Abstract
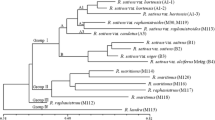
A simple and reliable multiplex polymerase chain reaction (PCR) method was developed using sequence-characterized amplified region (SCAR) markers to identify kiwifruit (Actinidia spp.) cultivars. We observed 183 different polymorphic random amplified polymorphic DNA (RAPD) markers using 23 arbitrary primers in 30 kiwifruit cultivars. The number of polymorphic bands per primer varied from five (OPM-15, OPN-05, and OPN-19) to 12 (OPG-01), with an average of eight. Fifty-eight cultivar-specific polymorphic RAPD fragments were converted into 16 SCAR markers after validating their specificity. Among them, 15 SCAR markers retained the dominant behavior of the original RAPD fragment, whereas the KA11_774 SCAR marker showed a co-dominant polymorphism. Based on RAPD and SCAR data sets, cluster analyses were performed with the unweighted pair-group method of arithmetic averages. The mean genetic similarities were estimated as 0.703 and 0.677 using the RAPD and SCAR markers, respectively. Thirty kiwifruit cultivars were divided into three clusters for RAPD analysis and into two clusters for SCAR analysis, based on a similarity value of 0.67. Overall, the kiwifruit cultivars with a similar breeding background tended to cluster together. Therefore, RAPD and SCAR markers could be successfully applied to assay genetic relationships for kiwifruit cultivar identification. Cluster analysis of combined RAPD and SCAR markers showed very similar results when compared to the RAPD analysis in dendrogram form. The kiwifruit cultivars tested in this study could be identified using only 10 different SCAR markers (KB08_182, KK08_287, KG19_295, KN15_316, KG01_411, KF07_433, KG09_468, KG09_477, KL01_764, and KA11_774). Furthermore, we optimized SCAR markers for simultaneous identification of the 30 kiwifruit cultivars by multiplex PCR, using two sets of five SCAR primer pairs. The developed SCAR markers and multiplex PCR approach facilitate efficient identification of kiwifruit cultivars.





Similar content being viewed by others
References
Agarwal M, Shrivastava N, Padh H (2008) Advances in molecular marker techniques and their applications in plant sciences. Plant Cell Rep 27:617–631
Bautista R, Crespillo R, Cánovas FM, Claros MG (2002) Identification of olive-tree cultivars with SCAR markers. Euphytica 129:33–41
Budak H, Shearman RC, Parmaksiz I, Dweikat I (2004) Comparative analysis of seeded and vegetative biotype buffalograsses based on phylogenetic relationship using ISSRs, SSRs, RAPDs, and SRAPs. Theor Appl Genet 109:280–288
Cho KH, Heo S, Kim HR, Kim JH, Shin IS, Han SE, Kim SE, Kim DH (2010) Discrimination of Korean apple cultivars using combination of RAPD-SCAR markers. Korean J Hortic Sci Technol 28:828–835
Cho KH, Kwack Y-B, Park SJ, Kim SH, Lee HC, Kim MY (2017) Genetic diversity in kiwifruit germplasm evaluated using RAPD and SRAP markers. J Plant Biotechnol 44:303–311
Cipriani G, Di Bella R, Testolin R (1996) Screening RAPD primers for molecular taxonomy and cultivar fingerprinting in the genus Actinidia. Euphytica 90:169–174
Edwards MC, Gibbs RA (1994) Multiplex PCR: advantages, development, and applications. Genome Res 3:S65–S75
El-Sayed NE, Hashad MM, Shaaban MM (2011) Molecular characterization and discrimination among grapevine cultivars by RAPD markers. Aust J Basic Appl Sci 5:230–235
Ferguson AR (2007) The need for characterisation and evaluation of germplasm: kiwifruit as an example. Euphytica 154:371–382
Ferguson AR, Huang H (2007) Genetic resources of kiwifruit: Domestication and breeding. Hortic Rev 33:1–121
Huang H, Li Z, Li J, Kubisiak TL, Layne DR (2002) Phylogenetic relationships in Actinidia as revealed by RAPD analysis. J Am Soc Hortic Sci 127:759–766
Ibáñez J, Vargas AM, Palancar M, Borrego J, de Andrés MT (2009) Genetic relationships among table-grape varieties. Am J Enol Vitic 60:35–42
Korir NK, Han J, Shangguan L, Wang C, Kayesh E, Zhang Y, Fang J (2013) Plant variety and cultivar identification: advances and prospects. Crit Rev Biotechnol 33:111–125
Korkovelos AE, Mavromatis AG, Huang WG, Hagidimitriou M, Giakoundis A, Goulas CK (2008) Effectiveness of SSR molecular markers in evaluating the phylogenetic relationships among eight Actinidia species. Sci Hortic 116:305–310
Lee MY, Doh EJ, Kim ES, Kim YW, Ko BS, Oh S-E (2008) Application of the multiplex PCR method for discrimination of Artemisia iwayomogi from other Artemisia Herbs. Biol Pharm Bull 31:685–690
Li G, Park Y-J (2012) SCAR markers for discriminating species of two genera of medicinal plants, Liriope and Ophiopogon. Genet Mol Res 11:2987–2996
Liu Y, Liu Y, Huang H (2010) Genetic variation and natural hybridization among sympatric Actinidia species and the implications for introgression breeding of kiwifruit. Tree Genet Genom 6:801–813
Liu C-Y, Li D-W, Zhou J-H, Zhang Q, Tian H, Yao X-H (2017) Construction of a SNP-based genetic linkage map for kiwifruit using next-generation restriction-site-associated DNA sequencing (RADseq). Mol Breed 37:139
Mu Q, Sun X, Zhong G, Wang X, Song C, Fang J (2012) Employment of a new strategy for identification of lemon (Citrus limon L.) cultivars using RAPD markers. Afr J Agric Res 7:2075–2082
Muralidharan K, Wakeland EK (1993) Concentration of primer and template qualitatively affects products in random-amplified polymorphic DNA PCR. Biotechniques 14:362–364
Palombi MA, Damiano C (2002) Comparison between RAPD and SSR molecular markers in detecting genetic variation in kiwifruit (Actinidia deliciosa A. Chev). Plant Cell Rep 20:1061–1066
Paran I, Michelmore RW (1993) Development of reliable PCR-based markers linked to downy mildew resistance genes in lettuce. Theor Appl Genet 85:985–993
Petri A, Pfannebecker J, Fröhlich J, König H (2013) Fast identification of wine related lactic acid bacteria by multiplex PCR. Food Microbiol 33:48–54
Polashock JJ, Vorsa N (2002) Deveolpment of SCAR markers for DNA fingerprinting and germplasm analysis of American cranberry. J Am Soc Hortic Sci 127:677–684
Powell W, Morgante M, Andre C, Hanafey M, Vogel J, Tingey S, Rafalski A (1996) The comparison of RFLP, RAPD, AFLP, and SSR (microsatellite) markers for germplasm analysis. Mol Breed 2:225–238
Prado MJ, Gonzalez MV, Romo S, Herrera MT (2007) Adventitious plant regeneration on leaf explants from adult male kiwifruit and AFLP analysis of genetic variation. Plant Cell Tissue Organ Cult 88:1–10
Scaglione D, Fornasiero A, Pinto C, Cattonaro F, Spadotto A, Infante R, Meneses C, Messina R, Lain O, Cipriani G, Testolin R (2015) A RAD-based linkage map of kiwifruit (Actinidia chinensis Pl) as a tool to improve the genome assembly and to scan the genomic region of the gender determinant for the marker-assisted breeding. Tree Genet Genom 11:115
Shirkot P, Sharma DR, Mohapatra T, Shirkot CK (2002) Partial molecular characterization of some kiwifruit cultivars by RAPD marker. Indian J Exp Biol 40:233–236
Sneath PHA, Sokal RR (1973) Numerical taxonomy: the principles and practice of numerical classification. W.H. Freeman and Co., San Francisco, pp 1–15
Testolin R, Messina R, Lain O, Cipriani G (2004) A natural sex mutant in kiwifruit (Actinidia deliciosa). N Z J Crop Hortic Sci 32:179–183
Turkec A, Sayar M, Heinze B (2006) Identification of sweet cherry cultivars (Prunus avium L.) and analysis of their genetic relationships by chloroplast sequence-characterised amplified regions (cpSCAR). Genet Resour Crop Evol 53:1635–1641
Vidal JR, Delavault P, Coarer M, Defontaine A (2000) Design of grapevine (Vitis vinifera L.) cultivar-specific SCAR primers for PCR fingerprinting. Theor Appl Genet 101:1194–1201
Wang Y-C, Zhang L, Man Y-P, Li Z-Z, Qin R (2012) Phenotypic characterization and simple sequence repeat identification of red-fleshed kiwifruit germplasm accessions. HortScience 47:992–999
Williams JGK, Hanafey MK, Rafalski JA, Tingey SV (1993) Genetic analysis using random amplified polymorphic markers. Meth Enzymol 218:704–740
Yan G, Yao J, Ferguson AR, Mcneilage MA, Seal AG, Murray BG (1997) New reports of chromosome numbers in Actinidia (Actinidiaceae). N Z J Bot 35:181–186
Yu M, Chu J, Ma R, Shen Z, Fang J (2013) A novel strategy for the identification of 73 Prunus domestica cultivars using random amplified polymorphic DNA (RAPD) markers. Afr J Agric Res 8:243–250
Zhen Y, Li Z, Huang H, Wang Y (2004) Molecular characterization of kiwifruit (Actinidia) cultivars and selections using SSR markers. J Am Soc Hortic Sci 129:374–382
Acknowledgements
This work was performed with the support of “Cooperative Research Program for Agriculture Science and Technology Development (Project Number PJ01448101)” from the Rural Development Administration in Wanju, Republic of Korea.
Author information
Authors and Affiliations
Contributions
KH designed and conducted the experiments; Y-B provided the materials and information for the kiwifruit cultivars; SJ and SE performed the data analysis; HC, KH, and JH helped revise the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Heakeun Yun, Ph.D.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cho, K.H., Kwack, YB., Park, S.J. et al. Sequence-characterized amplified region markers and multiplex-polymerase chain reaction assays for kiwifruit cultivar identification. Hortic. Environ. Biotechnol. 61, 395–406 (2020). https://doi.org/10.1007/s13580-020-00227-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13580-020-00227-9