lanatus (Thunb.) Matsum. and Nakai]. Sci Hortic 74:175–181. https://doi.org/10.1016/S0304-4238(97)00102-7
Hwang J, Oh J, Kim Z, Staub JE, Chung S-M, Park Y (2014) Fine genetic map** of a locus controlling short internode length in melon (Cucumis melo L.). Mol Breed 34:949–961. https://doi.org/10.1007/s11032-014-0088-1
Jang YJ, Yun HS, Rhee S-J, Seo M, Kim Y, Lee GP (2020) Exploring molecular markers and candidate genes responsible for watermelon dwarfism. Hortic Environ Biotechnol 61:173–182. https://doi.org/10.1007/s13580-020-00229-7
Jia Q, Li C, Shang Y, Zhu J, Hua W, Wang J, Yang J, Zhang G (2015) Molecular characterization and functional analysis of barley semi-dwarf mutant Riso no. 9265. BMC Genomics 16:1–11. https://doi.org/10.1186/s12864-015-2116-x
Kauffman C, Lower R (1976) Inheritance of an extreme dwarf plant type in the cucumber. J Am Soc Hortic Sci
Khush GS (2001) Green revolution: the way forward. Nat Rev Genet 2:815–822. https://doi.org/10.1038/35093585
Kieffer M, Master V, Waites R, Davies B (2011) TCP14 and TCP15 affect internode length and leaf shape in Arabidopsis. Plant J 68:147–158. https://doi.org/10.1111/j.1365-313X.2011.04674.x
Kim T-W, Hwang J-Y, Kim Y-S, Joo S-H, Chang SC, Lee JS, Takatsuto S, Kim S-K (2005) Arabidopsis CYP85A2, a cytochrome P450, mediates the Baeyer-Villiger oxidation of castasterone to brassinolide in brassinosteroid biosynthesis. Plant Cell 17:2397–2412. https://doi.org/10.1105/tpc.105.033738
Knavel DE (1990) Inheritance of a short-internode mutant of mainstream’muskmelon. HortScience 25:1274–1275. https://doi.org/10.21273/HORTSCI.25.10.1274
Kosambi D (1944) The estimation of map distance. Ann Eugenics 12:505–525
Kosugi S, Natsume S, Yoshida K, MacLean D, Cano L, Kamoun S, Terauchi R (2013) Coval: improving alignment quality and variant calling accuracy for next-generation sequencing data. PLoS ONE 8:e75402. https://doi.org/10.1371/annotation/cc88d2b5-36e8-441a-ab5f-58a9ed143d6b
Kubicki B, Sołtysiak U, Korzeniewska A (1986) Induced mutation in cucumber (Cucumis sativus L.). V: compact type of growth. Genetic Pol 27:289–298
Lee J, Son B, Choi Y, Kang J, Lee Y, Je BI, Park Y (2018) Development of an SNP set for marker-assisted breeding based on the genoty**-by-sequencing of elite inbred lines in watermelon. J Plant Biotechnol 45:242–249. https://doi.org/10.5010/JPB.2018.45.3.242
Levi A, Thomas CE, Keinath AP, Wehner TC (2001) Genetic diversity among watermelon (Citrullus lanatus and Citrullus colocynthis) accessions. Genet Resour Crop Evol 48:559–566. https://doi.org/10.1023/A:1013888418442
Levi A, Wechter WP, Thies JA, Ling K-S, Reddy U, Xu Y, Guo S, Zhang X (2011) Watermelon. Genetics, genomics and breeding of cucurbits:309–334
Li H, Durbin R (2009) Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25:1754–1760. https://doi.org/10.1093/bioinformatics/btp324
Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, Marth G, Abecasis G, Durbin R (2009) The sequence alignment/map format and SAMtools. Bioinformatics 25:2078–2079
Li H, Jiang L, Youn JH, Sun W, Cheng Z, ** T, Ma X, Guo X, Wang J et al (2013) A comprehensive genetic study reveals a crucial role of CYP90D2/D2 in regulating plant architecture in rice (Oryza sativa). New Phytol 200:1076–1088. https://doi.org/10.1111/nph.12427
Liu P (1972) Inheritance and morphology of two dwarf mutants in watermelon. J Am Soc Hort Sci 97:745–748
Mohr H (1956) Mode of inheritance of the bushy growth characteristics in watermelon. Proc Assn S Agr Workers 53:174
Multani DS, Briggs SP, Chamberlin MA, Blakeslee JJ, Murphy AS, Johal GS (2003) Loss of an MDR transporter in compact stalks of maize br2 and sorghum dw3 mutants. Science 302:81–84. https://doi.org/10.1126/science.1086072
Nagashima A, Suzuki G, Uehara Y, Saji K, Furukawa T, Koshiba T, Sekimoto M, Fujioka S, Kuroha T et al (2008) Phytochromes and cryptochromes regulate the differential growth of Arabidopsis hypocotyls in both a PGP19-dependent and a PGP19-independent manner. Plant J 53:516–529. https://doi.org/10.1111/j.1365-313X.2007.03358.x
Noh B, Bandyopadhyay A, Peer WA, Spalding EP, Murphy AS (2003) Enhanced gravi-and phototropism in plant mdr mutants mislocalizing the auxin efflux protein PIN1. Nature 423:999–1002. https://doi.org/10.1038/nature01716
Okamoto K, Ueda H, Shimada T, Tamura K, Koumoto Y, Tasaka M, Morita MT, Hara-Nishimura I (2016) An ABC transporter B family protein, ABCB19, is required for cytoplasmic streaming and gravitropism of the inflorescence stems. Plant Signal Behav 11:e1010947. https://doi.org/10.1080/15592324.2015.1010947
Pandey MK, Khan AW, Singh VK, Vishwakarma MK, Shasidhar Y, Kumar V, Garg V, Bhat RS, Chitikineni A et al (2017) QTL-seq approach identified genomic regions and diagnostic markers for rust and late leaf spot resistance in groundnut (Arachis hypogaea L.). Plant Biotechnol J 15:927–941. https://doi.org/10.1111/pbi.12686
Paris HS (2015) Origin and emergence of the sweet dessert watermelon, Citrullus lanatus. Ann Bot 116:133–148. https://doi.org/10.1093/aob/mcv077
Paris HS, Nerson H, Karchi Z (1984) Genelics of internode length in melons. J Hered 75:403–406. https://doi.org/10.1093/oxfordjournals.jhered.a109965
Poland JA, Rife TW (2012) Genoty**-by-sequencing for plant breeding and genetics. Plant Genome 5:92–102. https://doi.org/10.3835/plantgenome2012.05.0005
Robinson R (1965) A new dwarf cucumber. Veg Imp Nwsl 7:23
Shi P, Guy KM, Wu W, Fang B, Yang J, Zhang M, Hu Z (2016) Genome-wide identification and expression analysis of the ClTCP transcription factors in Citrullus lanatus. BMC Plant Biol 16:85. https://doi.org/10.1186/s12870-016-0765-9
Shifriss O (1947) Developmental reversal of dominance in Cucurbita pepo. Proc Am Soc Hort Sci 50:330–346
Singh VK, Khan AW, Jaganathan D, Thudi M, Roorkiwal M, Takagi H, Garg V, Kumar V, Chitikineni A et al (2016) QTL-seq for rapid identification of candidate genes for 100-seed weight and root/total plant dry weight ratio under rainfed conditions in chickpea. Plant Biotechnol J 14:2110–2119. https://doi.org/10.1111/pbi.12567
Spielmeyer W, Ellis MH, Chandler PM (2002) Semidwarf (sd-1), “green revolution” rice, contains a defective gibberellin 20-oxidase gene. Proc Natl Acad Sci USA 99:9043–9048. https://doi.org/10.1073/pnas.132266399
Takagi H, Abe A, Yoshida K, Kosugi S, Natsume S, Mitsuoka C, Uemura A, Utsushi H, Tamiru M et al (2013) QTL-seq: rapid map** of quantitative trait loci in rice by whole genome resequencing of DNA from two bulked populations. Plant J 74:174–183. https://doi.org/10.1111/tpj.12105
Tanabe S, Ashikari M, Fujioka S, Takatsuto S, Yoshida S, Yano M, Yoshimura A, Kitano H, Matsuoka M et al (2005) A novel cytochrome P450 is implicated in brassinosteroid biosynthesis via the characterization of a rice dwarf mutant, dwarf11, with reduced seed length. Plant Cell 17:776–790. https://doi.org/10.1105/tpc.104.024950
Tigchelaar E (1991) New vegetable variety list 23: compiled by the garden seed research committee American seed trade association. HortScience 26:343–357
Van Ooijen J (2006) JoinMap® 4. Software for the calculation of genetic linkage maps in experimental populations, Kyazma BV, Wageningen, p 33
Van Ooijen J (2009) MapQTL® 6, Software for the map** of quantitative trait loci in experimental populations of diploid species. Kyazma BV, Wageningen 59
Verrier PJ, Bird D, Burla B, Dassa E, Forestier C, Geisler M, Klein M, Kolukisaoglu Ü, Lee Y et al (2008) Plant ABC proteins–a unified nomenclature and updated inventory. Trends Plant Sci 13:151–159. https://doi.org/10.1016/j.tplants.2008.02.001
Wang H, Li W, Qin Y, Pan Y, Wang X, Weng Y, Chen P, Li Y (2017) The cytochrome P450 gene CsCYP85A1 is a putative candidate for super compact-1 (scp-1) plant architecture mutation in cucumber (Cucumis sativus L.). Front Plant Sci 8:266. https://doi.org/10.3389/fpls.2017.00266
Werner T, Motyka V, Laucou V, Smets R, Van Onckelen H, Schmülling T (2003) Cytokinin-deficient transgenic Arabidopsis plants show multiple developmental alterations indicating opposite functions of cytokinins in the regulation of shoot and root meristem activity. Plant Cell 15:2532–2550. https://doi.org/10.1105/tpc.014928
Wu S, Wang X, Reddy U, Sun H, Bao K, Gao L, Mao L, Patel T, Ortiz C et al (2019) Genome of ‘Charleston Gray’, the principal American watermelon cultivar, and genetic characterization of 1,365 accessions in the US National Plant Germplasm System watermelon collection. Plant Biotechnol J 17:2246–2258. https://doi.org/10.1111/pbi.13136
Yang H, Li YG (2009) Study on a new short stem gene of watermelon. Chin Garden Digest 32–33
Yaobin Q, Peng C, Yichen C, Yue F, Derun H, Tingxu H, **anjun S, Jiezheng Y (2018) QTL-Seq identified a major QTL for grain length and weight in rice using near isogenic F2 population. Rice Sci 25:121–131. https://doi.org/10.1016/j.rsci.2018.04.001
Ye K, Schulz M, Long Q, Apweiler R, Ning Z (2009) Pindel: a pattern growth approach to detect break points of large deletions and medium sized insertions from paired-end short reads. Bioinformatics 25(21):2865–2871
Zhang G, Ren Y, Sun H, Guo S, Zhang F, Zhang J, Zhang H, Jia Z, Fei Z et al (2015) A high-density genetic map for anchoring genome sequences and identifying QTLs associated with dwarf vine in pumpkin (Cucurbita maxima Duch.). BMC Genomics 16:1–13. https://doi.org/10.1186/s12864-015-2312-8
Zhang X, Wang W, Guo N, Zhang Y, Bu Y, Zhao J, ** to fine map a wild soybean allele characteristic of greater plant height. BMC Genomics 19:1–12. https://doi.org/10.1186/s12864-018-4582-4
Acknowledgements
This work was supported by a Grant (710001-07-5) from the Vegetable Breeding Research Center through the Agriculture, Food and Rural Affairs Research Center Support Program of the Ministry of Agriculture, Food and Rural Affairs (MAFRA), Korea. This study was also supported by a Grant (Project No: PJ014846012020) from National Institute of Horticultural Herbal Sciences, Rural Development Administration.
Author information
Authors and Affiliations
Contributions
YC, SL, JP, and SK carried out plant material preparation and phenotype evaluation. YC, SL, and GP carried out DNA extraction; YC, SL, and GP, and YP performed the NGS data analysis, genetic map**, and QTL-seq analysis; YC, SL, and YP wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
13580_2020_330_MOESM1_ESM.pdf
Figure S1 Genetic linkage maps based on an F2 population derived from a cross between “Bush Sugar Baby” and PCL-J1 using 336 single-nucleotide polymorphisms (SNPs) (PDF 1542 kb)
13580_2020_330_MOESM3_ESM.pdf
Figure S3 Gene sequence of ClCG09G018320 encoding the ABC B family 19 protein and sequence variants between BSB and PCL-J (PDF 107 kb)
13580_2020_330_MOESM4_ESM.jpg
Figure S4 Comparison of plant heights between the semi-dwarf type “Bush Sugar Baby” (BSB), the extreme dwarf type “Caupat-dw,” and F1 plants from a cross between BSB and “Caupat-dw” six weeks (the 17–20-true-leaf stage) after germination (JPG 99 kb)
13580_2020_330_MOESM5_ESM.xlsx
Table S1 Data for comparison of genome annotation and collinearity between the “97103” genome assembly version 1 and the “Charleston Gray” genome assembly version 2 (XLSX 269 kb)
13580_2020_330_MOESM6_ESM.xlsx
Table S2 Data for genes and SNPs/Indels located in the physical distance (1 Mbp) between the sdw-1-flanking SNPs (“Charleston Gray” genome assembly version 2) (XLSX 83 kb)
Rights and permissions
About this article
Cite this article
Cho, Y., Lee, S., Park, J. et al. Identification of a candidate gene controlling semi-dwarfism in watermelon, Citrullus lanatus, using a combination of genetic linkage map** and QTL-seq. Hortic. Environ. Biotechnol. 62, 447–459 (2021). https://doi.org/10.1007/s13580-020-00330-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13580-020-00330-x