Abstract
Purpose
Breast cancer is the most commonly diagnosed cancer in women, and triple-negative breast cancer (TNBC) accounts for approximately 15%-20% of all breast cancers. TNBC is highly invasive and malignant. Due to the lack of relevant receptor markers, the prognosis of TNBC is poor and the five-year survival rate is low. Paclitaxel is the first-line drug for the treatment of TNBC, which can inhibit cell mitosis. However, many patients develop drug resistance during treatment, leading to chemotherapy failure. Therefore, finding new therapeutic combinations to overcome TNBC drug resistance can provide new strategies for improving the survival rate of TNBC patients.
Methods
Cell viability assay, RT-qPCR, Colony formation assay, Western blot, and Xenogeneic transplantation methods were used to investigate roles and mechanisms of IRE1α/XBP1s pathway in the paclitaxel-resistant TNBC cells, and combined paclitaxel and IRE1α inhibitor in the treatment of TNBC was examined in vitro and in vivo.
Results
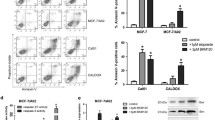
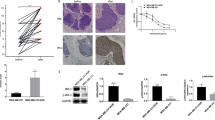
We found activation of UPR in paclitaxel-resistant cells, confirming that IRE1α/XBP1 promotes paclitaxel resistance in TNBC. In addition, we demonstrated that the combination of paclitaxel and IRE1α inhibitors can synergistically inhibit the proliferation of TNBC tumors both in vitro and in vivo,suggesting that IRE1α inhibitors combined with paclitaxel may be a new treatment option for TNBC.
Conclusions
In this study, we demonstrated the important role of IRE1α signaling in mediating paclitaxel resistance and identified that combination therapies targeting IRE1α signaling could overcome paclitaxel resistance and enhance chemotherapy efficacy.






Similar content being viewed by others
Data availability
All data yield or analyzed during this study included in this article.
References
R.L. Siegel, K.D. Miller, N.S. Wagle, A. Jemal, Cancer statistics, 2023. CA: A Cancer J. Clin. 73(1), 17–48 (2023). https://doi.org/10.3322/caac.21763
J.W. Zhu, P. Charkhchi, S. Adekunte, M.R. Akbari, What is known about breast cancer in young women? Cancers 15(6), 1917 (2023)
R.L. Siegel, K.D. Miller, H.E. Fuchs, A. Jemal, Cancer statistics, 2022. CA: A Cancer J. Clin. 72(1) (2022)
C. Luo, N. Li, B. Lu, J. Cai, M. Lu, Y. Zhang, H. Chen, M. Dai, Global and regional trends in incidence and mortality of female breast cancer and associated factors at national level in 2000 to 2019. Chinese Med. J. 135(01), 42–51 (2022)
J. Li, Z. Chen, K. Su, J. Zeng, Clinicopathological classification and traditional prognostic indicators of breast cancer. Int. J. Clin. Exp. Pathol. 8(7), 8500 (2015)
A.C. Garrido-Castro, N.U. Lin, K. Polyak, Insights into Molecular Classifications of Triple-Negative Breast Cancer: improving Patient Selection for Treatment. Cancer Discovery 9(2), 176–198 (2019). https://doi.org/10.1158/2159-8290.Cd-18-1177
S.G. Ahn, S.J. Kim, C. Kim, J. Jeong, Molecular classification of triple-negative breast cancer. J Breast Cancer 19(3), 223 (2016)
H. Muley, R. Fado, R. Rodriguez-Rodriguez, N. Casals, Drug uptake-based chemoresistance in breast cancer treatment. Biochem. Pharmacol. 177, 113959 (2020)
J. Shi, F. Liu, Y. Song, Progress: targeted therapy, immunotherapy, and new chemotherapy strategies in advanced triple-negative breast cancer, Cancer Manage. Res. 9375–9387 (2020)
A. Marra, D. Trapani, G. Viale, C. Criscitiello, G. Curigliano, Practical classification of triple-negative breast cancer: intratumoral heterogeneity, mechanisms of drug resistance, and novel therapies. NPJ. Breast Cancer 6(1), 54 (2020)
D. Zhao, C. Hu, Q. Fu, H. Lv, Combined chemotherapy for triple negative breast cancer treatment by paclitaxel and niclosamide nanocrystals loaded thermosensitive hydrogel. Eur. J. Pharm. Sci. 167, 105992 (2021). https://doi.org/10.1016/j.ejps.2021.105992
A.R.T. Bergin, S. Loi, Triple-negative breast cancer: recent treatment advances. F1000Res 8 (2019). https://doi.org/10.12688/f1000research.18888.1
S. Boichuk, P. Dunaev, I. Mustafin, S. Mani, K. Syuzov, E. Valeeva, F. Bikinieva, A. Galembikova, Infigratinib (BGJ 398), a Pan-FGFR Inhibitor, Targets P-Glycoprotein and Increases Chemotherapeutic-Induced Mortality of Multidrug-Resistant Tumor Cells. Biomedicines 10, 3 (2022). https://doi.org/10.3390/biomedicines10030601
L. Mosca, A. Ilari, F. Fazi, Y.G. Assaraf, G. Colotti, Taxanes in cancer treatment: activity, chemoresistance and its overcoming. Drug. Resist. Updates 54, 100742 (2021). https://doi.org/10.1016/j.drup.2020.100742
T. Avril, E. Vauléon, E. Chevet, Endoplasmic reticulum stress signaling and chemotherapy resistance in solid cancers. Oncogenesis 6(8), e373 (2017). https://doi.org/10.1038/oncsis.2017.72
M. Wang, R.J. Kaufman, Protein misfolding in the endoplasmic reticulum as a conduit to human disease. Nature 529(7586), 326–335 (2016). https://doi.org/10.1038/nature17041
M.C. Kopp, N. Larburu, V. Durairaj, C.J. Adams, M.M.U. Ali, UPR proteins IRE1 and PERK switch BiP from chaperone to ER stress sensor. Nat. Struct. Mol. Biol. 26(11), 1053–1062 (2019). https://doi.org/10.1038/s41594-019-0324-9
C. Hetz, K. Zhang, R.J. Kaufman, Mechanisms, regulation and functions of the unfolded protein response. Nat. Rev. Mol. Cell Biol. 21(8), 421–438 (2020). https://doi.org/10.1038/s41580-020-0250-z
A. Read, M. Schröder, The unfolded protein response: an overview. Biology 10(5) (2021). https://doi.org/10.3390/biology10050384
S.M. Park, T.I. Kang, J.S. So, Roles of XBP1s in transcriptional regulation of target genes. Biomedicines 9(7) (2021). https://doi.org/10.3390/biomedicines9070791
X. Chen, J.R. Cubillos-Ruiz, Endoplasmic reticulum stress signals in the tumour and its microenvironment. Nat. Rev. Cancer 21(2), 71–88 (2021). https://doi.org/10.1038/s41568-020-00312-2
P. Walter, D. Ron, The unfolded protein response: from stress pathway to homeostatic regulation. Science (New York, N.Y.) 334(6059), 1081–1086 (2011). https://doi.org/10.1126/science.1209038
Y. **e, C. Liu, Y. Qin, J. Chen, J. Fang, Knockdown of IRE1ɑ suppresses metastatic potential of colon cancer cells through inhibiting FN1-Src/FAK-GTPases signaling. Int. J. Biochem. Cell Biol. 114, 105572 (2019). https://doi.org/10.1016/j.biocel.2019.105572
D. Xu, Z. Liu, M.-X. Liang, Y.-J. Fei, W. Zhang, Y. Wu, J.-H. Tang, Endoplasmic reticulum stress targeted therapy for breast cancer. Cell Commun. Signal. 20(1), 174 (2022)
L. Sisinni, M. Pietrafesa, S. Lepore, F. Maddalena, V. Condelli, F. Esposito, M. Landriscina, Endoplasmic reticulum stress and unfolded protein response in breast cancer: the balance between apoptosis and autophagy and its role in drug resistance. Int. J. Mol. Sci. 20(4), 857 (2019)
P. Wang, D. Song, D. Wan, L. Li, W. Mei, X. Li, L. Han, X. Zhu, L. Yang, Y. Cai, R. Zhang, Ginsenoside panaxatriol reverses TNBC paclitaxel resistance by inhibiting the IRAK1/NF-κB and ERK pathways. PeerJ 8, e9281 (2020). https://doi.org/10.7717/peerj.9281
K. Xu, W. Zhu, A. Xu, Z. **ong, D. Zou, H. Zhao, D. Jiao, Y. Qing, M.A. Jamal, H.J. Wei, H.Y. Zhao, Inhibition of FOXO1‑mediated autophagy promotes paclitaxel‑induced apoptosis of MDA‑MB‑231 cells. Mol. Med. Rep. 25(2) (2022). https://doi.org/10.3892/mmr.2022.12588
G. Kim, S.K. Jang, Y.J. Kim, H.O. **, S. Bae, J. Hong, I.C. Park, J.H. Lee, Inhibition of Glutamine Uptake Resensitizes Paclitaxel Resistance in SKOV3-TR Ovarian Cancer Cell via mTORC1/S6K Signaling Pathway. Int. J. Mol. Sci. 23, 15 (2022). https://doi.org/10.3390/ijms23158761
J.A. Zundell, T. Fukumoto, J. Lin, N. Fatkhudinov, T. Nacarelli, A.V. Kossenkov, Q. Liu, J. Cassel, C.A. Hu, S. Wu, R. Zhang, Targeting the IRE1α/XBP1 endoplasmic reticulum stress response pathway in ARID1A-mutant ovarian cancers. Cancer Res. 81(20), 5325–5335 (2021). https://doi.org/10.1158/0008-5472.Can-21-1545
Y. Xu, P. Huangyang, Y. Wang, L. Xue, E. Devericks, H.G. Nguyen, X. Yu, J.A. Oses-Prieto, A.L. Burlingame, S. Miglani, et al., ERα is an RNA-binding protein sustaining tumor cell survival and drug resistance. Cell 184(20), 5215–5229.e5217 (2021). https://doi.org/10.1016/j.cell.2021.08.036
M.J.P. Crowley, B. Bhinder, G.J. Markowitz, M. Martin, A. Verma, T.A. Sandoval, C.S. Chae, S. Yomtoubian, Y. Hu, S. Chopra, et al., Tumor-intrinsic IRE1α signaling controls protective immunity in lung cancer. Nat. Commun. 14(1), 120 (2023). https://doi.org/10.1038/s41467-022-35584-9
X. Chen, D. Iliopoulos, Q. Zhang, Q. Tang, M.B. Greenblatt, M. Hatziapostolou, E. Lim, W.L. Tam, M. Ni, Y. Chen, et al., XBP1 promotes triple-negative breast cancer by controlling the HIF1α pathway. Nature 508(7494), 103–107 (2014). https://doi.org/10.1038/nature13119
Acknowledgements
This work was supported by the grants from National Natural Science Foundation of China (82073310, 82373146), ECNU Construction Fund of Innovation and Entrepreneurship Laboratory (44400-20201-532300/021). We thank the Instruments Sharing Platform of School of LifeSciences, East China Normal University.
Author information
Authors and Affiliations
Contributions
M.W., L.Z., and L.P. designed the experiments. M.W., L.Z., L.P., L.L., S.W. and H.P. performed the experiments. M.W., L.Z., L.P., L.L., S.W., H.P. and Z.Y. performed the data analysis. M.W., M.L. and Z.Y. wrote the manuscript.
Corresponding author
Ethics declarations
Animal ethical
All animals mentioned in this article were purchased from the Animal Center of East China Normal University. Animal care and maintenance complied with the guidelines of the Animal Investigation Committee of the Institute of Biomedical Sciences, East China Normal University.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, M., Zhang, L., Pi, L. et al. IRE1α inhibitor enhances paclitaxel sensitivity of triple-negative breast cancer cells. Cell Oncol. (2024). https://doi.org/10.1007/s13402-024-00961-7
Accepted:
Published:
DOI: https://doi.org/10.1007/s13402-024-00961-7