Abstract
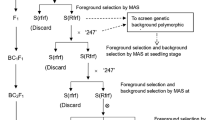
In China, the annual formulation of hybrid combinations in sugar beets is significantly lower than that in foreign countries, primarily due to the limited availability of monogerm sterile and maintainer line pairs. This limitation constrains the production of three-line matching hybrids, resulting in considerably fewer hybrid combinations compared with those developed by foreign breeding companies. Increasing the number of monogerm maintainer lines in sugar beets, as a way to address this limitation, can be achieved by improving these lines with sugar beet multigerm restorer lines. The more the monogerm maintainer lines in sugar beet, the more monogerm sterile lines can be created through backcrossing. Hence, a large number of pairs of monogerm sterile and maintainer lines with excellent multigerm traits can be obtained in sugar beet. This study aimed to pollinate monogerm maintainer lines using multigerm restorer lines in sugar beets. Seeds obtained from these monogerm maintainer lines were collected, and mother roots were obtained through southern propagation. After vernalization and self-pollination in the second year, nonhybrid monogerm plants were eliminated. Molecular marker-assisted selection was used to evaluate the fertility gene composition of 697 sugar beet lines. The monogerm maintainer lines were quickly selected based on the results of molecular marker fertility identification combined with field investigation of morphological and embryological characteristics. The experimental results revealed that out of 697 sugar beet breeding lines, 600 were multigerm lines, accounting for 86.08%, and 97 were monogerm lines, accounting for 13.92%. The fertility identification results of the nuclear Rf1 locus showed that 36 out of 97 monogerm lines contained recessive genes. Therefore, 36 sugar beet monogerm maintainer lines were obtained. The approach of using multigerm restorer lines to improve monogerm maintainer lines, coupled with molecular marker technology and the field investigation of morphological and embryological characteristics, contributed to an increased number of sugar beet monogerm maintainer lines. This method enabled the acquisition of more pairs of sugar beet monogerm sterile lines and maintainer lines, enriching the sugar beet germplasm resource bank and promoting the development of the sugar beet breeding industry.



Similar content being viewed by others
References
Arakawa, T., D. Uchiyama, T. Ohgami, R. Ohgami, T. Murata, Y. Honma, H. Hamada, Y. Kuroda, K. Taguci, K. Kitazaki, and T. Kubo. 2018. A fertility-restoring genotype of beet (Beta vulgaris L.) is composed of a weak restorer-of-fertility gene and a modifier gene tightly linked to the Rf1 locus. PLoS ONE 13 (6): e0198409. https://doi.org/10.1371/journal.pone.0198409.s006.
Coons, G.H., F.V. Owen, and D. Stewart. 1955. Improvement of the sugar beet in the United States. Advances in Agronomy 7: 89–139. https://doi.org/10.1016/S0065-2113(08)60336-3.
Duraisam, R., K. Salelgn, and A.K. Berekete. 2017. Production of beet sugar and bio-ethanol from sugar beet and it bagasse: a review. International Journal of Engineering Trends and Technology (IJETT) 43 (4): 222–233. https://doi.org/10.14445/22315381/IJETT-V43P237.
Fu, Z.J., C. Bai, H. Zhang, Y.Y. E, Z.Q. Zhang, X.D. Li, S.M. Zhao, L. Wang, W.Z. Zheng, B.Z. Zhang, and H.Z. Zhang. 2022. Selection and application of monogerm male sterile hybrid NT39106 in sugar beet. Bulletin of Agricultural Science and Technology 08: 223–227 (In Chinese).
Hagihara, E., N. Itchoda, Y. Habu, S. Iida, T. Mikami, and T. Kubo. 2005. Molecular map** of a fertility restorer gene for Owen cytoplasmic male sterility in sugar beet. Theoretical and Applied Genetics 111: 250–255. https://doi.org/10.1007/s00122-005-2010-z.
Jung, B., F. Ludewig, A. Schulz, G. Meissner, N. Woestefeld, U.I. Fluegge, B. Pommerrenig, P. Wirsching, N. Sauer, W. Koch, F. Sommer, T. Muhlhaus, M. Schroda, T.A. Cuin, D. Graus, I. Marten, R. Hedrich, and H. EkkehardNeuhaus. 2015. Identification of the transporter responsible for sucrose accumulation in sugar beet taproots. Nature Plants 1 (1): 14001. https://doi.org/10.1038/nplants.2014.1.
Liang, X.M., W.L. Kan, Z. Pi, and Z.D. Wu. 2024. Effect of RSAP primer screening and different detection methods on polymorphism of amplified products in sugar beet. Molecular Plant Breeding 1–8. https://kns.cnki.net/kcms2/detail/46.1068.S.20230705.1229.006.html(In Chinese).
McGrath, J.M., and B.J. Townsend. 2015. Sugar beet, energy beet, and industrial beet. Industrial Crops: Breeding for Bioenergy and Bioproducts. https://doi.org/10.1007/978-1-4939-1447-0_5.
Mikami, T., P.Y. Masayuki, H. Matsuhira, K. Kitazaki, and T. Kubo. 2011. Molecular basis of cytoplasmic male sterility in beets: An overview. Plant Genetic Resources 9 (2): 284–287. https://doi.org/10.1017/S1479262111000177.
Moritani, M., K. Taguchi, K. Kitazaki, H. Matsuhira, T. Katsuyama, T. Mikami, and T. Kubo. 2013. Identification of the predominant nonrestoring allele for owen-type cytoplasmic male sterility in sugar beet (Beta vulgaris L.): development of molecular markers for the maintainer genotype. Molecular Breeding 32 (1): 91–100. https://doi.org/10.1007/s11032-013-9854-8.
Pathak, A.D., R. Kapur, S. Solomon, R. Kumar, S. Srivastava, and P.R. Singh. 2014. Sugar beet: a historical perspective in Indian context. Sugar Tech 16: 125–132. https://doi.org/10.1007/s12355-014-0304-7.
Peng, F., S.N. Li, Z. Pi, and Z.D. Wu. 2023. Identification of molecular markers for fertility in sugar beet. Sugar Tech. https://doi.org/10.1007/s12355-023-01275-y.
Shi, H.Q. 2021. A research on the use of molecular markers technology to identification the fertility of sugar beet germplasm resources. China: Heilongjiang University. https://doi.org/10.27123/d.cnki.ghlju.2021.001568. (In Chinese).
Shi, H.Q., Z.D. Wu, W. **ng, L.H.Z. Ding, and Z. Pi. 2021a. Analysis of the fertility composition of sugar beet multigerm germplasm resources by molecular markers. Sugar Crops of China 43 (4): 26–31. https://doi.org/10.13570/j.cnki.scc.2021.04.004. (In Chinese).
Shi, H.Q., Z. Pi, and Z.D. Wu. 2021b. Different molecular markers to identify the fertility of sugar beet monogerm germplasm resources. Research Square. https://doi.org/10.21203/rs.3.rs-230220/v1.
Stevanato, P., C. Chiodi, C. Broccanello, G. Concheri, E. Biancardi, O. Pavli, and G. Skaracis. 2019. Sustainability of the sugar beet crop. Sugar Tech 21: 703–716. https://doi.org/10.1007/s12355-019-00734-9.
Yan, C.Y., Y. Zou, Z.D. Wu, W. **ng, and H.X. Li. 2018. A method for large-scale rapid extraction of DNA from sugar beet. Sugar Crops of China 40 (06): 44–46. https://doi.org/10.13570/j.cnki.scc.2018.06.013. (In Chinese).
Ye, L.H. 2014. Development of CAPS molecular markers based on ipal gene and assisted selection breeding. Nanchang: Jiangxi Agricultural University (In Chinese).
Yv, C., G.L. Li, Y.Q. Sun, N.N. Li, and S.Y. Zhang. 2023. Characteristics of respiratory metabolism in growth and development of sugar beet taproot. Acta Agronomica Sinica 1–12. https://kns.cnki.net/kcms2/detail/11.1809.S.20230526.1609.004.htm..
Zhang, Y., Y.D. Ning, X. Lan, C.X. Yv, S.B. An, H.Y. Zhang, Y.H. Zhang, Y.S. Shi, and Y.X. Guo. 2023. Present situation and prospect of sugar beet breeding industry development in China. Sugar Crops of China 45 (01): 8–13. https://doi.org/10.13570/j.cnki.scc.2023.01.002. (In Chinese).
Funding
This study was supported by the Ministry of Finance and the Ministry of Agriculture and Rural Affairs of China—National Modern Agricultural Industry Technology System (CARS-170111).
Author information
Authors and Affiliations
Contributions
ZW and LS participated in the conceptualization and design of the study. JZ and YZ were involved in material preparation, data collection, and analysis. JZ wrote the first draft of the manuscript. JZ, ZW, and SL revised the manuscript, and ZW, SL, and JZ commented on a previous version of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, JJ., Li, SN., Wu, ZD. et al. Application of Multigerm Restorer Lines for Improving Monogerm Maintainer Lines in Sugar Beet. Sugar Tech (2024). https://doi.org/10.1007/s12355-024-01376-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12355-024-01376-2