Abstract
The excessive price of nutrients needed for microalgal cultivation is a vital constraint, although mass production is essential for attaining economically viable production of algal fuels and value-added products. Increasing energy demand has prompted the world to look for alternative oil resources. There is a crucial demand for sustainable and economically viable source of energy due to the reduction in conventional fossil fuel supplies. In this study, potato peel and sweet lime peels were exploited in place of conventional growth mediums for the cultivation of microalgal cells. These wastes are zero-value material, which contains vital nutrients and elements for the cultivation of oleaginous microorganisms like microalgae. The growth of the isolated strains Chlorella sorokiniana KMBM_K and KMBM_I in the pretreated peel hydrolysates was investigated. The biomass and lipid extracted can be further investigated for potential applications in biofuel, feed, and food additives. A new insight can be obtained with this study as it integrates the concept of lipid extracted microalgal biomass utilization (LEMB) approach along with waste disposal, thereby serving in the management of these agricultural wastes. While performing the optimization studies, pH 7 and 25% and 40% dilution, respectively, for potato peel and sweet lime were found to support more biomass and lipid yield. The highest biomass yield of 2.1 g L−1 and highest lipid productivity of 49.93 mg L−1 day−1 are obtained in Chlorella sorokiniana KMBM_K when cultivated in potato peel extract. This study showed that these agricultural wastes can be used as a natural, cost effective, ecofriendly, efficient, and affordable substrate for the cultivation of the microalgal isolates.
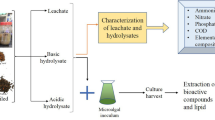
Graphical abstract






Similar content being viewed by others
Data availability
Data used for the research work will be shared upon request.
References
Yang J, Li X, Hu H, Zhang X, Yu Y, Chen Y (2011) Growth and lipid accumulation properties of a freshwater microalga, Chlorella ellipsoidea YJ1, in domestic secondary effluents. Appl Energy 88:3295–3299
Chisti Y (2007) Biodiesel from microalgae. Biotechnol Adv 25(3):294–306
Hashem AH, Suleiman WB, Abu-Elrish GM, El-Sheikh HH (2021) Consolidated bioprocessing of sugarcane bagasse to microbial oil by newly isolated oleaginous fungus: Mortierella wolfii. Arab J Sci Eng 46:199–211
Limbu B, Sibi G (2017) Fruit wastes hydrolysates as feedstock: pre-treatment strategies for cost-saving and sustainable microalgae cultivation. Int J Res Environ Sci 3(2):35–40
Slade R, Bauen A (2013) Micro-algae cultivation for biofuels: cost, energy balance, environmental impacts and future prospect. Biomass Bioenergy 53:29–38
Bhatnagar A, Bhatnagar M, Chinnasamy S, Das KC (2010) Chlorella minutissima – a promising fuel alga for cultivation in municipal wastewaters. Appl Biochem Biotechnol 161:523–536
Wong MH (1985) Cultivation of microalgae in refuse compost and soy-bean waste extracts. Agric Wastes 12(3):225–233
Hashem AH, Suleiman WB, Abu-elreesh G, Shehabeldine AM, Khalil AMA (2020) Sustainable lipid production from oleaginous fungus Syncephalastrum racemosum using synthetic and watermelon peel waste media. Bioresour Technol Rep 12:100569
Hasanin MS, Hashem AH (2020) Eco-friendly, economic fungal universal medium from watermelon peel waste. J Microbiol Methods 168:105802
Hashem AH, Hasanin MS, Khalil AMA, Suleiman WB (2020) Eco-green conversion of watermelon peels to single cell oils using a unique oleaginous fungus: Lichtheimia corymbifera AH13. Waste Biomass Valoriz 11:5721–5732
Park WK, Moon M, Kwak MS, Jeon S, Choi GG, Yang JW, Lee B (2014) Use of orange peel extract for mixotrophic cultivation of Chlorella vulgaris: increased production of biomass and FAMEs. Bioresour Technol 171:343–349
Amit GUK (2019) Utilization of kinnow peel extract with different wastewaters for cultivation of microalgae for potential biodiesel production. J Environ Chem Eng 7:103135
Mathias DJ, Kumar S, Rangarajan V (2019) An investigation on citrus peel as the lignocellulosic feedstock for optimal reducing sugar synthesis with an additional scope for the production of hydrolytic enzymes from the aqueous extract waste. Biocatal Agric Biotechnol 20:101259
Park WK, Moon M, Shin SE, Cho JM, Suh WI, Chang YK, Lee B (2018) Economical DHA (Docosahexaenoic acid) production from Aurantiochytrium sp. KRS101 using orange peel extract and low cost nitrogen sources. Algal Res 29:71–79
Barros HRM, Ferreira TAPC, Genovese MI (2012) Antioxidant capacity and mineral content of pulp and peel from commercial cultivars of citrus from Brazil. Food Chem 134:1892–1898
Arapoglou D, Varzakas Th, Vlyssides A, Israilides C (2010) Ethanol production from potato peel waste (PPW). Waste Manage 30:1898–1902
Jekayinfa SO, Linke B, Pecenka R (2015) Biogas production from selected crop residues in Nigeria and estimation of its electricity value. Int J Renewable Energy Technol 6(2):101–118
Malakar B, Das D, Mohanty K (2020) Optimization of glucose yield from potato and sweet lime peel waste through different pre-treatment techniques along with enzyme assisted hydrolysis towards liquid biofuel. Renewable Energy 145:2723–2732
Sadiq IM, Dalai S, Chandrasekaran N, Mukherjee A (2011) Ecotoxicity study of titania (TiO2) NPs on two microalgae species: Scenedesmus sp. and Chlorella sp. Ecotoxicol Environ Saf 74:1180–1187
Rosgaard L, Pedersen S, Anne S (2007) Comparison of different pretreatment strategies for enzymatic hydrolysis of wheat and barley straw. Appl Biochem Biotechnol 143:284–296
Sininart C, Abdullateef D (2012) Alkali-pretreatment and acid-hydrolysis of banana peels. The 10th International PSU Engineering Conference
Lee YK, Ding SK, Hoe CH, Low CS (1996) Mixotrophic growth of Chlorella sorokiniana in outdoor enclosed photobioreactor. J Appl Phycol 8(2):163–169
Shi XM, Liu HJ, Zhang XW, Chen F (1999) Production of biomass and lutein by Chlorella protothecoides at various glucose concentrations in heterotrophic cultures. Process Biochem 34(4):341–347
Choi IS, Kim JH, Wi SG, Kim KH, Bae HJ (2013) Bioethanol production from mandarin (Citrus unshiu) peel waste using pop** pretreatment. Appl Energy 102:204–210
Yang Y, Xu J, Vail D, Weathers P (2011) Ettlia oleoabundans growth and oil production on agricultural anaerobic waste effluents. Bioresour Technol 102:5076–5082
Abreu AP, Fernandes B, Vicente AA, Teixeira J, Dragone G (2012) Mixotrophic cultivation of Chlorella vulgaris using industrial dairy waste as organic carbon source. Bioresour Technol 118:61–66
Yan D, Lu Y, Chen YF, Wu Q (2011) Waste molasses alone displaces glucose-based medium for microalgal fermentation towards cost-saving biodiesel production. Bioresour Technol 102:6487–6493
Pleissner D, Lam WC, Sun Z, Lin CSK (2013) Food waste as nutrient source in heterotrophic microalgae cultivation. Bioresour Technol 137:139–146
Wu S, Xu XR, Sun KF, Li HB (2014) Effects of fruit waste hydrolysates on biomass and chlorophyll a fluorescence parameters of Chlorella pyrenoidosa. Int J Environ Bioenery 9(2):105–121
Liang Y, Sarkany N, Cui Y, Yesuf J, Trushenski J, Blackburn JW (2010) Use of sweet sorghum juice for lipid production by Schizochytrium limacinum SR21. Bioresour Technol 101:3623–3627
Gupta A, Abraham RE, Barrow CJ, Puri M (2015) Omega-3 fatty acid production from enzyme saccharified hemp hydrolysate using a novel marine thraustochytrid strain. Bioresour Technol 184:373–378
Ji M-K, Yun H-S, Park S, Lee H, Park Y-T, Bae S, Ham J, Choi J (2015) Effect of food wastewater on biomass production by a green microalga Scenedesmus obliquus for bioenergy generation. Bioresour Technol 179:624–628
Guldhe A, Kumari S, Ramanna L, Ramsundar P, Singh P, Rawat I, Bux F (2017) Prospects, recent advancements and challenges of different wastewater streams for microalgal cultivation. J Environ Manage 203:299–315
Hemaiswarya S, Raja R, Carvalho IS, Ravikumar R, Zambare V, Barh D (2012) An Indian scenario on renewable and sustainable energy sources with emphasis on algae. Appl Microbiol Biotechnol 96:1125–1135
Arora N, Patel A, Pruthi PA, Pruthi V (2016) Recycled de-oiled algal biomass extract as a feedstock for boosting biodiesel production from Chlorella minutissima. Appl Biochem Biotechnol 180:1534–1541
Abomohra AEF, Eladel H, El-Esawi M, Wang S, Wang Q, He Z, Feng Y, Shang H, Hanelt D (2018) Effect of lipid-free microalgal biomass and waste glycerol on growth and lipid production of Scenedesmus obliquus: innovative waste recycling for extraordinary lipid production. Bioresour Technol 249:992–999
Kim SS, Ly HV, Kim J, Lee EY, Woo HC (2015) Pyrolysis of microalgae residual biomass derived from Dunaliella tertiolecta after lipid extraction and carbohydrate saccharification. Chem Eng J 263:194–199
Bui HH, Tran KQ, Chen WH (2015) Pyrolysis of microalgae residues — a Kinetic study. Bioresour Technol 199:362–366
Acknowledgements
The authors would like to acknowledge Central Instruments Facility (CIF), IIT Guwahati for the FESEM analysis and Analytical Laboratory, School of Energy Science and Engineering, IIT Guwahati, for various analysis.
Author information
Authors and Affiliations
Contributions
Barasa Malakar: conceptualization; formal analysis; investigation; methodology; roles/writing — original draft.
Debasish Das: supervision; writing — review and editing.
Kaustubha Mohanty: conceptualization; resources; supervision; funding acquisition; writing — review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Potato and sweet lime peel hydrolysate were used for microalgal cultivation.
• Microalgal strains were isolated from biodiversity-rich waterbodies of IIT Guwahati, Assam.
• The isolated strains were identified as Chlorella sorokiniana KMBM_K and KMBM_I.
• The peel hydrolysates support the cultivation of the isolated microalgae.
Rights and permissions
About this article
Cite this article
Malakar, B., Das, D. & Mohanty, K. Utilization of waste peel extract for cultivation of microalgal isolates: a study of lipid productivity and growth kinetics. Biomass Conv. Bioref. 13, 17017–17026 (2023). https://doi.org/10.1007/s13399-022-02313-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-022-02313-7