Abstract
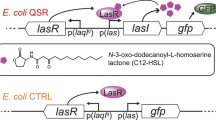
Quorum sensing (QS) is defined as a cellto- cell signaling process that collectively regulates the gene expression of bacteria via small signaling molecules called autoinducers (AIs). It was reported that QS-regulated gene expression in Pseudomonas aeruginosa failed to occur at a high Reynolds number (Re= 3,000), since AI-2, a secreted interspecies signaling molecule, was washed away and so could not reach the minimum concentration required for QS. In this study, we describe the effects of flow speed on QS-stimulated biofilm formation in Escherichia coli O157:H7 inside a very thin microchannel (3 cm×1 cm×45 μm). In microtiter plates, the wild-type strain produced high amounts of exopolysaccharide, whereas its QS mutants ΔluxS and ΔlsrK, defective in AI-2 production and phosphorylation, made less exopolysaccharide. Confocal laser scanning microscopy showed that at a flow rate of 1 μL/min the wild-type strain formed rounded biofilms, whereas such biofilms formed by the QS mutants were fewer and thinner. At a flow rate of 10 μL/min, none of the tested strains formed mature biofilms. Our results suggest that QS is essential in the biofilm maturation under static or very slow laminar flow conditions where the biofilm signal can be easily accumulated and transported to the sessile cells.
Similar content being viewed by others
References
Costerton, J.W., Stewart, P.S. & Greenberg, E. Bacterial biofilms: a common cause of persistent infections. Science 284, 1318–1322 (1999).
Oh, Y., Jo, W., Yang, Y. & Park, S. Influence of culture conditions on Escherichia coli O157: H7 biofilm formation by atomic force microscopy. Ultramicroscopy 107, 869–874 (2007).
Stewart, P.S. & Costerton, J.W. Antibiotic resistance of bacteria in biofilms. Lancet 358, 135–138 (2001).
Li, J. et al. Quorum sensing in Escherichia coli is signaled by AI-2/LsrR: effects on small RNA and biofilm architecture. J. Bacteriol. 189, 6011–6020 (2007).
Anand, S. & Griffiths, M.W. Quorum sensing and expression of virulence in Escherichia coli O157:H7. Int. J. Food Microbiol. 85, 1–9 (2003).
Barrios, A.F.G. et al. Autoinducer 2 controls biofilm formation in Escherichia coli through a novel motility quorum-sensing regulator (MqsR, B3022). J. Bacteriol. 188, 305–316 (2006).
Wu, S.-Y., Hulme, J. & An, S.S.A. Recent trends in the detection of pathogenic Escherichia coli O157:H7. BioChip J. 9, 173–181 (2015).
Kirisits, M.J. et al. Influence of the hydrodynamic environment on quorum sensing in Pseudomonas aeruginosa biofilms. J. Bacteriol. 189, 8357–8360 (2007).
Shumi, W. et al. Shear stress tolerance of Streptococcus mutans aggregates determined by microfluidic funnel device (µFFD). J. Microbiol. Methods 93, 85–89 (2013).
Park, S. et al. Motion to form a quorum. Science 301, 188-188 (2003).
Park, S. et al. Influence of topology on bacterial social interaction. Proc. Natl. Scad. Sci. USA. 100, 13910–13915 (2003).
Jackson, D.W., Simecka, J.W. & Romeo, T. Catabolite repression of Escherichia coli biofilm formation. J. Bacteriol. 184, 3406–3410 (2002).
Kim, E.H. et al. Effect of cyclic stretching on cell shape and division. BioChip J. 9, 306–312 (2015).
Surette, M.G., Miller, M.B. & Bassler, B.L. Quorum sensing in Escherichia coli, Salmonella typhimurium, and Vibrio harveyi: a new family of genes responsible for autoinducer production. Proc. Natl. Acad. Sci. USA 96, 1639–1644 (1999).
Danese, P.N., Pratt, L.A. & Kolter, R. Exopolysaccharide production is required for development of Escherichia coli K-12 biofilm architecture. J. Bacteriol. 182, 3593–3596 (2000).
Dewanti, R. & Wong, A.C. Influence of culture conditions on biofilm formation by Escherichia coli O157: H7. Int. J. Food Microbiol. 26, 147–164 (1995).
Xavier, K.B. & Bassler, B.L. Regulation of uptake and processing of the quorum-sensing autoinducer AI-2 in Escherichia coli. J. Bacteriol. 187, 238–248 (2005).
Andersson, H. & Van Den Berg, A. Microfabrication and microfluidics for tissue engineering: state of the art and future opportunities. Lab. Chip. 4, 98–103 (2004).
Cormack, B.P., Valdivia, R.H. & Falkow, S. FACS-optimized mutants of the green fluorescent protein (GFP). Gene 173, 33–38 (1996).
Spoering, A.L. & Lewis, K. Biofilms and planktonic cells of Pseudomonas aeruginosa have similar resistance to killing by antimicrobials. J. Bacteriol. 183, 6746–6751 (2001).
Miller, V.L. & Mekalanos, J.J. A novel suicide vector and its use in construction of insertion mutations: osmoregulation of outer membrane proteins and virulence determinants in Vibrio cholerae requires toxR. J. Bacteriol. 170, 2575–2583 (1988).
Yanisch-Perron, C., Vieira, J. & Messing, J. Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mpl8 and pUC19 vectors. Gene 33, 103–119 (1985).
Donnenberg, M.S. & Kaper, J.B. Construction of an eae deletion mutant of enteropathogenic Escherichia coli by using a positive-selection suicide vector. Infect. Immun. 59, 4310–4317 (1991).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lim, J., Lee, KM., Park, C.Y. et al. Quorum sensing is crucial to Escherichia coli O157:H7 biofilm formation under static or very slow laminar flow conditions. BioChip J 10, 241–249 (2016). https://doi.org/10.1007/s13206-016-0310-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13206-016-0310-9