Abstract
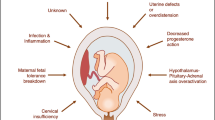
Preterm birth (PTB) is the main condition related to perinatal morbimortality worldwide. The aim of this study was to identify associations of spontaneous PTB with genetic variants, exposures, and interactions between and within them. We carried out a retrospective case–control study including parental sociodemographic and obstetric data, and fetal genetic variants. We sequenced the coding and flanking regions of five candidate genes from the placental blood cord of 69 preterm newborns and 61 at term newborns. We identify the characteristics with the greatest predictive power of PTB using penalized regressions, in which we include exposures (E), genetic variants (G), and two-way interactions. Few prenatal visits (< 5) was the main predictor of PTB from 26 G, 35 E, 299 G × G, 564 E × E, and 875 G × E evaluated terms. Within the fetal genetic characteristics, we observed associations of rs4845397 (KCNN3, allele T) variant; G × G interaction between rs12621551 (COL4A3, allele T) and rs73993878 (COL4A3, allele A), which showed sensitivity to anemia; and G × G interaction between rs11680670 (COL4A3, allele T) and rs2074351 (PON1, allele A), which showed sensitivity to vaginal discharge. The results of this exploratory study suggest that social disparities and metabolic pathways linked to uterine relaxation, inflammation/infections, and collagen metabolism would be involved in PTB etiology. Future studies with a larger sample size are necessary to confirm these findings and to analyze a greater number of exposures.
Similar content being viewed by others
References
Ayers KL, Cordell HJ (2010) SNP selection in genome-wide and candidate gene studies via penalized logistic regression. Genet Epidemiol 34(8):879–891. https://doi.org/10.1002/gepi.20543
Babitt JL, Lin HY (2012) Mechanisms of anemia in CKD. J Am Soc Nephrol 23(10):1631–1634. https://doi.org/10.1681/ASN.2011111078
Barrera-Gómez J, Agier L, Portengen L et al (2017) A systematic comparison of statistical methods to detect interactions in exposome-health associations. Environ Health 16(1):74. https://doi.org/10.1186/s12940-017-0277-6
Bekkar B, Pacheco S, Basu R, DeNicola N (2020) Association of air pollution and heat exposure with preterm birth, low birth weight, and stillbirth in the US: a systematic review. JAMA Netw Open 3(6):e208243. https://doi.org/10.1001/jamanetworkopen.2020.8243
Bien J, Taylor J, Tibshirani R (2013) A LASSO for hierarchical interactions. Ann Stat 41(3):1111–1141. https://doi.org/10.1214/13-AOS1096
Black RE, Victora CG, Walker SP et al (2013) Maternal and child undernutrition and overweight in low-income and middle-income countries. Lancet 382(9890):427–451. https://doi.org/10.1016/S0140-6736(13)60937-X
Brainard AM, Korovkina VP, England SK (2007) Potassium channels and uterine function. Semin Cell Dev Biol 18(3):332–339. https://doi.org/10.1016/j.semcdb.2007.05.008
Browning BL, Zhou Y, Browning SR (2018) A one-penny imputed genome from next-generation reference panels. Am J Hum Genet 103(3):338–348. https://doi.org/10.1016/j.ajhg.2018.07.015
Camps J, Iftimie S, García-Heredia A, Castro A, Joven J (2017) Paraoxonases and infectious diseases. Clin Biochem 50(13–14):804–811. https://doi.org/10.1016/j.clinbiochem.2017.04.016
Canty AJ (2002) Resampling methods in R: the boot package. Newsl R Proj Volume 2(3). http://cran.fhcrc.org/doc/Rnews/Rnews_2002-3.pdf. Accessed 19 April 2021
Castilla EE, Orioli IM (2004) ECLAMC: the Latin-American collaborative study of congenital malformations. Community Genet 7(2–3):76–94. https://doi.org/10.1159/000080776
Chanrachakul B, Matharoo-Ball B, Turner A et al (2003) Immunolocalization and protein expression of the alpha subunit of the large-conductance calcium-activated potassium channel in human myometrium. Reproduction 126(1):43–48. https://doi.org/10.1530/rep.0.1260043
Chawanpaiboon S, Vogel JP, Moller AB et al (2019) Global, regional, and national estimates of levels of preterm birth in 2014: a systematic review and modelling analysis. Lancet Glob Health 7(1):e37–e46. https://doi.org/10.1016/S2214-109X(18)30451-0
Chen D, Hu Y, Chen C et al (2004) Polymorphisms of the paraoxonase gene and risk of preterm delivery. Epidemiology 15(4):466–470. https://doi.org/10.1097/01.ede.0000129509.59912.b2
Danecek P, Bonfield JK, Liddle J et al (2021) Twelve years of SAMtools and BCFtools. Gigascience 10(2):giab008. https://doi.org/10.1093/gigascience/giab008
Day LJ, Schaa KL, Ryckman KK et al (2011) Single-nucleotide polymorphisms in the KCNN3 gene associate with preterm birth. Reprod Sci 18(3):286–295. https://doi.org/10.1177/1933719110391277
Dirección de Estadísticas e Información de Salud - Ministerio de Salud de Argentina (2021) Estadísticasvitales Información Básica 2019. https://www.argentina.gob.ar/sites/default/files/serie5numero63.pdf. Accessed 19 April 2021
Dotters-Katz SK, Grotegut CA, Heine RP (2013) The effects of anemia on pregnancy outcome in patients with pyelonephritis. Infect Dis Obstet Gynecol 2013:780960. https://doi.org/10.1155/2013/780960
Efron B, Hastie T (2021) Computer age statistical inference, student edition: algorithms, evidence, and data science, vol 6. Cambridge University Press, Cambridge
Elias D, Gimenez L, Poletta F et al (2021) Preterm birth and genitourinary tract infections: assessing gene-environment interaction. Pediatr Res 90(3):678–683. https://doi.org/10.1038/s41390-020-01200-z
Elias D, Campaña H, Poletta FA et al (2022) Preterm birth etiological pathways: a Bayesian networks and mediation analysis approach. Pediatr Res 91(7):1882–1889. https://doi.org/10.1038/s41390-021-01659-4
Emmert-Streib F, Dehmer M (2019) High-dimensional LASSO-based computational regression models: regularization shrinkage and selection. Mach Learn Knowl Extr 1(1):359–383. https://doi.org/10.3390/make1010021
Firth D (1993) Bias reduction of maximum likelihood estimates. Biometrika 80:27–38. https://doi.org/10.2307/2336755
Gimenez LG, Krupitzki HB, Momany AM et al (2016) Maternal and neonatal epidemiological features in clinical subtypes of preterm birth. J Matern Fetal Neonatal Med 29(19):3153–3161. https://doi.org/10.3109/14767058.2015.1118035
Gimenez LG, Momany AM, Poletta FA et al (2017) Association of candidate gene polymorphisms with clinical subtypes of preterm birth in a Latin American population. Pediatr Res 82(3):554–559. https://doi.org/10.1038/pr.2017.109
Girling J, de Swiet M (1998) Inherited thrombophilia and pregnancy. Curr Opin Obstet Gynecol 10(2):135–144. https://doi.org/10.1097/00001703-199804000-00010
Gómez LM, Sammel MD, Appleby DH et al (2010) Evidence of a gene-environment interaction that predisposes to spontaneous preterm birth: a role for asymptomatic bacterial vaginosis and DNA variants in genes that control the inflammatory response. Am J Obstet Gynecol 202(4):386.e1-386.e3866. https://doi.org/10.1016/j.ajog.2010.01.042
Gonçalves LF, Chaiworapongsa T, Romero R (2002) Intrauterine infection and prematurity. Ment Retard Dev Disabil Res Rev 8(1):3–13. https://doi.org/10.1002/mrdd.10008
Heinze G, Ploner M, Dunkler D, Southworth H (2020) Package “logistf”. https://cran.r-project.org/web/packages/logistf/logistf.pdf. Accessed 17 February 2021
Jo BS, Choi SS (2015) Introns: the functional benefits of introns in genomes. Genomics Inform 13(4):112–118. https://doi.org/10.5808/GI.2015.13.4.112
Jones C, Chan C, Farine D (2011) Sex in pregnancy. CMAJ 183(7):815–818. https://doi.org/10.1503/cmaj.091580
Keller MC (2014) Gene × environment interaction studies have not properly controlled for potential confounders: the problem and the (simple) solution. Biol Psychiatry 75(1):18–24. https://doi.org/10.1016/j.biopsych.2013.09.006
Ketterlinus RD, Henderson SH, Lamb ME (1990) Maternal age, sociodemographics, prenatal health and behavior: influences on neonatal risk status. J Adolesc Health Care 11(5):423–431. https://doi.org/10.1016/0197-0070(90)90090-o
Kruegel J, Rubel D, Gross O (2013) Alport syndrome—insights from basic and clinical research. Nat Rev Nephrol 9(3):170–178. https://doi.org/10.1038/nrneph.2012.259
Krupitzki HB, Gadow EC, Gili JA et al (2013) Environmental risk factors and perinatal outcomes in preterm newborns, according to family recurrence of prematurity. Am J Perinatol 30(6):451–461. https://doi.org/10.1055/s-0032-1326990
Kuhn M (2008) Building predictive models in R using the caret package. J Stat Softw 28(5):1–26. https://doi.org/10.18637/jss.v028.i05
Laurin C, Boomsma D, Lubke G (2016) The use of vector bootstrap** to improve variable selection precision in Lasso models. Stat Appl Genet Mol Biol 15(4):305–320. https://doi.org/10.1515/sagmb-2015-0043
Leifert JA (2008) Anaemia and cigarette smoking. Int J Lab Hematol 30(3):177–184. https://doi.org/10.1111/j.1751-553X.2008.01067.x
Lim M, Hastie T (2015) Learning interactions via hierarchical group-lasso regularization. J Comput Graph Stat 24(3):627–654. https://doi.org/10.1080/10618600.2014.938812
Lindenbaum P (2015) JVarkit: java-based utilities for Bioinformatics. https://doi.org/10.6084/m9.figshare.1425030.v1
Mackness MI, Mackness B, Durrington PN, Connelly PW, Hegele RA (1996) Paraoxonase: biochemistry, genetics and relationship to plasma lipoproteins. Curr Opin Lipidol 7(2):69–76. https://doi.org/10.1097/00041433-199604000-00004
Macones GA, Parry S, Elkousy M, Clothier B, Ural SH, Strauss JF 3rd (2004) A polymorphism in the promoter region of TNF and bacterial vaginosis: preliminary evidence of gene-environment interaction in the etiology of spontaneous preterm birth. Am J Obstet Gynecol 190(6):1504–1513. https://doi.org/10.1016/j.ajog.2004.01.001
Matharoo-Ball B, Ashford ML, Arulkumaran S, Khan RN (2003) Down-regulation of the alpha- and beta-subunits of the calcium-activated potassium channel in human myometrium with parturition. Biol Reprod 68(6):2135–2141. https://doi.org/10.1095/biolreprod.102.010454
Maymon E, Romero R, Pacora P et al (2000) Matrilysin (matrix metalloproteinase 7) in parturition, premature rupture of membranes, and intrauterine infection. Am J Obstet Gynecol 182(6):1545–1553. https://doi.org/10.1067/mob.2000.107652
McLaren W, Gil L, Hunt SE et al (2016) The ensembl variant effect predictor. Genome Biol 17(1):122. https://doi.org/10.1186/s13059-016-0974-4
Mitchell H (2004) Vaginal discharge–causes, diagnosis, and treatment. BMJ 328(7451):1306–1308. https://doi.org/10.1136/bmj.328.7451.1306
Moore CM, Jacobson SA, Fingerlin TE (2019) Power and sample size calculations for genetic association studies in the presence of genetic model misspecification. Hum Hered 84(6):256–271. https://doi.org/10.1159/000508558
Morales JO, Dipierri JE, Alfaro E, Bejarano IF (2000) Distribution of the ABO system in the Argentine Northwest: miscegenation and genetic diversity. Interciencia 25(9):432–435
Muglia LJ, Katz M (2010) The enigma of spontaneous preterm birth. N Engl J Med 362(6):529–535. https://doi.org/10.1056/NEJMra0904308
Phaloprakarn C, Tangjitgamol S (2013) Maternal ABO blood group and adverse pregnancy outcomes. J Perinatol 33(2):107–111. https://doi.org/10.1038/jp.2012.73
Price AL, Patterson NJ, Plenge RM, Weinblatt ME, Shadick NA, Reich D (2006) Principal components analysis corrects for stratification in genome-wide association studies. Nat Genet 38(8):904–909. https://doi.org/10.1038/ng1847
Purcell S, Neale B, Todd-Brown K et al (2007) PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 81(3):559–575. https://doi.org/10.1086/519795
Rahmati S, Azami M, Badfar G, Parizad N, Sayehmiri K (2020) The relationship between maternal anemia during pregnancy with preterm birth: a systematic review and meta-analysis. J Matern Fetal Neonatal Med 33(15):2679–2689. https://doi.org/10.1080/14767058.2018.1555811
Ratowiecki J, Santos MR, Poletta F et al (2021) Inequidades sociales en madres adolescentes y la relación con resultados perinatales adversos en poblaciones sudamericanas. Cad Saude Publica 36(12):e00247719. https://doi.org/10.1590/0102-311X00247719
Romero R, Friel LA, Velez Edwards DR et al (2010a) A genetic association study of maternal and fetal candidate genes that predispose to preterm prelabor rupture of membranes (PROM). Am J Obstet Gynecol 203(4):361.e1-361.e30. https://doi.org/10.1016/j.ajog.2010.05.026
Romero R, Velez Edwards DR, Kusanovic JP et al (2010b) Identification of fetal and maternal single nucleotide polymorphisms in candidate genes that predispose to spontaneous preterm labor with intact membranes. Am J Obstet Gynecol 202(5):431.e1-431.e4334. https://doi.org/10.1016/j.ajog.2010.03.026
Soydinç HE, Sak ME, Evliyaoğlu O et al (2012) Maternal plasma prolidase, matrix metalloproteinases 1 and 13, and oxidative stress levels in pregnancies complicated by preterm premature rupture of the membranes and chorioamnionitis. J Turk Ger Gynecol Assoc 13(3):172–177. https://doi.org/10.5152/jtgga.2012.23
Soydinc HE, Sak ME, Evliyaoglu O et al (2013) Prolidase, matrix metalloproteinases 1 and 13 activity, oxidative-antioxidative status as a marker of preterm premature rupture of membranes and chorioamnionitis in maternal vaginal washing fluids. Int J Med Sci 10(10):1344–1351. https://doi.org/10.7150/ijms.4802
Stephenson CD, Lockwood CJ, Ma Y, Guller S (2005) Thrombin-dependent regulation of matrix metalloproteinase (MMP)-9 levels in human fetal membranes. J Matern Fetal Neonatal Med 18(1):17–22. https://doi.org/10.1080/14767050500123632
Stevenson M, Stevenson MM, BiasedUrn I. Package ‘epiR’. https://cran.r-project.org/web/packages/epiR/epiR.pdf. Last accessed 19 September 2021
Strauss JF 3rd (2013) Extracellular matrix dynamics and fetal membrane rupture. Reprod Sci 20(2):140–153. https://doi.org/10.1177/1933719111424454
Tsai HJ, Liu X, Mestan K et al (2008) Maternal cigarette smoking, metabolic gene polymorphisms, and preterm delivery: new insights on GxE interactions and pathogenic pathways. Hum Genet 123(4):359–369. https://doi.org/10.1007/s00439-008-0485-9
UNICEF, WHO, World Bank Group, United Nations (2019) Levels and trends in child mortality: report 2019. United Nations Children’s Fund, New York
Vanderweele TJ, Ko YA, Mukherjee B (2013) Environmental confounding in gene-environment interaction studies. Am J Epidemiol 178(1):144–152. https://doi.org/10.1093/aje/kws439
Vilhjálmsson BJ, Nordborg M (2013) The nature of confounding in genome-wide association studies. Nat Rev Genet 14(1):1–2. https://doi.org/10.1038/nrg3382
Vittinghoff E, McCulloch CE (2007) Relaxing the rule of ten events per variable in logistic and Cox regression. Am J Epidemiol 165(6):710–718. https://doi.org/10.1093/aje/kwk052
Wang X, Zuckerman B, Pearson C et al (2002) Maternal cigarette smoking, metabolic gene polymorphism, and infant birth weight. JAMA 287(2):195–202. https://doi.org/10.1001/jama.287.2.195
Zhang Y, Pan W (2015) Principal component regression and linear mixed model in association analysis of structured samples: competitors or complements? Genet Epidemiol 39(3):149–155. https://doi.org/10.1002/gepi.21879
Acknowledgements
The authors want to thank Mrs. Mariana Piola and Alejandra Mariona.
Funding
This work was supported by Agencia Nacional de Promoción Científica y Tecnológica (ANPCyT-MINCyT), grants: PICT-2018–4275 to JSLC and PICT-2018–4285 to LGG; Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET); Instituto Nacional de Genética Médica Populacional (INAGEMP) [Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) grant 465549/2014–4; Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), grant 88887.136366/2017–00; and Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS), grant 17/2551–0000521-0]; and from Fundo de Incentivo à Pesquisa e Eventos do Hospital de Clínicas de Porto Alegre (FIPE/HCPA), grant 17–0445. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
DEE, LGG, and JSLC contributed in the conception of the study; CS, VC, JSLC, and LGG contributed in the acquisition of data; VC, HBK, MR, HC, RU, LGG, DEE, and SLH contributed in the review and curation of data; LGG, DRM, ABON, and ACBF contributed in DNA sequencing; DEE, LGG, JSLC, FAP, JAG, MRS, and JR contributed in the analysis of data. All authors contributed in the drafting of the manuscript, critical revision for important intellectual content, and final approval of the published version.
Corresponding author
Ethics declarations
Ethics approval
Study protocols were approved by the Centro de Educación Médica e Investigaciones Clínicas (CEMIC) Ethics Committee (IRB 00001745–IORG 0001315) and the University of Iowa Institutional Review Board (IRB 200411759).
Consent to participate
Parents provided written informed consent for themselves and the neonates.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Elias, D.E., Santos, M.R., Campaña, H. et al. Genes, exposures, and interactions on preterm birth risk: an exploratory study in an Argentine population. J Community Genet 13, 557–565 (2022). https://doi.org/10.1007/s12687-022-00605-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12687-022-00605-z