Abstract
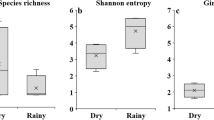
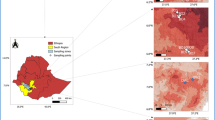
Arid zones represent 44% of the world's agricultural land where crops are exposed to stressful conditions, mainly water deficiency and high solar radiation. Under such conditions, arbuscular mycorrhizal fungi (AMF) may improve plant stress resistance by increasing water and nutrient uptake but their diversity and community in dryland agroecosystems remains underexplored. The objective of this study was to investigate the AMF species diversity and community associated with five economically relevant crops (alfalfa, pepper, maize, onion and walnut) grown in an arid zone of northern Mexico. DNA was extracted and PCR-amplified with AMF specific primers from root and rhizosphere soil samples. Libraries were constructed and sequenced using the Illumina MiSeq system; representative sequences were assigned to 30 species in 12 genera. AMF community composition was similar among the crops but significantly different between the roots and the rhizosphere soil. A strong dominance, in terms of DNA sequence read abundance of Septoglomus sp. was found in root samples. On the contrary, in rhizosphere soil Rhizophagus spp. and Funneliformis mosseae were more abundant. Most of the Septoglomus sequences were affiliated to a node in-between the branches of the species S. viscosum and S. titan, indicating an undescribed or uncharacterized highly abundant species in this ecosystem. Describing and understanding plant associated AMF communities in drylands may provide valuable information for future tailored made inocula efforts.





Similar content being viewed by others
Data availability
The sequencing run has been stored at the Sequence Read Archive at the NCBI with accession number PRJNA772040.
Code availability
Not applicable
References
Abdedaiem R, Rejili M, Mahdhi M, de Lajudie P, Mars M (2020) Soil properties shape species diversity and community composition of native arbuscular mycorrhizal fungi in Retama raetam roots growing on arid ecosystems of Tunisia. Int J Agric Biol 23(2):438–446. https://doi.org/10.17957/IJAB/15.1307
Al-Arjani AB, Hashem A, Abd-Allah EF (2020) Arbuscular mycorrhizal fungi modulates dynamics tolerance expression to mitigate drought stress in Ephedra foliata Boiss. Saudi J Biol Sci 27(1):380–394. https://doi.org/10.1016/j.sjbs.2019.10.008
Alaux PL, Mison C, Senés-Guerrero C, Moreau V, Manssens G, Foucart G et al (2021) Diversity and species composition of arbuscular mycorrhizal fungi across maize fields in the southern part of Belgium. Mycorrhiza 31(2):265–272. https://doi.org/10.1007/s00572-020-01007-0
Alguacil MDM, Schlaeppi K, López-García Á, van der Heijden MG, Querejeta JI (2021) Contrasting responses of arbuscular mycorrhizal fungal families to simulated climate warming and drying in a semiarid shrubland. Microb Ecol: 1-4. https://doi.org/10.1007/s00248-021-01886-6
Alrajhei K, Saleh I, Abu-Dieyeh MH (2022) Biodiversity of arbuscular mycorrhizal fungi in plant roots and rhizosphere soil from different arid land environment of Qatar. Plant Direct 6(1):e369. https://doi.org/10.1002/pld3.369
Antunes PM, Koch AM, Morton JB, Rillig MC, Klironomos JN (2011) Evidence for functional divergence in arbuscular mycorrhizal fungi from contrasting climatic origins. New Phytol 189(2):507–514. https://doi.org/10.1111/j.1469-8137.2010.03480.x
Armada E, Probanza A, Roldán A, Azcón R (2016) Native plant growth promoting bacteria Bacillus thuringiensis and mixed or individual mycorrhizal species improved drought tolerance and oxidative metabolism in Lavandula dentata plants. J Plant Physiol 192:1–12. https://doi.org/10.1016/j.jplph.2015.11.007
Augé RM, Toler HD, Saxton AM (2015) Arbuscular mycorrhizal symbiosis alters stomatal conductance of host plants more under drought than under amply watered conditions: a meta-analysis. Mycorrhiza 25(1):13–24. https://doi.org/10.1007/s00572-014-0585-4
Badi OBM, Abdelhalim TS, Eltayeb MM, Gorafi YSA, Tsujimoto H, Taniguchi T (2019) Dominance of limited arbuscular mycorrhizal fungal generalists of Sorghum bicolor in a semi-arid region in Sudan. Soil Sci Plant Nutr 65(6):570–578. https://doi.org/10.1080/00380768.2019.1680573
Baharuddin FA, Zainal Z, Sukiran NL (2019) Morphological changes analysis of rice cv. IR64 under drought stress. In AIP Conference Proceedings. AIP Publishing LLC, vol. 2111, no. 1, p. 040003. https://doi.org/10.1063/1.5111242
Baon JB, Smith SE, Alston AM (1994) Phosphorus uptake and growth of barley as affected by soil temperature and mycorrhizal infection. J Plant Nutr 17:479–492. https://doi.org/10.1080/01904169409364742
Begum N, Ahanger MA, Su Y, Lei Y, Mustafa NSA, Ahmad P, Zhang L (2019) Improved drought tolerance by AMF inoculation in maize (Zea mays) involves physiological and biochemical implications. Plants 8(12):579. https://doi.org/10.3390/plants8120579
Berger SA, Stamatakis A (2011) Aligning short reads to reference alignments and trees. Bioinformatics 27:2068–2075. https://doi.org/10.1093/bioinformatics/btr320
Berger SA, Krompass D, Stamatakis A (2011) Performance, accuracy, and web server for evolutionary placement of short sequence reads under maximum-likelihood. Syst Biol 60:291–302. https://doi.org/10.1093/sysbio/syr010
Bolyen E, Rideout JR, Dillon MR, Bokulich NA, Abnet CC et al (2019) Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat Biotechnol 37:852–857. https://doi.org/10.1038/s41587-019-0209-9
Callahan BJ, McMurdie PJ, Rosen MJ, Han AW, Johnson AJ, Holmes SP (2016) DADA2: High-resolution sample inference from Illumina amplicon data. Nat Methods 13(7):581–583. https://doi.org/10.1038/nmeth.3869
Chimal-Sánchez E, Senés-Guerrero C, Varela L, Montaño NM, García-Sánchez R, Pacheco A et al (2020) Septoglomus mexicanum, a new species of arbuscular mycorrhizal fungi from semiarid regions in Mexico. Mycologia 112(1):121–132. https://doi.org/10.1080/00275514.2019.1671147
Compant S, Van Der Heijden MG, Sessitsch A (2010) Climate change effects on beneficial plant–microorganism interactions. FEMS Microbiol Ecol 73(2):197–214. https://doi.org/10.1111/j.1574-6941.2010.00900.x
Davies FT, Olalde-Portugal V, Aguilera-Gomez L, Alvarado MJ, Ferrera-Cerrato RC, Boutton TW (2002) Alleviation of drought stress of Chile ancho pepper (Capsicum annuum L. cv. San Luis) with arbuscular mycorrhiza indigenous to Mexico. Sci Hortic 92(3–4):347–359. https://doi.org/10.1016/S0304-4238(01)00293-X
de Mendiburu F (2021) Agricolae: Statistical Procedures for Agricultural Research. https://CRAN.R-project.org/package=agricolae
Faggioli VS, Cabello MN, Grilli G, Vasar M, Covacevich F, Öpik M (2019) Root colonizing and soil borne communities of arbuscular mycorrhizal fungi differ among soybean fields with contrasting historical land use. Agric Ecosyst Environ 269:174–182. https://doi.org/10.1016/j.agee.2018.10.002
Fernandes RA, Ferreira DA, Saggin-Junior OJ, Stürmer SL, Paulino HB, Siqueira JO, Carneiro MAC (2016) Occurrence and species richness of mycorrhizal fungi in soil under different land use. Can J Soil Sci 96(3):271–280. https://doi.org/10.1139/cjss-2015-0011
Fitter AH, Heinemeyer A, Staddon PL (2000) The impact of elevated CO2 and global climate change on arbuscular mycorrhizas: a mycocentric approach. New Phytol 147(1):179–187. https://doi.org/10.1046/j.1469-8137.2000.00680.x
Gao C, Montoya L, Xu L, Madera M, Hollingsworth J, Purdom E, Hutmacher RB, Dahlberg JA, Coleman-Derr D, Lemaux PG, Taylor JW (2019) Strong succession in arbuscular mycorrhizal fungal communities. ISME J 13:214–226. https://doi.org/10.1038/s41396-018-0264-0
Gargouri M, Karray F, Chebaane A, Mhiri N, Partida-Martínez LP, Sayadi S, Mliki A (2021) Increasing aridity shapes beta diversity and the network dynamics of the belowground fungal microbiome associated with Opuntia ficus-indica. Sci Total Environ 773:145008. https://doi.org/10.1016/j.scitotenv.2021.145008
Gaur MK, Squires VR (2018) Geographic Extent and Characteristics of the World’s Arid Zones and Their Peoples. In: Climate Variability Impacts on Land Use and Livelihoods in Drylands. Springer, Cham, pp 3–20. https://doi.org/10.1007/978-3-319-56681-8_1
Golla B (2021) Agricultural production system in arid and semi-arid regions. J Agric Sc Food Technol 7(2):234–244. https://doi.org/10.17352/2455-815X.000113
Hontoria C, García-González I, Quemada M, Roldán A, Alguacil MM (2019) The cover crop determines the AMF community composition in soil and in roots of maize after a ten-year continuous crop rotation. Sci Total Environ 660:913–922. https://doi.org/10.1016/j.scitotenv.2019.01.095
Jerbi M, Labidi S, Lounès-Hadj Sahraoui A, Chaar H, BenJeddi F (2020) Higher temperatures and lower annual rainfall do not restrict, directly or indirectly, the mycorrhizal colonization of barley (Hordeum vulgare L.) under rainfed conditions. PLoS One 15(11):e0241794. https://doi.org/10.1371/journal.pone.0241794
Jochum M, McWilliams KM, Borrego E, Kolomiets M, Niu G, Pierson E, Jo YK (2019) Bioprospecting Plant growth-promoting rhizobacteria that mitigate drought stress in grasses. Front Microbiol 10:2106. https://doi.org/10.3389/fmicb.2019.02106
Kavadia A, Omirou M, Fasoula D, Trajanoski S, Andreou E, Ioannides IM (2020) Genotype and soil water availability shape the composition of AMF communities at chickpea early growth stages. Appl Soil Ecol 150:103443. https://doi.org/10.1016/j.apsoil.2019.103443
Khalediyan N, Weisany W, Schenk PM (2021) Arbuscular mycorrhizae and rhizobacteria improve growth, nutritional status and essential oil production in Ocimum basilicum and Satureja hortensis. Ind Crops Prod 160:113163. https://doi.org/10.1016/j.indcrop.2020.113163
Klichowska E, Nobis M, Piszczek P, Błaszkowski J, Zubek S (2019) Soil properties rather than topography, climatic conditions, and vegetation type shape AMF–feathergrass relationship in semi-natural European grasslands. Appl Soil Ecol 144:22–30. https://doi.org/10.1016/j.apsoil.2019.07.001
Krüger M, Stockinger H, Krüger C, Schüßler A (2009) DNA-based species level detection of Glomeromycota: one PCR primer set for all arbuscular mycorrhizal fungi. New Phytol 183:212–223. https://doi.org/10.1111/j.1469-8137.2009.02835.x
Krüger M, Krüger C, Walker C, Stockinger H, Schüßler A (2012) Phylogenetic reference data for systematics and phylotaxonomy of arbuscular mycorrhizal fungi from phylum to species level. New Phytol 193(4):970–984. https://doi.org/10.1111/j.1469-8137.2011.03962.x
Loján P, Senés-Guerrero C, Suárez JP, Kromann P, Schüßler A, Declerck S (2017) Potato field-inoculation in Ecuador with Rhizophagus irregularis: no impact on growth performance and associated arbuscular mycorrhizal fungal communities. Symbiosis 73:45–56. https://doi.org/10.1007/s13199-016-0471-2
Lumini E, Vallino M, Alguacil MM, Romani M, Bianciotto V (2011) Different farming and water regimes in Italian rice fields affect arbuscular mycorrhizal fungal soil communities. Ecol Appl 21(5):1696–1707. https://doi.org/10.1890/10-1542.1
Luo Y, Wang Z, He Y, Li G, Lv X, Zhuang L (2020) High-throughput sequencing analysis of the rhizosphere arbuscular mycorrhizal fungi (AMF) community composition associated with Ferula sinkiangensis. BMC Microbiol 20(1):1–14. https://doi.org/10.1186/s12866-020-02024-x
Mahanty T, Bhattacharjee S, Goswami M, Bhattacharyya P, Das B, Ghosh A, Tribedi P (2017) Biofertilizers: a potential approach for sustainable agriculture development. Environ Sci Pollut Res 24(4):3315–3335. https://doi.org/10.1007/s11356-016-8104-0
Mangiafico S (2022) Rcompanion: Functions to Support Extension Education Program Evaluation. https://CRAN.R-project.org/package=rcompanion
Manoharan L, Rosenstock NP, Williams A, Hedlund K (2017) Agricultural management practices influence AMF diversity and community composition with cascading effects on plant productivity. Appl Soil Ecol 115:53–59. https://doi.org/10.1016/j.apsoil.2017.03.012
Meteored (2021) Histórico del clima en Chihuahua. https://www.meteored.mx/chihuahua/historico. Accessed 20 August 2021
Miller MA, Pfeiffer W, Schwartz T (2010) "Creating the CIPRES Science Gateway for inference of large phylogenetic trees" in Proceedings of the Gateway Computing Environments Workshop (GCE), 14 Nov. 2010, New Orleans, LA pp 1 - 8
Monz CA, Kunt HW, Reeves FB, Elliot ET (1994) The response of mycorrhizal colonization to elevated CO2 and climate change in Pascopyrum smithii and Bouteloua gracilis. Plant Soil 165:75–80
National Park Service (2018) Chihuahuan Desert Ecoregion. https://www.nps.gov/im/chdn/ecoregion.htm. Accessed 20 August 2021
Ogle DH, Doll JC, Wheeler P, Dinno A (2022) FSA: Fisheries Stock Analysis. https://github.com/fishR-Core-Team/FSA
Ojuederie OB, Olanrewaju OS, Babalola OO (2019) Plant growth promoting rhizobacterial mitigation of drought stress in crop plants: implications for sustainable agriculture. Agronomy 9(11):712. https://doi.org/10.3390/agronomy9110712
Oksanen J, Blanchet FG, Fiendly M, Kindt R, Legendre P, McGlinn D, Minchin P, O’Hara R, Simpson G, Solymos P, Henry H, Stevenes M, Szoecs E, Wagner H, (2020) vegan: Community Ecology Package. R Package Version 2.5.7. https://CRAN.R-project.org/package=vegan
Ouledal S, Lumini E, Bianciotto V, Khemira H, Ennajeh M (2022) Diversity of Arbuscular Mycorrhizal Fungi in olive orchard soils in arid regions of Southern Tunisia. Arid Land Res Manag: 1-17. https://doi.org/10.1080/15324982.2022.2037787
Pedersen TL (2020) patchwork: The Composer of Plots. https://CRAN.R-project.org/package=patchwork
Peterson GA (2018) Dryland Farming. Reference Module in Earth Systems and Environmental Sciences, Elsevier. https://doi.org/10.1016/B978-0-12-409548-9.11401-0
Ren C, Chen J, Lu X, Doughty R, Zhao F, Zhong Z et al (2018) Responses of soil total microbial biomass and community compositions to rainfall reductions. Soil Biol Biochem 116:4–10. https://doi.org/10.1016/j.soilbio.2017.09.028
SEMARNAT (2020) Precipitación media histórica por entidad federativa. http://dgeiawf.semarnat.gob.mx:8080/ibi_apps/WFServlet?IBIF_ex=D3_AGUA01_01&IBIC_user=dgeia_mce&IBIC_pass=dgeia_mce&NOMBREENTIDAD=*&NOMBREANIO=*. Accessed 20 August 2021
Sendek A, Karakoç C, Wagg C, Domínguez-Begines J, do Couto GM, van der Heijden MG, et al (2019) Drought modulates interactions between arbuscular mycorrhizal fungal diversity and barley genotype diversity. Sci Rep 9(1):1–15. https://doi.org/10.1038/s41598-019-45702-1
Senés-Guerrero C, Schüßler A (2016) A conserved arbuscular mycorrhizal fungal core-species community colonizes potato roots in the Andes. Fungal Divers 77(1):317–333. https://doi.org/10.1007/s13225-015-0328-7
Senés-Guerrero C, Torres-Cortés G, Pfeiffer S, Rojas M, Schüßler A (2014) Potato-associated arbuscular mycorrhizal fungal communities in the Peruvian Andes. Mycorrhiza 24(6):405–417. https://doi.org/10.1007/s00572-013-0549-0
Senés-Guerrero C, Giménez S, Pacheco A, Gradilla-Hernández MS, Schüßler A (2020) New MiSeq based strategy exposed plant-preferential arbuscular mycorrhizal fungal communities in arid soils of Mexico. Symbiosis 81(3):235–246. https://doi.org/10.1007/s13199-020-00698-5
Séry D, Kouadjo ZG, Voko BR, Zézé A, (2016) Selecting native arbuscular mycorrhizal fungi to promote Cassava growth and increase yield under field conditions. Front Microbiol 7:2063. https://doi.org/10.3389/fmicb.2016.02063
Šmilauer P, Košnar J, Kotilínek M, Šmilauerová M (2020) Contrasting effects of host identity, plant community, and local species pool on the composition and colonization levels of arbuscular mycorrhizal fungal community in a temperate grassland. New Phytol 225(1):461–473. https://doi.org/10.1111/nph.16112
Stewart BA (2016) Dryland farming. Reference Module in Food Sciences. https://doi.org/10.1016/B978-0-08-100596-5.02937-1
Teixeira-Rios T, da Silva DKA, Goto BT, Yano-Melo AM (2018) Seasonal differences in arbuscular mycorrhizal fungal communities in two woody species dominating semiarid caatinga forests. Folia Geobot 53(2):191–200. https://doi.org/10.1007/s12224-018-9314-7
United Nations (2020) United Nations Decade for deserts and the fight against desertification. https://www.un.org/en/events/desertification_decade/whynow.shtml. Accessed 20 August 2021
Vieira LC, da Silva DKA, de Melo MAC, Escobar IC, Oehl F, da Silva GA (2019) Edaphic factors influence the distribution of arbuscular mycorrhizal fungi along an altitudinal gradient of a tropical mountain. Microbial Ecol 78(4):904–913. https://doi.org/10.1007/s00248-019-01354-2
Vieira LC, Silva AD, Escobar EC, Silva MD, Moura AD, Oehl F, Silva AD (2020) Changes in an arbuscular mycorrhizal fungi community along an environmental gradient. Plants 9(1):52. https://doi.org/10.3390/plants9010052
Wickham H (2010) ggplot2: Elegant Graphics for Data Analysis
Wilson JM, Witcombe JR (1985) Crops for arid lands. In Plants for Arid Lands. Springer, Dordrecht, pp. 35–52. https://doi.org/10.1007/978-94-011-6830-4_4
Xu X, Chen C, Zhang Z, Sun Z, Chen Y, Jiang J, Shen Z (2017) The influence of environmental factors on communities of arbuscular mycorrhizal fungi associated with Chenopodium ambrosioides revealed by MiSeq sequencing investigation. Sci Rep 7(1):1–11. https://doi.org/10.1038/srep45134
Zhang J, Wang F, Che R, Wang P, Liu H, Ji B, Cui X (2016a) Precipitation shapes communities of arbuscular mycorrhizal fungi in Tibetan alpine steppe. Sci Rep 6(1):1–10. https://doi.org/10.1038/srep23488
Zhang T, Yang X, Guo R, Guo J (2016b) Response of AM fungi spore population to elevated temperature and nitrogen addition and their influence on the plant community composition and productivity. Sci Rep 6(1):1–12. https://doi.org/10.1038/srep24749
Zhao L, Zhang K, Sun X, He X (2022) Dynamics of arbuscular mycorrhizal fungi and glomalin in the rhizosphere of Gymnocarpos przewalskii in Northwest Desert, China. Appl Soil Ecol 170:104251. https://doi.org/10.1016/j.apsoil.2021.104251
Funding
This study was funded as a Mexican-German bilateral scientific and technological cooperation project, by CONACYT-National Council of Science and Technology of Mexico (grant no. 267782) and BMBF Germany (grant no. 01DN17031). CEGM would like to thank CONACYT (CVU No. 875797) and Tecnologico de Monterrey for academic scholarships.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
Not applicable
Consent to participate
Not applicable
Consent for publication
Not applicable
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
13199_2022_851_MOESM1_ESM.jpg
Supplementary file1 Supplementary Fig. 1. Root and soil sampling in crop fields. a-b) Walnut sampling (30 cm depth), south-central Chihuahua. c-d) Alfalfa sampling (30 cm deep), south-center region. e–f) Maize sampling (12 cm depth) south-east region. g) Pepper sampling (10 cm depth).(JPG 981 KB)
13199_2022_851_MOESM2_ESM.pdf
Supplementary file2 Supplementary Fig. 2. Phylogenetic backbone tree used for EPA analysis. Every tree branch contains an i-number that can be correlated to affiliated Reference Sequences (RS).(PDF 85 KB)
13199_2022_851_MOESM3_ESM.pdf
Supplementary file3 Supplementary Fig. 3. Rarefaction curve of AMF sequences annotated at the species level.(PDF 139 KB)
13199_2022_851_MOESM4_ESM.xlsx
Supplementary file4 Supplementary Table 1. Read relative abundance of AMF species per plant species and habitat. Letter R at the end of the crop represents root samples and letter S represents rhizosphere soil samples.(XLSX 15 KB)
13199_2022_851_MOESM5_ESM.xlsx
Supplementary file5 Supplementary Table 2. Annotated Reference Sequences (RS) per sample and their read abundances.(XLSX 239 KB)
Rights and permissions
About this article
Cite this article
Guardiola-Márquez, C.E., Pacheco, A., Mora-Godínez, S. et al. Septoglomus species dominate the arbuscular mycorrhiza of five crop plants in an arid region of northern Mexico. Symbiosis 87, 93–106 (2022). https://doi.org/10.1007/s13199-022-00851-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13199-022-00851-2