Abstract
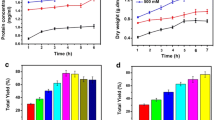
The work presented in this paper focuses on the extraction of keratin from chicken feather waste by the following two methods: (1) alkaline hydrolysis or (2) the reduction by L-cysteine reducing agent; resulting in the extracted yields of 35% and 55%, respectively. The keratin samples produced from the two extraction methods possessed different physical and chemical properties including molecular weight, crystallinity index, thermal behavior, metal binding ability (iron, copper, silver and zinc), antimicrobial activity, and cytotoxicity. The alkaline hydrolysis method yielded keratin of a lower molecular weight, crystallinity index and thermal degradation than the reduction method by L-cysteine. However, their chemical identity was determined to be qualitatively similar as characterized by infrared spectroscopy. The keratin produced by alkaline hydrolysis give rise to a higher metal content adsorbability and thus subsequently led to greater antimicrobial activity with an acceptable value of cytotoxicity. Overall, the work presented a biomaterial from chicken waste with outstanding properties particularly using the alkaline hydrolysis extraction method, which can be applied in a variety of biomedical industries.
Graphical Abstract











Similar content being viewed by others
Data Availability
The datasets generated and analyzed during the current study are not publicly available due to time limitations but are available from the corresponding author on reasonable request.
References
Tesfaye, T., Sithole, B., Ramjugernath, D.: Valorisation of chicken feathers: a review on recycling and recovery route—current status and future prospects. Clean. Technol. Environ. Policy. (2017). https://doi.org/10.1007/s10098-017-1443-9
Brenner, M., Weichold, O.: Protein hydrolysates from biogenic waste as an ecological flame retarder and binder for fiberboards. ACS Omega (2020). https://doi.org/10.1021/acsomega.0c03819
Tamreihao, K., Mukherjee, S., Khunjamayum, R., Devi, L.J., Asem, R.S., Ningthoujam, D.S.: Feather degradation by keratinolytic bacteria and biofertilizing potential for sustainable agricultural production. J. Gen. Microbiol. (2019). https://doi.org/10.1002/jobm.201800434
Suzuki, Y., Tsujimoto, Y., Matsui, H., Kunihiko Watanabe, K.: Decomposition of extremely hard-to-degrade animal proteins by thermophilic bacteria. J. Biosci. Bioeng. (2006). https://doi.org/10.1263/jbb.102.73
Ullah, A., Vasanthan, T., Bressler, D., Elias, A.L., Wu, J.: Bioplastics from feather quill. Biomacromol (2011). https://doi.org/10.1021/bm201112n
Lange, L., Huang, Y., Busk, P.K.: Microbial decomposition of keratin in nature—a new hypothesis of industrial relevance. Appl. Microbiol. Biotechnol. (2016). https://doi.org/10.1007/s00253-015-7262-1
Sinkiewicz, I., Śliwińska, A., Staroszczyk, H., Kołodziejska, I.: Alternative methods of preparation of soluble keratin from chicken feathers. Waste. Biomass. Valorization. (2017). https://doi.org/10.1007/s12649-016-9678-y
Yin, J., Rastogi, S., Terry, A.E., Popescu, C.: Self-organization of oligopeptides obtained on dissolution of feather keratins in superheated water. Biomacromol (2007). https://doi.org/10.1021/bm060811g
Li, Q.: Progress in microbial degradation of feather waste. Front. Microbiol. (2019). https://doi.org/10.3389/fmicb.2019.027
Sillapawattana, P., Wongputtisin, P.: Primary quality assessment of keratin extracted from chicken feather waste as feed component. FAB. J. 9, 51–60 (2021)
Saul, J.M., Ellenburg, M.D., De Guzman, R.C., Dyke, M.V.: Keratin hydrogels support the sustained release of bioactive ciprofloxacin. Part A. J. Biomed. Mater. Res A. (2011). https://doi.org/10.1002/jbm.a.33147
Tran, C.D., Prosenc, F., Franko, M., Benzi, G.: Synthesis, structure and antimicrobial property of green composites from cellulose, wool, hair and chicken feather. Carbohydr. Polym. (2016). https://doi.org/10.1016/j.carbpol.2016.06.021
Feroz, S., Muhammad, N., Ratnayake, J., Dias, G.: Keratin-Based materials for biomedical applications. Bioact. Mater. (2020). https://doi.org/10.1016/j.bioactmat.2020.04.007
Sharma, S., Kumar, A., Adelere, I., Latee, A.: Keratin as a protein biopolymer. Springer, Cham (2019)
Chilakamarry, C.R., Mahmood, S., Saffe, S.N.B.M., Arifin, M.A.B., Gupta, A., Sikkandar, M.Y., Begum, S.S., Narasaiah, B.: Extraction and application of keratin from natural resources: a review. Biotech (2021). https://doi.org/10.1007/s13205-021-02734-7
Oluba, O.M., Obi, C.F., Akpor, O.B., Ojeaburu, S.I., Ogunrotimi, F.D., Adediran, A.A., Oki, M.: Fabrication and characterization of keratin starch biocomposite film from chicken feather waste and ginger starch. Sci. Rep. (2021). https://doi.org/10.1038/s41598-021-88002-3
de Guzman, R.C., Tsuda, S.M., Ton, M.N., Zhang, X., Esker, A.R., Van Dyke, M.E.: Binding interactions of keratin-based hair fiber extract to gold, keratin, and BMP-2. PLoS ONE (2015). https://doi.org/10.1371/journal.pone.0137233
Nikiforova, T., Kozlov, V., Islyaikin, M.: Sorption of d-metal cations by keratin from aqueous solutions. J. Environ. Chem. Eng. (2019). https://doi.org/10.1016/j.jece.2019.103417
Havryliak, V., Mykhaliuk, V., Petrina, R., Fedorova, O., Lubenets, V., Novikov, V.: Adsorbents based on keratin for lead and cadmium removal. Curr. Appl. Sci. Technol. 20(1), 136–143 (2020)
Ma, B., Qiao, X., Hou, X., Yang, Y.: Pure keratin membrane and fibers from chicken feather. Int. J. Biol. Macromol. (2016). https://doi.org/10.1016/j.ijbiomac.2016.04.039
AOAC 2013.06–2013, Arsenic, Cadmium, Mercury, and Lead in Foods.
Hudzicki, J.: Kirby-Bauer disk diffusion susceptibility test protocol. ASM 15, 55–63 (2009)
ISO (2009)-10993–5, 3rd Edition, Biological evaluation of medical devices-Tests for in vitro cytotoxicity.
Helmerhorst, E., Stokes, G.: Generation of an acid-stable and protein-bound persulfide-like residue in alkali or sulfhydryl-treated insulin by a mechanism consonant with the .beta-elimination hypothesis of disulfide bond lysis. Biochem. (1983). https://doi.org/10.1021/bi00270a010
Nashef, A.S., Osuga, D.T., Lee, H.S., Ahmed, A.I., Whitaker, J.R., Feene, R.E.: Effects of alkali on proteins Disulfides and their products. J. Agric. Food Chem. (1977). https://doi.org/10.1021/jf60210a020
Brenner, M., Weichold, O.: Autogenous cross-linking of recycled keratin from poultry-feather waste to hydrogels for plant-growth media. Polymers (2021). https://doi.org/10.3390/polym13203581
Shavandi, A., Bekhit, A.E.D.A., Carne, A., Bekhit, A.: Evaluation of keratin extraction from wool by chemical methods for bio-polymer application. J Bioact. Compat. Polym. (2017). https://doi.org/10.1177/0883911516662069
Tesfaye, T., Sithole, B., Ramjugernath, D.: Valorisation of chicken feather barbs: Utilisation in yarn production and technical textile applications. Sustain. Chem. Pharm. (2018). https://doi.org/10.1016/j.scp.2018.02.002
Idris, A., Vijayaraghavan, R., Rana, U.A., Fredericks, D., Patti, A.F., Macfarlane, D.R.: Dissolution of feather keratin in ionic liquids. Green Chem. (2013). https://doi.org/10.1039/C2GC36556A
Xu, H., Yang, Y.: Controlled de-cross-linking and disentanglement of feather keratin for fiber preparation via a novel process. ACS Sustain. Chem. Eng. (2014). https://doi.org/10.1021/sc400461d
Welu, K.T., Beyan, S.M., Balakrishnan, S., Admassu, H.: Chicken feathers based keratin extraction process data analysis using response surface-box-Behnken design method and characterization of keratin product. Curr. Appl. Sci. Technol, 163–177 (2020).
Cao, J., Joko, K., Cook, J.R.: DSC studies of the melting behavior of α-form crystallites in wool keratin. Text. Res. J. (1997). https://doi.org/10.1177/004051759706700207
Banaszak, L. Metal ions bound to proteins. J. Struct. Biol. 1, (2000).
Xu, H., Xu, D.C., Wang, Y.: Natural indices for the chemical hardness/softness of metal cations and ligands. ACS Omega (2017). https://doi.org/10.1021/acsomega.7b01039
Sutirman, Z.A., Sanagi, M.M., Karim, K.J.A., Naim, A.A., Ibrahim, W.A.W.: Chitosan-based adsorbents for the removal of metal ions from aqueous solutions. Malaysian J. Anal. Sci. 22(5), 839–850 (2018)
Pinho, E., Magalhães, L., Henriques, M., Oliveira, R.: Antimicrobial activity assessment of textiles: standard methods comparison. Ann. Microbiol. (2011). https://doi.org/10.1007/s13213-010-0163-8
Smoot, E., Kucan, J.O., Roth, A., Mody, N., Debs, N.: In vitro toxicity testing for antibacterials against human keratinocytes. Plast. Reconstr. Surg. (1991). https://doi.org/10.1097/00006534-199105000-00017
Ward, R., Hubbard, A., Sulley, H., Garle, M., Clothier, R.: Human keratinocyte cultures in an in vitro approach for the assessment of surfactant-induced irritation. Toxicol. In Vitro. (1998). https://doi.org/10.1016/S0887-2333(97)00098-2
Acknowledgements
This work was supported by the National Research Council of Thailand (NRCT). The authors would like to thank Ms. Pennapa Takam and Mr. Thanawat Inkham for their effort in laboratory works.
Funding
This work was supported by the National Research Council of Thailand (NRCT). Arunee Kongdee Aldred and Panwad Sillapawattana have received the research support from NRCT.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Arunee Kongdee Aldred: conceptualization, methodology, writing original draft, supervision. Prapaipat Klungsupya: methodology, validation. Wasin Charerntantanakul: methodology, writing-reviewing and editing. Oliver Weichlold: conceptualization, validation, writing-reviewing and editing. Panwad Sillapawattana: conceptualization, methodology, data collection and analysis, writing original draft.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kongdee Aldred, A., Klungsupya, P., Charerntantanakul, W. et al. Preparation of Keratin-Metal Complexes Derived from Different Treatments of Chicken Feather Waste. Waste Biomass Valor 15, 115–125 (2024). https://doi.org/10.1007/s12649-023-02154-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-023-02154-z