Abstract
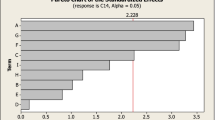
Optimization for enhanced phytase production by Aspergillus oryzae SBS50 in submerged fermentation was investigated using Taguchi design. In first step design, starch, beef extract, magnesium sulphate, ferrous sulphate and Tween 80 were identified as significant factors affecting phytase production. These significant factors were further optimized at four different levels using a second Taguchi design and were observed that 1% starch, 2% beef extact, 3% Tween 80, 0.1% magnesium sulphate and 0.225% ferrous sulphate supported maximum phytase production (47,432 U/L). The use of Taguchi designed experiments resulted in 14.9-fold enhancement in phytase production compared to the medium optimized by ‘one variable at a time’ approach. Furthermore, 4% agar immobilized conidiospores of A. oryzae supported high phytase production compared with free cells and other matrices. Agar-immobilized conidiospores resulted in sustained phytase production up to eight repeated batch cycles followed by a decrease in enzyme titres.



Similar content being viewed by others
References
Assemi H, Rezapanah M, Vafaei-Shoushtari R, Mehrvar A (2012) Modified artificial diet for rearing of tobacco budworm, Helicoverpa armigera, using the Taguchi method and Derringer’s desirability function. J Insect Sci 12:100
Awad GEA, Esawy AM, El-Gammal EW, Ahmed HM, Elnashar MM, Atwa NA (2015) Comparative studies of free and immobilized phytase, produced by Penicillium purpurogenu GE1, using grafted alginate/carrageenan beads. Egypt Pharm J 14:87–93
Bajaj K, Wani MA (2011) Enhanced phytase production from Nocardia sp. MB 36 using agro-residues as substrates: potential application for animal feed production. Eng Life Sci 11(6):620–628
Bajaj K, Wani MA (2015) Purification and characterization of a novel phytase from Nocardia sp. MB 36. Biocatal Biotransform 33:141–149
Bhavsar K, Kumar VR, Khire JM (2012) Downstream processing of extracellular phytase from Aspergillus niger: chromatography process vs. aqueous two phase extraction for its simultaneous partitioning and purification. Process Biochem 47(7):1066–1072
Bucke C (1987) Cell immobilization in calcium alginate. In: Mosbach K (ed) Methods in ezymology, vol 135B. Academic press, New York, pp 175–189
Chadha BS, Gulati H, Minhas M, Saini HS, Singh N (2004) Phytase production by the thermophilic fungus Rhizomucor pusillus. World J Microbiol Biotechnol 20:105–109
Coban HB, Demisa A (2014) Screening of phytase producers and optimization of culture conditions for submerged fermentations. Bioprocess Biosyst Eng 37(4):609–616
Das S, Ghosh U (2014) Effect of nutritional supplementation of solid state fermentation medium on biosynthesis of phytase fom Aspergillus niger NCIM 612. J Sci Indust Res 73:593–597
Fiske CH, Subbarow YP (1925) The colorimetric determination of phosphorus. J Biol Chem 66:375–400
Hemachander C, Boss N, Puvanakrishnan R (2001) Whole cell immobilization of Ralstonia pickettii for lipase production. Process Biochem 36(7):629–633
Heravi KM, Eftekhar F, Yakhchali B, Tabandeh F (2008) Isolation and identification of a lipase producing Bacillus sp. from soil. Pak J Biol Sci 11(5):740–745
Jain J, Sapna, Singh B (2016) Characteristics and biotechnological applications of bacterial phytases. Process Biochem 51:159–169
Jalasutram V, Jetty A (2011) Optimization of 1, 3-Propanediol production by Klebsiella pneumoniae 141B using Taguchi methodology: improvement in production by cofermentation studies. Res Biotechnol 2(2):90–104
Kaur P, Satyanarayana T (2010) Improvement in cell-bound phytase activity of Pichia anomala by permeabilization and applicability of permeabilized cells in soymilk dephytinization. J Appl Microbiol 108(6):2041–2049
Kuek C (1991) Production of glucoamylase using Aspergillus phoenicus immobilized in calcium alginate beads. Appl Microbiol Biotechnol 35:466–470
Kumar P, Satyanarayana T (2007) Optimization of culture variables for improving glucoamylase production by alginate-entrapped Thermomucor indicae-seudaticae using statistical methods. Bioresour Technol 98:1252–1259
Kumar V, Yadav AN, Verma P, Sangwan P, Saxena A, Kumar K, Singh B (2017) β-Propeller phytases: diversity, catalytic attributes, biotechnological improvements and applications. Int J Biol Macromol 98:595–609
Kumari A, Singh B, Satyanarayana T (2016) Mixed substrate fermentation for enhanced phytase production by thermophilic mould Sporotrichum thermophile and its application in beneficiation of poultry feed. Appl Biochem Biotechnol 178:197–210
Maller A, Vici CA, Facchini FDA, da Silva TM, Kamimura ES, Rodrigues MI, Jorge JA, Terenzi HF, Polizeli MLTM (2014) Increase of the phytase production by Aspergillus japonicus and its biocatalyst on chicken feed treatment. J Basic Microbiol 54:S152–S160
Park JK, Chang HN (2000) Microencapsulation of microbial cells. Biotechnol Adv 18(4):303–319
Quan CS, Fan SD, Ohta Y (2003) Immobilization of Candida krusei cells producing phytase in alginate gel beads: an application of the preparation of myo-inositol phosphates. Appl Microbiol Biotechnol 62:41–47
Sedghi M, Golian A, Esmaeilipour O, Van Krimpen MM (2014) Application of the Taguchi method in poultry science: estimation of the in vitro optimum intrinsic phytase activity of rye, wheat and barley. British Poul Sci 55(2):246–252
Sapna, Singh B (2013) Improved production of protease-resisitant phytase by Aspergillus oryzae and its applicability in the hydrolysis of insoluble phytates. J Indust Microbiol Biotechnol 40(8):891–899
Sapna, Singh B (2014) Phytase production by Aspergillus oryzae in solid state fermentation and its applicability in dephytinization of wheat bran. Appl Biochem Biotechnol 173(7):1885–1895
Sapna, Singh B (2015) Biocatalytic potential of protease-resistant phytase of Aspergillus oryzae SBS50 in ameliorating food nutrition. Biocatal Biotransfom 33(3):167–174
Singh B, Satyanarayana T (2006) A marked enhancement in phytase production by a thermophilic mould Sporotrichum thermophile using statistical designs in a cost-effective cane molasses medium. J Appl Microbiol 101:344–352
Singh B, Satyanarayana T (2008) Phytase production by Sporotrichum thermophile in a cost-effective cane molasses medium in submerged fermentation and its application in bread. J Appl Microbiol 105:1858–1865
Singh B, Satyanarayana T (2015) Fungal phytases: characteristics and amelioration of nutritional quality and growth of non ruminants. J Anim Physiol Nutr 99:646–660
Singh B, Kunze G, Satyanarayana T (2011) Developments in biochemical aspects and biotechnological applications of microbial phytases. Biotechnol Mol Biol Rev 6(3):69–87
Vats P, Banerjee UC (2004) Production studies and catalytic properties of phytases (myo-inositolhexakisphosphate phosphohydrolases): an overview. Enzyme Microbial Technol 35:3–14
Vohra A, Satyanarayana T (2002) Statistical optimization of the medium components by response surface methodology to enhance phytase production by Pichia anomala. Process Biochem 37:999–1004
Acknowledgements
The authors acknowledge the financial assistance from University Grants Commission [F. No. 39-203/2010 (SR)], New Delhi during the course of this investigation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Sapna, Singh, B. Free and immobilized Aspergillus oryzae SBS50 producing protease-resistant and thermostable phytase. 3 Biotech 7, 213 (2017). https://doi.org/10.1007/s13205-017-0804-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-017-0804-8